PROXIMATE COMPOSITION AND FATTY ACID PROFILES OF SELECTED FISH SPECIES FROM PAKISTAN
M. Ahmed1,2, M. Liaquat1, A. S. Shah3, I. B. Abdel-Farid4,5 and M. Jahangir1*
1Department of Food Science and Technology, The University of Haripur, Khyber-Pakhtunkhwa, Pakistan
2Khyber-Pakhtunkhwa Food Safety and Halal Food Authority, Abbottabad, Pakistan
3Khyber-Pakhtunkhwa Food Safety and Halal Food Authority, Peshawar, Pakistan
4Department of Botany, Faculty of Science, Aswan University, Aswan 81528, Egypt
5Department of Biology, Faculty of Science, Aljouf University, Sakaka, Aljouf, KSA
*Corresponding author E-mail address: mj_awan@hotmail.com
ABSTRACT
In present study, the economically most important fish species, namely Mahseer (Tor putitora), Silver carp (Hypophthalmichthys molitrix), Common carp (Cyprinus carpio), Thela fish (Catla catla) and Rainbow trout (Oncorhynchus mykiss) from Pakistan, were evaluated for their proximate compositions and fatty acid profiles. All of the analyzed fish were collected from the Northwestern regions (Khyber-Pakhtunkhwa) of Pakistan. Proximate analysis was done by using standard AOAC methods, whereas the detailed fatty acid composition was evaluated by using gas liquid chromatography. A significant difference (p< 0.05) was noticed for various proximate parameters and fatty acids profiles among fish species. Palmatic acid (C16:0) was the copious fatty acid of each fish species, with a range from 16.7% to 24.6%. Furthermore, all fish species were found as good sources of polyunsaturated fatty acids (PUFAs) including alpha linolenic acids (ALA C18:3), arachidonic acids (AA C20:4), eicosapentaenoic (EPA C20:5) and docosahexaenoic acids (DHA C22:6) and protein. Whereas, Rainbow trout ranked highest in PUFAs while Mahseer proved to have highest protein contents.
Keywords: Fish composition, Fatty acids profile, Omega-3/n-3fatty acids, Polyunsaturated fatty acids.
https://doi.org/10.36899/JAPS.2020.4.0102
Published online April 25, 2020
INTRODUCTION
Although Pakistan has a low per capita (i.e. 2 kg) fish consumption, still during 2016-17 the fish production was estimated as 520,000 million tons, where 103,277 million tons has been exported (GoP, 2017). Furthermore, there is a promising value (i.e. $ 266 millions) of export from fish and fishery products that is estimated to be 1.2 % of total merchandise trade of Pakistan, as reported previously (FAO, 2009). Among others, the Mahseer, Silver carp, Common carp, Thela and Rainbow trout are commonly grown, having good economic value and well reputed among consumers and producers, in Pakistan (FAO, 2005).
Fish is a main source of protein, vitamins, minerals, and polyunsaturated fatty acids (PUFAs) including omega-3 fatty acids. Among other constituents, especially the fish lipids are considered as healthy diet for human consumption all over the world (Erkan and Ozkan, 2007; Hossain, 2011). The lipid fraction of fish mainly consists of PUFAs, including eicosapentaenoic acid (n-3 C20:5EPA), docosapentaenoic acid (n-3 C22:5DPA) and docosahexaenoic acid (n-3 C22:6 DHA) (Strobel et al., 2012). The health promoting effects of PUFAs (especially omega-3 fatty acids) have reported benefits for human metabolic and biological functions (Kocatepe and Turan, 2012), which have been linked through the eicosanoids synthesis process, such as thromboxanes, leukotrienes and prostaglandins into anti-thrombotic prostaglandins (Inhamuns and Franco, 2008; Calder, 2013). Furthermore, the benefits of PUFAs includes coronary artery disease avoidance (by enhancement in endothelial functions), lessen breast cancer & prostate cancer risks and reduce depressive symptoms in parkinsonian patients (Park et al., 2009; Emsley et al., 2008; Kaur et al., 2009; Shannon et al., 2010).
Although there is variability in the recommendations of WHO and FAO for the intake of n-6/n-3, with a ratio of 20:1 to 30:1 for various communities around the world, but it is recommended as 5:1 in western diets, so this value can be taken as standard (Lunn and Theobald, 2006). General suggestion for daily intake of DHA (n-3 C22:6) to EPA (n-3 C20:5) is 0.5g for toddlers and 1g for the patients of heart disease and adults (Hossain, 2011). A diet is considered as unbalanced, if it includes excessive amount of omega-6 PUFAs, along with small amount of omega-3 PUFAs (Kris-Etherton et al., 2002). The large amount of n-3 long chain (LC) PUFAs of aquatic fish oils depend on fatty acid composition of their normal diet. The fatty acid contents of fish lipids depend on several factors, including the variation due to fish species, nutrition of fish, environmental condition, temperature and geographical location (Kocatepe and Turan, 2012; O¨zogul and O¨zogul, 2007). Similarly, the PUFA's compositions of various fish oils might also differ but little interest has been paid to this specific subject, when selecting fish species for human diet or processing for product development (Osman et al., 2001).
Although, there are several research studies available on fatty acid profiles of a range of fish types from several geographical areas but here is the first published report for the proximate analysis and fatty acid profiles of Mahseer (Tor putitora), Silver carp (H. molitrix), Common carp (C. carpio), Thela fish (C. catla) and Rainbow trout (O. mykiss) from the northwestern region (Khyber-Pakhtunkhwa) of Pakistan.
MATERIALS AND METHODS
Collections of fish samples: Fresh fish samples were purchased from the fishermen at the site of harvesting. The Mahseer, Silver carp, Common carp, and Thela fish were collected from Tarbeela lake (Khalabat Township, District Haripur, Khyber-Pakhtunkhwa, Pakistan), whereas Rainbow trout (900 g + 100 g) were purchased from the collection point of river Swat, District Swat, Khyber-Pakhtunkhwa, Pakistan). The fish samples having similar weight (2 kg + 100 g) were selected and instantaneously stocked up in a cooler filled with dry ice in order to limit the biological processes, and transported to the Foodomics laboratory, Department of Food Science & Technology, The University of Haripur. Fish were de-scaled, washed and dissected to get various organs (head, tale, viscera) separated before further proximate analysis and fatty acid profile of fleshy tissue.
Fish proximate composition analysis: The proximate analyses were done according to the standard procedures as reported previously (AOAC, 1990). Percent moisture content was determined by drying the fish samples (100 g / sample) in an oven at 105 ºC, until a stable weight. The ash content was determined by incineration of samples in a muffle furnace at 550 ºC for 10 hours. Crude lipids (F) content were determined by Soxhlet extraction and crude proteins (CP) content was determined by the Kjeldahl method (N% x 6.25). The carbohydrate (C) value of fish was calculated by the formula [%Carbohydrate value = 100 - (% Moisture + % Ash + % Crude protein + % Crude lipid)] (Jabeen and Chaudhry, 2011). The energy value of each ingredient in fish sample was determined by multiplications of the percentages (%) of crude fat (F), total carbohydrate (C) and crude protein (CP) contents with their respective energy values of 9, 4 and 4kcal/100g, as per bellow mentioned formula (Jabeen and Chaudhry, 2011):
Caloric value (CV) = (9F +4C+ 4CP) kcal / 100g weight
Fatty acid analysis: Fatty acids composition was analyzed by gas liquid chromatography (Gas-LC) of fatty acid methyl esters, as previously reported (Pickova et al., 1997).
Statistical analysis: Principal component analysis (PCA) was performed (for fatty acid profile of fish) with SIMCA-P+ software (v. 12.0.1, Umetrics, Umeå, Sweden) by using unit variance (UV) scaling method, as previously reported by Jahangir et al., 2014. Further, statistical analysis was performed for proximate parameters and fatty acid profile, by using the Statistica 8 (version 8.1) software, where completely randomize design (CRD) was used to analyze the significance for differences between mean compositions in the different fish types and means were separated out by LSD. A significance level of 0.05 was applied for this analysis.
RESULTS AND DISCUSSION
Proximate composition of five fish species under this research (Table 1), proved to have significant difference in moisture (70.22– 77.69 %) with lowest in Mahseer and highest in Thela fish, ash on wet weight bases (0.95 % – 1.42 %) with lowest for Common carp and highest for Rainbow trout, carbohydrate (0.94 – 5.88%) with lowest for Thela and highest for Common carp, crude protein (18.5 – 22.2%) with lowest for Silver carp and highest for Mahseer and crude fat 1.20–3.26 % with lowest in Thela fish and highest in Common carp. As fish species are classified on the bases of their body fat, where the lean fish species contain fat lesser than 5% (Jabeen and Chaudhry, 2011), so we can categorize these fish as lean fish species (Bennion and Scheule, 2003).
Fish species with less fat content always contain high water content and their fleshy tissue is white in color because it stores 50 to 80 % of its fat in the liver as compared to muscles (Osman et al., 2001). On the basis of above proximate analysis of crude fat, carbohydrate and protein, the caloric value of the fish samples was calculated, that proves highly significant different (p<0.01), with a range from 90.9 - 130.1 kcal/100g for different species (Table 1).
Fatty acids; such as myristic acid C14:0, palmatic acid C16:0, palmitoleic acid C16:1 n-7, stearic acid C18:0, oleic acid C18:1n-9, linoleic acid C18:2 n-6, alpha-linolenic acid ALA C18:3 n-3, eicosapentaenoic acid EPA C20:5n-3 and docosahexaenoic acid DHA C22:6 n-3 were reported in various proportions among different fish species (Bayir et al.,2006; O¨zogul et al., 2007; 2009; Memon et al., 2011; Usydus et al., 2011). Similarly, in our findings, the SFAs were found variant in all fish samples. Myristic acid was 1.89% as least in Common carp and 4.80% as highest in Mahseer, whereas stearic acid was observed 0.08% as least in Mahseer and 4.77% as highest Thela fish, among all analyzed fish types. The highest palmitic acid content (24.6%) was noted in the Mahseer, while the lowest value (16.7%) was observed in Common carp.
The saturated fatty acids (SFAs) contents ranged from 23.6 – 33.3% with lowest for Common carp and highest for Mahseer, monounsaturated fatty acids (MUFAs) 18.8 – 39.1% with lowest for Silver carp and highest for Common carp and poly unsaturated fatty acids (PUFAs) ranged 15.2–24.6% with lowest for Silver carp and highest for Rainbow trout (Table 2; Figure 2). Inter-comparison of fatty acids profile among fish species, by using principal component analysis (PCA), proves a similarity of fatty acids in Rainbow trout, Common carp and Thela fish as in one group, while Mahseer and Silver carp are discriminated from all other species (Figure 3) in principal component (PC 1), whereas these two are different from each other in PC 2. The discrimination of Silver carp and Mahseer from other fish species (namely Common carp, Rainbow trout and Thela) is generally due to the high percentage of C14:0 Myristic, ∑n-3, n-3/n-6, docosanoic acid (C22:0), lignoceric acid (C24:0), eicosenoic acid (C20:1(n-9)), nervonic acid (C24:1), γ-linolenic acid (C18:3(n-6)), α-linolenic acid (C18:3(n-3)), arachidonic acid (C20:4(n-6)), eicosapentaenoic acid (C20:5(n-3)) and docosapentanoic acid (C22:5(n-3)), whereas in specific for Silver carp higher contents of SFAs, heptadecanoic acid (C17:0), erucic acid (C22:1(n-9)), eicosadienoic acid (C20:2(n-6)), palmitic acid (C16:0), arachidic acid (C20:0) was higher, while on the other hand the docosahexaenoic acid (C22:6 (n-3)) was higher for Masheer (Figure 2).
On the other hand the principal component containing the Common carp, Rainbow trout and Thela species has proven the discrimination of these species from Silver carp and Mahseer due to higher contents of the overall ∑MUFAs, ∑PUFAs in general, whereas in specific higher amount of DHA/EPA, lauric acid (C12:0), pentadecanoic acid (C15:0), stearic acid (C18:0), myristoleic acid (C14:1), palmitoleic acid (C16:1 (n-9)), heptadecanoic acid (C17:1 cis-10), elaidic acid (C18:1(n-7)), oleic acid (C18:1(n-9)), linoleic acid (C18:2(n-6)), eicosatrienoic acid (C20:3 (n-6)) and eicosatrienoic acid (C20:3(n-3)) (Figure 2).
All though there were similarities as observed grouping in principal component analysis, the MUFAs values were still found different in all five analyzed fish samples. The maximum MUFAs were present in Common carp (39.1%), whereas the lowest quantity was found in the Silver carp (18.8%) among all samples. The oleic acid was most prominent fatty acid ranged from 0.35 – 35.05% of the entire MUFAs. The highest oleic acid content was observed in Common carp (35.05%), whereas the lowest value was found in the Thela fish (0.35%). The highest palmitoleic acid (C16:1 n-9) value was obtained in Thela fish (5.97%), while the lowest value was found in the rainbow trout (0.07%). Palmitoleic acid (C16:1, n-9) and oleic acid (C18:1, n-9) have been reported as the major MUFAs in our findings, which is in line with the muscles tissues from the Mazurian great lakes (Luczynska et al., 2008), marine fish species from Turkish waters (Bayir et al., 2006) and anchovy (Kaya and Turan, 2008).
The PUFAs values were also different among all five analyzed fish. The highest PUFAs content were found in Rainbow trout (24.6%), whereas the lowest value was noticed in the Silver carp (15.2%). The foremost fatty acids distinguished as PUFAs were alpha-linolenic acid, eicosapentanoic acid and docosahexaneoic acid respectively. The highest proportion of DHA in total PUFAs was recorded from the Mahseer (5.25%), whereas, the Common carp contained lower DHA contents (0.49%). This study proves that the selected five fish species generally contains considerable amount of n-3 PUFAs contents, with high level of linoleic acid in Rainbow trout, Thela fish and Common carp (Table 2). The superior content of linoleic acid within the fish based upon the diet and the capacities of desaturation and elongation of carbon chain (Alasalvar et al., 2002).
All these five fish species contained lower level of arachidonic acid (AA, C20:4 n-6), that is a precursor intended for biosynthesis of series-2 eicosanoids such as thromboxane and prostaglandin (Pompeia et al., 2002). The arachidonic acid is important for the clotting process of blood, attached to endothelial cells through wound curing and takes part a vital role in the retina, brain and growth of infants (Shen et al., 2007). Maximum amount of the arachidonic acid among five fish species was found in Rainbow trout 1.1%, followed by Silver carp 0.7% and the least amount of this important constituent was observed in Common carp 0.1% (Table 2). In this study % DHA is higher than % EPA (Table 2; Figure 2). The proportional ratio between omega-3 PUFAs and omega-6 PUFAs is a valuable gauge for determining the dietetic value of fish oil and generally consumption of 0.4g per day of n-3 PUFAs decreases the number of deaths in the patients suffering from coronary heart disease (Ozyurt and Polat, 2006). It was suggested by Zuraini et al., (2006) that the proportional ratio of 1:1 to 1:5 is suitable in vigorous diet for human. The proportional ratio of n-3: n-6 PUFAs was found higher in Mahseer followed by Silver carp while the ratio for the Common carp found lowest among all fish species. The total percentage n-3 PUFAs is less than that of n-6 PUFAs, which is similar to previous findings for other fish species (Jabeen and Chaudhry, 2011; Ugoala et al., 2009). Fresh water fish species have been mostly characterized by the prominent levels of omega-6 PUFAs, specifically linoleic acid and arachidonic acids (Aras et al., 2003). All selected fish species were found rich in linoleic acid (LA, n-6 C18:2) content except Silver carp, which is essential fatty acid that cannot be synthesized in the human body and must be taken in diet and is required for the development of tissues. The analyzed fish species are found to be healthy diet for human consumption, where it is very interesting that Rainbow trout which is very expensive has similar fatty acid profile with Common carp and Thela fish, which are comparatively available in lower cost.
Table 1. Chemical composition (on wet bases) of muscles of selected fish species from Tarbela lake and river Swat (Mean value ± SD).
|
Variables
|
Mahseer
|
Rainbow Trout
|
Silver Carp
|
Thela Fish
|
Common Carp
|
|
Moisture (%)
|
70.2c ± 0.59
|
72.6b ± 0.38
|
77.1a ± 0.53
|
77.7a ± 0.21
|
70.6c ± 0.36
|
|
Ash (%)
|
1.2abc ± 0.17
|
1.4a ± 0.24
|
1.3ab ±0.17
|
1.1bc ± 0.09
|
1.0c ± 0.03
|
|
Crude protein (%)
|
22.2a ± 0.10
|
21.8a ± 0.45
|
18.5c ±0.16
|
19.1b ± 0.13
|
19.3b ± 0.36
|
|
Crude fat (%)
|
1.4c ± 0.01
|
2.6b ± 0.05
|
1.4c ± 0.09
|
1.2d ± 0.00
|
3.3a ± 0.08
|
|
Total carbohydrates (%)
|
5.0a ± 0.74
|
1.6b ± 0.89
|
1.7b ± 0.57
|
0.9b ± 0.32
|
5.9a ± 0.05
|
|
Caloric value (kcal/100g)
|
119.7b± 5.14
|
116.8b ±2.71
|
93.7c ±2.99
|
90.9c ± 0.88
|
130.1a ± 1.85
|
Levels of significance = 0.05; SD = Standard Deviation
Similar alphabets represent non-significant differences
Table 2. Fatty acid composition (%) of selected fish species from Tarbela lake and river Swat.
|
Name of sample
|
Mahseer
|
Rainbow Trout
|
Silver Carp
|
Thela Fish
|
Common Carp
|
|
C12:0, Lauric
|
0.1a ± 0.01
|
0.1z ± 0.01
|
0.2y ± 0.01
|
0.1y ± 0.01
|
0.8o ± 0.01
|
|
C14:0, Myristic
|
4.8j ± 0.01
|
3.1l ± 0.01
|
4.0j ± 0.01
|
3l ± 0.01
|
1.9l ± 0.01
|
|
C15:0, Pentadecanoic
|
0.5t ± 0.01
|
0.3u ± 0.01
|
0.1b ± 0.01
|
0.4r ± 0.01
|
0.5q ± 0.01
|
|
C16:0, Palmitic
|
24.6c ± 0.01
|
17.7i ± 0.01
|
18d ± 0.01
|
18.2g ± 0.01
|
16.7g ± 0.01
|
|
C17:0, Heptadecanoic
|
0.8s ± 0.01
|
0.1a ± 0.01
|
0.4v ± 0.01
|
0.7q ± 0.01
|
0.1y ± 0.01
|
|
C18:0, Stearic
|
0.1a ± 0.01
|
4.5j ± 0.01
|
0.8r ± 0.01
|
4.8j± 0.01
|
3.5i ± 0.01
|
|
C20:0, Arachidic
|
1.3p ± 0.01
|
0.5s ± 0.01
|
0.03c ± 0.01
|
0.01b ± 0.01
|
0.03z ± 0.01
|
|
C22:0, Docosanoic
|
0.1z ± 0.01
|
0.3v ± 0.01
|
1.8n ± 0.01
|
0.3u ± 0.01
|
0.1vw ± 0.01
|
|
C24:0, Lignoceric
|
1.1q ± 0.01
|
0.2x ± 0.01
|
1.2o ± 0.01
|
0.4s ± 0.01
|
0.1vw ± 0.01
|
|
∑SFAs
|
33.3b ± 0.01
|
26.9d ± 0.01
|
26.5b ± 0.01
|
27.9d ± 0.01
|
23.6d ± 0.01
|
|
C14:1, Myristoleic
|
0.1y ± 0.01
|
0.1a ± 0.01
|
0.2y ± 0.01
|
0.1y ± 0.01
|
0.5p ± 0.01
|
|
C16:1(n-9), Palmitoleic
|
0.3w ± 0.01
|
0.1ab ± 0.01
|
0.3w ± 0.01
|
6i ± 0.01
|
0.2t ± 0.01
|
|
C17:1, cis-10 Heptadecanoic
|
0.1b ± 0.01
|
0.2x ± 0.01
|
0.03c ± 0.01
|
0.01b ± 0.01
|
0.1x ± 0.01
|
|
C18:1(n-7), Elaidic
|
2.6m ± 0.01
|
2.6n ± 0.01
|
3.4k ± 0.01
|
28.4c ± 0.01
|
2.6 j± 0.01
|
|
C18:1(n-9), Oleic
|
18e ± 0.01
|
29.8c ± 0.01
|
13.5f ± 0.01
|
0.4t ± 0.01
|
35c ± 0.01
|
|
C20:1(n-9), Eicosenoic
|
0.1b ± 0.01
|
0.2y ± 0.01
|
0.2z ± 0.01
|
0.1za ± 0.01
|
0.04yz ± 0.01
|
|
C22:1(n-9), Erucic
|
1.6o ± 0.01
|
0.9q ± 0.01
|
0.1b ± 0.01
|
0.1a ± 0.01
|
0.4s ± 0.01
|
|
C24:1, Nervonic
|
0.2w ± 0.01
|
0.4t ± 0.01
|
1.1p ± 0.01
|
0.02b ± 0.01
|
0.3t ± 0.01
|
|
∑MUFAs
|
23d ± 0.01
|
34.3b ± 0.01
|
18.8c ± 0.01
|
35b ± 0.01
|
39.1b ± 0.01
|
|
C18:2(n-6), Linoleic
|
4.8j ± 0.01
|
18.3h ± 0.01
|
0.02c ± 0.01
|
17.1h ± 0.01
|
15.3h ± 0.01
|
|
C18:3(n-6), γ-linolenic
|
2.9k ± 0.01
|
0.8r ± 0.01
|
7.9h ± 0.01
|
1.1p ± 0.01
|
1.6m ± 0.01
|
|
C18:3(n-3), α-linolenic (ALA)
|
0.1x ± 0.01
|
0.3w ± 0.01
|
0.3x ± 0.01
|
0.01b ± 0.01
|
0.1x ± 0.01
|
|
C20:2(n-6), Eicosadienoic
|
0.2w ± 0.01
|
0.1b ± 0.01
|
0.1b ± 0.01
|
0.1a ± 0.01
|
0.1v ± 0.01
|
|
C20:3(n-6), Eicosatrienoic (hGL)
|
0.2w ± 0.01
|
1p ± 0.01
|
0.02c ± 0.01
|
0.01b ± 0.01
|
0.5r ± 0.01
|
|
C20:3(n-3), Eicosatrienoic
|
0.1b ± 0.01
|
0.1z ± 0.01
|
0.1a ± 0.01
|
0.1x ± 0.01
|
0.1w ± 0.01
|
|
C20:4(n-6), Arachidonic
|
0.3v ± 0.01
|
1.1o ± 0.01
|
0.7t ± 0.01
|
0.1z ± 0.01
|
0.1x ± 0.01
|
|
C20:5(n-3), Eicosapentaenoic (EPA)
|
1.9n ± 0.01
|
0.1z ± 0.01
|
3l ± 0.01
|
1.2o ± 0.01
|
1.1n ± 0.01
|
|
C22:5(n-3), Docosapentanoic (DPA)
|
0.4u± 0.01
|
0.2x ± 0.01
|
0.4u ± 0.01
|
0.2w ± 0.01
|
0.2u ± 0.01
|
|
C22:6(n-3), Docosahexaenoic (DHA)
|
5.3i ± 0.01
|
2.8m ± 0.01
|
2.7m ± 0.01
|
2.6m ± 0.01
|
0.5q ± 0.01
|
|
∑PUFAs
|
16.2f ± 0.01
|
24.6f ± 0.01
|
15.2e ± 0.01
|
22.5e ± 0.01
|
19.5e ± 0.01
|
|
∑n-3
|
7.7h ± 0.01
|
3.5k ± 0.01
|
6.5i ± 0.01
|
4.1k ± 0.01
|
2k ± 0.01
|
|
∑n-6
|
8.5g ± 0.01
|
21.2g ± 0.01
|
8.7g ± 0.01
|
18.4f ± 0.01
|
17.6f ± 0.01
|
|
n-3/n-6
|
0.9r ± 0.01
|
0.2y ± 0.01
|
0.7s ± 0.01
|
0.2v ± 0.01
|
0.1vw ± 0.01
|
|
DHA/EPA
|
2.8l ± 0.01
|
26.7e ± 0.01
|
0.91q ± 0.01
|
2.3n ± 0.01
|
0.4s ± 0.01
|
|
Total
|
72.5a ± 0.01
|
85.8a ± 0.01
|
60.5a ± 0.01
|
85.4a ± 0.01
|
82.3a ± 0.01
|
Levels of significance = 0.05; SD = Standard Deviation
Similar alphabets represent non-significant differences
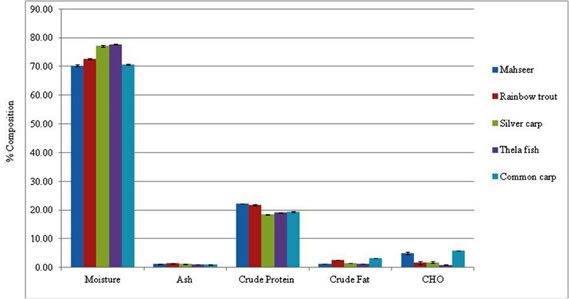
Figure 1. Proximate composition of selected fish species ( Mahseer, Rainbow Trout, Silver carp, Thela, Common carp).

Figure 2. Principal component analysis loading bi plot of fatty acid profile of selected fish species. M = Mahseer, R = Rainbow trout, S = Silver carp, T = Thela Fish, C = Common carp, 1 =C12:0 Lauric,2 = C14:0 Myristic, 3 = C15:0 Pentadecanoic, 4 = C16:0 Palmitic, 5 = C17:0 Heptadecanoic, 6 =C18:0 Stearic, 7 = C20:0 Arachidic, 8 = C22:0 Docosanoic, 9 =C24:0 Lignoceric, 10 = ∑SFAs, 11 = C14:1 Myristoleic, 12= C16:1 (n-9) Palmitoleic, 13 = C17:1 cis-10 Heptadecanoic, 14 = C18:1(n-7) Elaidic, 15 = C18:1(n-9) Oleic, 16 = C20:1(n-9) Eicosenoic, 17 = C22:1(n-9) Erucic, 18 = C24:1 Nervonic, 19 = ∑MUFAs, 20 = C18:2(n-6) Linoleic, 21 = C18:3(n-6) γ-linolenic, 22 =C18:3(n-3) α-linolenic (ALA), 23 = C20:2(n-6) Eicosadienoic, 24 = C20:3(n-6) Eicosatrienoic (hGL), 25 = C20:3(n-3) Eicosatrienoic, 26 = C20:4(n-6) Arachidonic, 27 = C20:5(n-3) Eicosapentaenoic (EPA), 28 = C22:5(n-3) Docosapentanoic (DPA), 29 = C22:6(n-3) Docosahexaenoic (DHA), 30 = ∑PUFAs, 31 = ∑n-3, 32 = ∑n-6, 33 = n-3/n-6, 34 = DHA/EPA, 35 = Total.
Conclusion: To date no authentic report on the chemical compositions and fatty acids profiles of local Pakistani selected fish species exists. This research reports the proximate composition and fatty acid profiles of five most commonly used local fish species that serves as a data base for aquaculture reports and will be authentic source of information for human diet leading to health benefit. All fish species, in our finding, contain important fatty acids, specifically ALA, EPA and DHA which are valuable for good human health and fitness. The fish species contained substantial amount of omega-3 PUFAs that suggests fish species, especially Rainbow Trout, as a good resource of vigorous diet for human beings. Findings of current research shows that Rainbow trout and Thela fish are an excellent source of PUFAs and total fatty acids, whereas Mahseer and Rainbow Trout were found to be an excellent source of protein, with a low level of fat. Interesting similar profiles of other fish species with that of Rainbow trout makes valuable information to consumers, as with lower cost similar level of diet can be opted.
Conflict of interest: The authors do not have conflict of interest in this research work.
Acknowledgements: The technical support (for proximate analysis) of Taufiq Ahmad, from the department of Food Technology at Nuclear Institute for Food and Agriculture (NIFA), Peshawar, Pakistan, and financial support of Higher Education Commission (HEC), Pakistanis gratefully acknowledged. Furthermore, Prof. Dr. Jana Pickova, Department of Food Science, Swedish University of Agricultural Sciences, Sweden is also acknowledged for the analysis of fatty acids.
REFERENCES
- Alasalvar, C., K.D.A. Taylor, E. Zubcov, F. Shahidi, and M. Alexis (2002). Differentiation of cultured and wild sea bass (Dicentrarchus labrax): total lipid content, fatty acid and trace mineral composition. Food. Chem. 79: 145-150.
- AOAC, (1990). Association of official analytical chemists. 15th(edn) Procedure 984.25
- Aras, N.M., H.I. Halilog?lu, H. Yetim and Ö. Ayik (2003). Comparison of fatty acid profiles of different tissues of mature trout (Salmo trutta labrax, Pallas, 1811) caught from Kazandere Creek in the Oruh Region, Erzurum, Turkey. J. Vet. Anim. Sci.,27: 311–316.
- Bayir, A., H.I. Halilog?lu, A.N. Sirkeciog?lu and N.M. Aras (2006). Fatty acid composition in some selected marine fish species living in Turkish waters. J. Sci. Food Agric., 86:163–168.
- Bennion, M. and B. Scheule (2003). Introductory foods. 12th Prentice Hall, New York. 145 p
- Calder, P.C. (2013). Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology?. Br J Clin Pharmacol, 75: 645-662.
- Emsley, R., D.J. Niehaus, P.P. Oosthuizen, L. Koen, B.Ascott-Evans, B. Chiliza, S.J. Van Rensburg and R.M. Smit (2008). Safety of the omega-3 fatty acid, eicosapentaenoic acid (EPA) in psychiatric patients: results from a randomized, placebo-controlled trial. Psych Res 161:284–291.
- Erkan, N. and O¨. O¨zkan (2007). Proximate composition and mineral contents in aqua culturedsea bass (Dicentrarchus Labrax), sea bream (Sparusaurata) analyzed by ICP-MS. Food Chem, 102:721–725.
- FAO (Food and Agriculture Organization) (2005). National Aquaculture Sector Overview, Pakistan. Country Profile Fact Sheets. Text by Hayat, M. In: FAO Fisheries and Aquaculture Department. Rome.
- FAO (Food and Agriculture Organization) (2009). Fishery and Aquaculture Country Profiles. Pakistan, Country Profile Fact Sheets. In: FAO Fisheries and Aquaculture Department [online]. Rome. Updated 1 December 2017. http://www.fao.org/fishery/
- GoP (Government of Pakistan) (2017). Pakistan Economic survey report (2016 – 17). Chapter 2, p.19 – 40.
- Hossain, M.A. (2011). Fish as source of n-3 polyunsaturated fatty acids (PUFAs), Which one is better-farmed or wild?. Adv J Food Sci. Technol, 3: 455-466.
- Inhamuns, A.J. and M.R.B. Franco (2008). EPA and DHA quantification in two species of fresh water fish from Central Amazonia. Food Chem 107: 587–591.
- Jabeen, F. and A.S. Chaudhry (2011). Chemical compositions and fatty acid profiles of three fresh water fish species. Food Chem, 125: 991–996.
- Jahangir, M., I.B. Abdel-Farid, C.H.R. de. Vos, H.H. Jonker, Y.H. Choi and R. Verpoorte (2014). Metabolomic variation of Brassica Rapa rapa (var. raapstelen) and Raphanus sativus L. at different developmental stages. Pak J Bot 46: 1445-1452.
- Kaur, B., A. Jørgensen and A.K. Duttaroy (2009). Fatty acid uptake by breast cancer cells (MDA-MB-231): effects of insulin, leptin, adiponectin, and TNFa. Prosta Leuko and Ess Fatty Acids, 80: 93–99.
- Kaya, Y. and H. Turan (2008). Comparison of protein, lipid and fatty acids composition of anchovy (Engraulis encrasicolus 1758) during the commercial catching season. J, Muscle Foods, 21:474–483.
- Kocatepe, D. and H. Turan (2012). Proximate and fatty acid composition of some commercially important fish species from the Sinop region of the Black Sea. Lipids, 47: 635–641.
- Kris-Etherton, P.M., W.S. Harris and L.J. Appel (2002). Fish consumption, fish oil, omega-3 fatty acids and cardiovascular disease. Circulation, 106: 2747–2757.
- Luczynska, J., Z.Borejszo, M.J. and Luczynski (2008). The composition of fatty acids in muscles of six freshwater fish species from the Mazurian great lakes (Northeastern Poland). Arch Pol Fish, 16: 167–178.
- Lunn, J. and H.E. Theobald (2006). The health effects of dietary unsaturated fatty acids. Nutr Bull, 31: 178–224.
- Memon, N.N., F.N. Talpur, M.I. Bhanger and A. Balouch (2011). Changes in fatty acid composition in muscle of three farmed carp fish species (Labeorohita, Cirrhinusmrigala, Catlacatla) raised under the same conditions. Food Chem, 126:405–410.
- O¨zogul, Y. and Z.O¨zogul (2007). Fatty acid profiles of commercially important fish species from the Mediterranean, Aegean and Black Seas. Food Chem, 100: 1634–1638.
- Osman, H., A.R. Suriah, and E.C. Law, (2001). Fatty acid composition and cholesterol content of selected marine fish in Malaysian water. Food Chem, 73:55–60.
- Ozogul, Y., F. Ozogul and S. Alagoz (2007). Fatty acid profiles and fat contents of commercially important seawater and freshwater fish species of Turkey: A comparative study. Food Chem, 103: 217–223.
- Ozogul, Y., F.Ozogul, E. Cicek, A. Polat and E. Kulay (2009). Fat content and fatty acid compositions of 34 marine water fish species from the Mediterranean Sea. Inter J Food Sci Nutr, 60: 464–475.
- Ozyurt, G. and A. Polat (2006). Amino acid and fatty acid composition of wild sea bass (Dicentrarchus labrax): A seasonal differentiation. Eur Food Res Technol 222: 316–320.
- Park, Y., J. Lim, Y. Kwon and J. Lee (2009). Correlation of erythrocyte fatty acid composition and dietary intakes with markers of atherosclerosis in patients with myocardial infarction. Nutr Res, 29: 391–396.
- Pickova, J., P.C. Dutta, P.O. Larsson and A. Kiessling (1997). Early embryonic cleavage pattern, hatching success, and egg-lipid fatty acid composition: comparison between two cod (Gadusmorhua) stocks. Can J Fish Aquat Sci, 54: 2410-2416.
- Pompeia, C., J.S. Freitas, S. Kimj, S.B. Zyngier and R. Curi (2002). Arachidonic acid cytotoxicity in leukocytes: Implications of oxidative stress and eicosanoid synthesis. Biol Cell, 94: 251–265.
- Shannon, J., J.O’Malley, M. Mori, M. Garzotto, A.J. Palma and I.B. King (2010). Erythrocyte fatty acids and prostate cancer risk: a comparison of methods. Prosta Leuko Essen Fatty Acids, 83: 161–169.
- Shen, C., J. Xie and X. Xu (2007). The components of cuttlefish (Sepiella maindroni De Rochebruns) oil. Food Chem 102: 210–214.
- Strobel, C., J. Gerhard and K. Katrin (2012). Survey of n-3 and n-6 polyunsaturated fatty acids in fish and fish products. Lipids Health Dis, 11: 1-10.
- Ugoala, C., G.I. Ndukwe and T.O. Audu (2009). Investigation of the constituent fatty acids of some freshwater fishes common in Nigeria. Braz J Aqu Sci Technol, 13: 65–70.
- Usydus, Z., J.Szlinder-Richert, M. Adamczyk and U. Szatkowska (2011). Marine and farmed fish in the Polish market: comparison of the nutritional value. Food Chem, 126:78–84.
- Zuraini, A., M.N. Somchit, M.H. Solihah, Y.M. Goh, A.K. Arifah, M.S. Zakaria, Somchit, M.A. Rajion, Z.A. Zakaria and A.M. Mat Jais (2006). Fatty acid and amino acid composition of three local Malaysian Channa spp. fish. Food Chem, 97: 674–678.
|





