POMEGRANATE JUICE
AMELIORATES ATENOLOL TOXIC EFFECTS ON EMBRYO DEVELOPMENT
S. Zafar1, Asmatullah1and C. Ara1, *
1Department of
Zoology, New
Campus, University of the Punjab, Lahore, Pakistan
*Corresponding author’s email: dr.chamanara@yahoo.com; chaman.zool@pu.edu.pk
ABSTRACT
Hypertension is an
increasing issue experienced by females during pregnancy. Gestational
hypertension is treated by many antihypertensive drugs including methyldopa,
beta blockers, and calcium channel blockers. Atenolol is usually the drug of
choice by obstetricians among beta blockers. Objective of this study was to
find the fetotoxic effects of Atenolol in albino mice and protective potential
of pomegranate juice against potential atenolol toxicity. Embryotoxic effect of
atenolol was determined in pregnant mice, force fed orally through gavage three
different atenolol concentrations (3.30µg/g, 2.50µg/g, 1.65µg/g body weight of
treated mice, respectively) from 6th -12th day of gestation. Other three groups
were made for antidote study and 50% diluted pomegranate juice was provided to
each group along with above mentioned atenolol concentrations as an antidote. During
osteogenesis, fetuses in atenolol exposure groups have varying degree of
incomplete ossification in dose dependent way. Histological analysis had also
shown teratogenic potential of atenolol in developing mice embryos. The present
study revealed that administration of atenolol during organo-genetic period upset
prenatal development in mice fetuses. To minimize the toxic effects of drug,
pomegranate juice was provided to experimental mice. Positive outcomes
represented that pomegranate juice has protective potential against Atenolol
induced fetotoxicity.
Key
words: Atenolol, Fetotoxity, Hypertension, Ameliorative, Pomegranate.
https://doi.org/10.36899/JAPS.2020.5.0131
Published online June 25, 2020
INTRODUCTION
Hypertension
during pregnancy is one of the major problems faced by obstetricians (Andrade et al.,
2004).
A recent study in United Kingdom reported that about one-third of the total
maternal morbidity was caused by hypertensive disorders. Another study by
National institute for health and care excellence stated that 1 out of 20 women
with severe hypertension during pregnancy were admitted to hospital (Alexander &
Wilson, 2013; McCormack et al., 2012). Many disorders caused by hypertension in
pregnancy bear risks for the woman and the baby. It is a significant cause of morbidity
and mortality during pregnancy. Recent indication by few clinical trials had
revealed that medicinal management is beneficial for the reduction of
hypertension and other outcomes related to the hypertension (James et al.,
2014).
There
are a lot of antihypertensive drugs available that belong to different classes.
In Churchill’s pocketbook of hypertension, beta-blockers are said to be
appropriate as initial choice for young patients in early phase of hypertension
in non-obstetric patients as well as pregnancy-associated hypertension (Isla et
al., 2005; Hind and Sara, 2014). Among beta-blockers, atenolol is a
commonly used antihypertensive drug during pregnancy (Butters et al.,
1990).
So in pregnancy the risk of preeclampsia and eclampsia is greatly reduced in
cases where atenolol is used as antihypertensive treatments (Lydakis et al.,
1999).
Some
reports of atenolol related fetopathy in newborns were published in last few
decades. These reports were featuring few individual case reports where mothers
were treated for hypertension (Freyer, 2009). However, there
is a lack of systemized data. Keeping in view this deficiency; it seems
unavoidable to conduct experiments on animal models. In NICE clinical
guidelines it is advised that the expected benefits should be measured against
potential threats of drug use in pregnancy. Informed approval on the use of
atenolol in these situations should be acquired and documented (Moussa et al.,
2014).
Antihypertensive
medication is indicated if the systolic blood pressure rises above 150-160 mmHg
or diastolic blood pressure rises above 100-110 mmHg or there is any end organ
damage (Chobanian et al.,
2003).
Atenolol remained the drug of choice for the last few decades. Its wide range
uses, success and comparatively less side effects have added to its reputation.
Furthermore, the absence of severe metabolic alterations enhanced its value as
antihypertensive (Mancia et al.,
2009).
The reports of atenolol related fetotoxicity in newborns revealed its major
drawback (Reynolds et al.,
1984).
Present study was conducted to evaluate fetotoxity of Atenolol and to minimize
its effects by using pomegranate juice as a natural remedy for improving
maternal and fetal health. As our research is on mammalian model (mice) so
results can be extrapolated to humans.
MATERIALS AND METHODS
Swiss
Webster male and female albino mice of 6-week old were taken from Veterinary
Research Institute, Lahore, Pakistan, having weight about 28 ± 2g. They were
put into sanitized steel cages (1 male:2 female) and were allowed to mate
freely for raising colony. Mice were placed in animal house with good
ventilation and maintained temperature (27 ± 2oC). Cages were well
supplied with water and mice Feed # 13, manufactured by National Feeds Ltd.,
Lahore, Pakistan. Mated females were recognized by the presence of semen as
white colored plug. The date was noted as day of conception. The next day is
counted as first day of gestational period. The pregnant mice were then placed
in separate cages to avoid any interference.
Experimental
design and dose administration: Mice were randomly divided into 8 groups designated
as control (C), vehicle control (VC) and 3 atenolol treated (LD), (MD), (HD)
and 3 atenolol + antidote treated groups (LD+AD), (MD+AD), (HD+AD). Each group
contains 10 pregnant females. Different concentrations of atenolol were
prepared in such a way that 0.1 ml. of solution contained the desired amount of
drug. The high dose contains Atenolol as 3.30µg/ g body weight (B.wt.) of
treated mice, medium dose contains 2.50 µg/g B.wt. and low dose contain 1.65
µg/g B.wt. In Atenolol treated groups females were forced fed and desired concentrations
were administered orally via gavage from 6th -12th day of gestation regularly
once in a day.
Fresh
pomegranates (Punica granatum) were bought from local market and washed
carefully to remove surface adherents. The peel of pomegranates was removed and
the seeds were crushed by using blender. The residue was passed through filter
paper no.40 (Whatmans) and juice was obtained. The juice collected was prepared
by adding distilled water in 50% proportion (Awari et al.,
2009).
Three
groups were made for antidote study and 50% diluted pomegranate juice was
provided to each high (3.30 µg/g B.wt.), medium (2.50 µg/g B.wt.) and low dose
(1.65 µg/g B.wt) group (HD+AD), (MD+AD), (LD+AD) along with the drug dose from
6th-12th gestational day as a sole treatment. Each mouse
approximately consumes 5ml pomegranate juice in a day.
Fetal Skeletal
analysis: Method
of Kawamura et al. (1990) was followed to prepare fetal skeleton. The
selected fetuses from exposure groups and antidote groups were given an
abdominal incision and viscera were removed. Then fetuses were placed in 2% KOH
for clearing flesh from bones. 2-3 drops of 1% KOH were added to Alizarin Red S
and the solution is used to stain fetuses. 20% glycerinated 1% KOH was used to enhance
visibility and then specimens were preserved in 50% ethanolic glycerol.
Macro-photography was done by using Panasonic Lumix TZ15 camera.
Fetal Histological
analysis: All
females were anaesthetized with 5% isoflurane inhalation and dissected at 18th gestational day and fetuses were fixed in Bouin’s fixative for further studies (Baker, 1958). Some fetuses
were selected from all dose groups for histological studies including control
and VC groups. Fetuses after fixation in Bouin’s fixative were washed with 70%
alcohol several times to completely remove the color of picric acid. Then dehydrated
with higher grades of ethanol i.e. 70%, 90% and 100% gradually. Fetuses were
left over night in xylene for clearance and then immersed in paraffin wax for
infiltration.
Fetuses
embedded in molten paraffin wax in special molds for the process of section
cutting. After solidification, molds were removed and wax blocks were trimmed.
These blocks with embedded fetuses were then subjected to microtome for cutting
4-5µ thick transverse sections. To reduce the wrinkles, these sections were
spread in water bath with warm water at 37- 40°C. Egg albumin was coated on
slides before mounting the sections. These slides were stained with
hematoxylin, and eosin was used as a counter stain. Canada balsam was applied
on stained sections for microbial protection and for clearance. All slides were
then studied using stereoscopic compound microscope following atlas of mouse
development (Matthew H.
Kaufman & Kaufman, 1992) to observe congenital anomalies. Microphotographs
were taken by using digital camera.
RESULTS AND DISCUSSION
Skeletal Studies: The structures of
all specimens from control and vehicle control group were well ossified and stained
(Fig.1).
Fetuses
in atenolol exposure groups have less ossified skeleton in carpals,
metacarpals, phalanges, tarsals and metatarsals. In high dose group
ossification of forelimb and hind limb was also affected (Figs. 2& 3). Simple
arrows showed less degree of ossification while bold arrows showed no
ossification in three different dose groups as well as in antidote groups.
Similar decrease in skeletal ossification and length of umbilical cord was also
observed previously along with structural deviations indicate intrauterine
growth retardation (Tabacova et al.,
2003).
Fitzgerald reported that renal pelvic enlargement was also observed in some
cases (Fitzgerald et
al., 1978).
The damaging oxidative effect of low density lipids is minimized by
Polyphenolic flavonoids in pomegranate juice which inhibits the development of
atherosclerosis (Aviram &
Rosenblat, 2012).
Lablels:
F: Frontal, N: Nasal, Pm: Pre Maxila, Md: Mandible, H: Humerus, R: Radius, U:
Ulna, Ri: Ribs, Ti: Tibia, Fi: Fibula, Fe: Femur, A: Atlas, Eo: Exocipital, So:
Supraocipiltal, Ip: Interperitonial, P: Parietal

Figure 1: Fetuses obtained from Control and Vehicle Control
Group showing well ossified skeleton.

Figure 2: Fetuses obtained from mothers administered with
atenolol showing defective and less ossified skeleton [A: High dose, B: Medium
dose, C: Low dose]
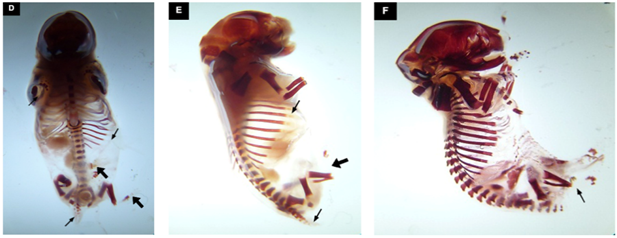
Figure 3: Fetuses obtained from mothers administered with
atenolol + pomegranate juice clearly indicating protective effect of
pomegranate juice [D: High dose+ AD; E: Medium dose + AD ; F: Low dose+ AD].
Histological
studies: Histological
studies of major fetal viscera including spinal cord, lung, liver, heart and
brain was done to understand histopathological changes caused by atenolol. For
histological studies some fetuses were randomly selected from each group.
Control: All fetuses belong
to control group depicted well developed structures with normal features as
shown in fig. 4
Vehicle Control
(VC): Fetuses
in the vehicle control group were normal and have quite developed cardiac and other
viscera (fig. 5)
Low Dose Group (1.65
µg/g B.wt): This
exposure group had fetuses with normal structures a few histological defects
like misshapen or distorted structures were observed in few cases (fig. 6)
Medium Dose Group (2.50µg/g B.wt.): Middle dose group had fetuses with internal defects like
misshapen and with mal positioned body structures. In figure shown below
undifferentiated neuroglial cells in the lateral part of cerebellum and
herniation in 4th ventricle was obvious (Fig. 7). In heart region,
hypoplasia of atrium and thick walled ventricles were observed.
High Dose Group (3.30µg/
g B.wt.): High
dose group had fetuses with internal defects like reduced sized organs,
degenerated muscles was observed. Enlargement of fourth ventricle, degeneration
of submandibular gland on right side was also clear in fig. 8.
Developmental
toxicity of atenolol is assumed previously and different researchers have
established more
or less similar
findings in this regard (Bayliss et al.,
2002; Lip et al., 1997; Lydakis et al., 1999; Tabacova et al., 2003).

Figure 4: Histological sections of control group fetuses
through cardiac and hepatic regions
Labels:
Sc: Spinal Cord, Cp: Cartilage Primordium of Basisphenoid, SVc: Superior Vena
Cava, Ll: Left Lung, La: Left Atrium, Lu: Left Ulna, Rv: Right Ventricle, Pa:
Pulmonary Artery, Pv: Pulmonary Vein, Rl: Right Lung, Ri: Ribs, Ra: Right
Atrium, Fs: Fundus Region of Stomach, LlL: Left Lobe of Liver, Vt: Vagal Trunk,
IVc: Inferior Vena Cava, RcD: Right Crus of Diaphragm, CpTv: Catilage
Primordium of Upper Thoracic Vertebral Body, RlL: Right Lobe of Liver, Liz:
Lobar Inter Zone, LdD: Lumen of Decending part of Duodenum, LSi: Lumen of Small
Intestine, Uc: Umbilical Cord, D: Diaphragm, Pv: Portal Vein, P: Pancreas, Pd:
Pancreatic Duct, Si: Small Intestine, RT: Right Tibia, Pp: Prepuce, Gp: Glans
Penis, Rf: Right Fibula, T: Tarsus, Ph: Phalanges, Tl: Tail, Tc: Tentorium
Cerebelli,
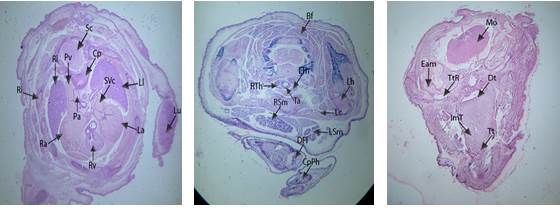
Figure 5: Histological sections of vehicle control group
fetuses through cardiac and sublingual regions
Labels:
Sc: Spinal Cord, Cp: Cartilage Primordium of Basisphenoid, SVc: Superior Vena
Cava, Ll: Left Lung, La: Left Atrium, Lu: Left Ulna, Rv: Right Ventricle, Pa:
Pulmonary Artery, Pv: Pulmonary Vein, Rl: Right Lung, Ri: Ribs, Ra: Right
Atrium, Bf: Brown Fat, Ml: Mantle Layer, Ft: Foramen Transversarium of Cervical
Vertebra, Ls: Left Scapula, Cc: Cricoid Cartilage, LTh: Left Lobe of Thyroid Gland,
Lh: Left Humerus, Tr: Trachea, ItG: Intertubercular Groove, RTh: Right Lobe of
Thyroid Gland, RSm: Right Submandibular gland, Rc: Right Clavicle, Lc: Left
Clavicle, LSm: Left Submandibular gland, DFI: Digit of Right Forelimb, CpPh:
Cartilage Primordium of Phalangeal Bones, Cm: Constrictor Muscle, Cp: Cartilage
Primordium of Basisphenoid, Dt: Dorsum of Tongue, Eam: External Auditory
Meatus, ImT: Intrinsic Muscle of Tongue, Tt: Tip of Tongue, TtR: Tubo –
Tympanic recess.
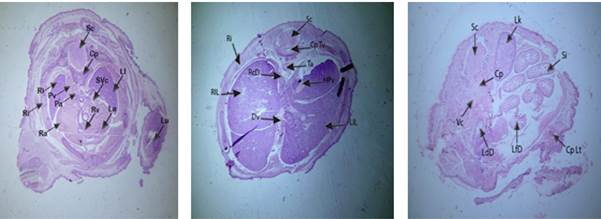
Figure 6: Photomicrographs of fetuses from low dose group
through cardiac, hepatic and intestinal region
Labels:
Cp: Cartilage Primordium of Basisphenoid, La: Left Atrium, Ll: Left Lung, Lu:
Left Ulna, Pa: Pulmonary Artery, Pv: Pulmonary Vein, Ra: Right Atrium, Ri:
Ribs, Rl: Right Lung, Rv: Right Ventricle, Sc: Spinal Cord, SVc: Superior Vena
Cava, Cp: Cartilage Primordium of Basisphenoid, CpLt: Cartilage Primordium of
Left Femur, LdD: Lumen of Decending part of Duodenum, LfD: Lumen of First part
of Duodenum, Lk: Left Kidney, Sc: Spinal Cord, Si: Small Intestine, Vc: Vena
Cava, CpTv: Catilage Primordium of Upper Thoracic Vertebral Body, Dv: Ductus
Venosus, HPv: Hepatic Portal Vein, LlL: Left Lobe of Liver, Ri: Ribs, RlL:
Right Lobe of Liver, LcD: Left Crus of Diaphragm, Sc: Spinal Cord, Ta: Trachea
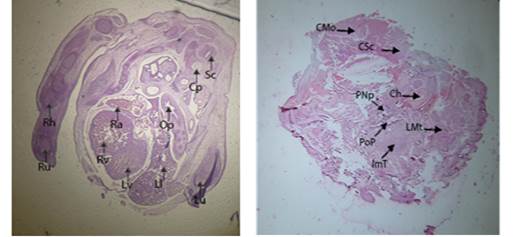
Figure 7 : Photomicrographs of fetuses from medium dose
group through cardiac and sublingual regions.
Labels:
Cp: Cartilage Primordium of Basisphenoid, Ll: Left Lung, Lv: Left Ventricle,
Op: Esophagus, Ra: Right Atrium, Rh: Right Humerus, Ru: Right Ulna, Rv: Right
Ventricle, Sc: Spinal Cord, CMo: Caudal part of Medulla Oblongata, CSc: Clivus,
ImT: Intrinsic Muscle of Tongue, PNp: Posterior Nasopharynx, POp: Posterior
Part of Pharynx, LMt: Left Molar Tooth, Ch: Cochlea

Figure 8: Photomicrographs of fetuses from high dose group
brain region.
Labels:
DgM: Degenerated Muscles, Np: Nucleus Pulposus, Sc: Spinal Cord, Fv*: Fourth
Ventricle, Tv: Third Ventricle, CpPh: Cartilage Primordium of Phalangeal Bones,
Lh: Left Humerus, LSm: Left Submandibular gland, LTh: Left Lobe of Thyroid
Gland, RTh: Right Lobe of Thyroid Gland, Sj: Shoulder Joint, Ta: Trachea, Sc:
Spinal Cord, Fv: Fourth Ventricle, Hc: Hyaloid Cavity, IrS: Intra Retinal
Space, L: Lens, Nr: Neural Layer of Retina, PRe: Pupil of Right Eye, Sg: Serous
Gland, Tc: Tentorium Cerebelli, Tv: Third Ventricle.
Low Dose (1.65
µg/g B.wt) + Antidote Group: This dose group has similar results to
control and vehicle control groups (fig. 9), with normal and well developed
fetuses.
Medium
Dose (2.50 µg/g B.wt.) + Antidote
Group: This group also shows abnormalities like mishappened structures
(fig. 10). Poorly formed ventricular chambers and degeneration of cardiac
muscles especially in atrium was observed.
These results are
comparable with the other studies. The fruit juice of punica granatum is
reported for cardiotonic activity. Studies demonstrated positive inotropic
activity of pomegranate extract on isolated frog’s heart (Ravindra et al.,
2012).
Pomegranate juice used as an antidote in present study significantly minimized
the detrimental effects of atenolol during pregnancy. Recent clinical trials
have demonstrated many advantages of pomegranate juice consumption. Pomegranate
contains polyphenols and anthocyanins which are beneficial to cardiac health as
these compounds improve vascular function and have anti-inflammatory effects (Aviram et al.,
2004).
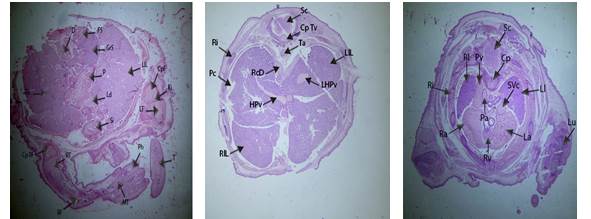
Figure 9: Photomicrographs of fetuses from low dose +
antidote group through hepatic and cardiac regions
Labels:
Cp: Cartilage Primordium of Basisphenoid, La: Left Atrium, Ll: Left Lung, Lu:
Left Ulna, Sc: Spinal Cord, Pa: Pulmonary Artery, Pv: Pulmonary Vein, Ra: Right
Atrium, Ri: Ribs, Rl: Right Lung, Rv: Right Ventricle, SVc: Superior Vena Cava,
Ae: Anterior Chamber of Eye, CPt: Cartilage Primordium of Turbinate Bone, DVs:
Transverse Dural Venous Sinus, RcD: Right Crus of Diaphragm, Ri: Ribs, RlL:
Right Lobe of Liver, Sc: Spinal Cord, Ta: Trachea, CpF: Cartilage Primordium of
Femur, Cp RF: Cartilage Primordium of Right Femur, D: Diencephalon, FS: Fundus
Region of Stomach, GrS: Glandular Region of Stomach, Kj: Knee Joint, Ld: Lumen
of Duodenum, LlL: Left Lobe of Liver, RT: Right Tibia, MT: Meta Tarsus, P:
Pancreas, Ph: Phalanges, RF: Right Fibula, RlL: Right Lobe of Liver, Si: Small
Intestine, T:Tarsus.
Labels:
Cp: Cartilage Primordium of Basisphenoid, Ll: Left Lung, Sc: Spinal Cord, Cp
Cv: Cartilage Primordium of Centrum of Eighth Cervical Vertibra, LC: Left
Clavicle, LSm: Left Submandibular gland, LT: Lumen of Trachea, Op: Oesiphagus,
Ps: Pectoralis Super Ficialis Muscle, RSm: Right Submandibular gland, RsV:
Right Sub Clavian Vein, Sc: Spinal Cord, Cp Tv: Catilage Primordium of Upper
Thoracic Vertebral Body, HPv: Hepatic Portal Vein, LlL: Left Lobe of Liver, Pc:
Pleural Cavity, Ri: Ribs, RlL: Right Lobe of Liver, Ta: Trachea.

Figure 10: : Photomicrographs of fetuses from medium dose +
antidote group through hepatic and cardiac regions
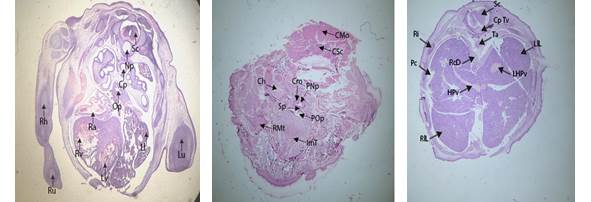
Figure 11: Cross sections of fetuses obtained from mothers
from high dose + antidote group abdominal region.
Labels:
Cp: Cartilage Primordium of Basisphenoid, Ll: Left Lung, Lu: Left Ulna, Lv:
Left Ventricle, Np: Nucleus Pulposus, Op: Oesiphagus, Ra: Right Atrium, Rh:
Right Humerus, Ru: Right Ulna, Rv: Right Ventricle, Sc: Spinal Cord, Ch:
Cochlea, Cm: Cephalic Mesenchyme, CMo: Caudal part of Medulla Oblongata, CSc:
Clivus, ImT: Intrinsic Muscle of Tongue, PNp: Posterior Nasopharynx, POp: Posterior
Part of Pharynx, RMt: Right Molar Tooth, Sp: Soft Palate, Cp Tv: Catilage
Primordium of Upper Thoracic Vertebral Body, HPv: Hepatic Portal Vein, LlL:
Left Lobe of Liver, LHPv: Left Hepatic Portal Vein, Pc: Pleural Cavity, RcD:
Right Crus of Diaphragm, Ri: Ribs, RlL: Right Lobe of Liver, Ta: Trachea
High Dose (3.30µg/
g B.wt.) + Antidote Group: This exposure group show many abnormalities like
thickened myocardium and degenerated lung tissue indicating congenital
emphysema.
In
all the exposure as well as antidote groups, liver seem to be normal with
respect to histology and anatomy. The lungs in control, vehicle control and low
dose (1.65 µg/g B.wt.) groups were normal in their gross structures, while
small lesions were observed in medium and high dose (3.30µg/ g B.wt.) groups
(fig. 11). Atenolol like that of other ß-blockers can cross the Placental
barrier and its level in maternal and fetal blood is more or less equal (Rasanen &
Jouppila, 1995).
Cardiac function and umbilico-placental circulation of fetus are directly
affected by atenolol. ß-adrenergic receptors are present in placenta and
umbilical vessels and atenolol causes reduced umbilical blood flow in humans. Thus
placental weight is significantly reduced after maternal atenolol treatment
during pregnancy (Montan et al.,
2009).
This decrease is correlated with intrauterine growth retardation (lUGR),
anemia, oxidative stress and lower birth weight in human and rodent species
independent of gestational age (Lip et al., 1997).
Conclusion: Atenolol
can be used in pregnancy however there is insufficient data to prove its safety
during first trimester. The present study revealed that the administration of
atenolol during organo-genetic period can cause skeletal and histopathological
deformities in developing mice embryos, which can be reduced by using
Pomegranate juice.
If
drug therapy is necessary, then to minimize risks and maintain maternal-fetal health
fresh punica granatum juice should be used.
Acknowledgments: Authors are
thankful to Department of Zoology, University of the Punjab, Lahore for
providing research facilities.
Conflict of
interest Statement: Authors
declares no any kind of financial and authorship conflict of interest.
REFERENCES
- Alexander, J. M. and K. L. Wilson (2013). Hypertensive emergencies of pregnancy. Obstet. and Gynecol. 40(1): 89–101. https://doi.org/10.1016/j.ogc.2012.11.008
- Andrade, S. E., J. H. Gurwitz, R. L. Davis, K. A. Chan, J. A. Finkelstein, K. Fortman, … R. Platt (2004). Prescription Drug Use in Pregnancy. Am. J. Obstet. Gynecol. 191(2): 398–407. https://doi.org/10.1016/j.ajog.2004.04.025
- Aviram, M. (2004). Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clin. Nutr. 23(3):423-33.
- Aviram, M. and M. Rosenblat (2012). Pomegranate Protection against Cardiovascular Diseases. In Evidence-based complementary and alternative medicine: eCAM (Vol. 2012).
- Awari, D. M., V. M. Mute, and B. B. Thube (2009). Cardiotonic Activity from the Fruit Juice of Punica Granatum. J. Pharm. Res. 2(2), 182–184.
- Baker J. R. J. (1958). Histochem. Cytochem.: Official. J. Histochem. Soc. 6:303–308.
- Bayliss, H., D. Churchill, M. Beevers, and D. G. Beevers (2002). Anti-Hypertensive Drugs in Pregnancy and Fetal Growth: Evidence for “Pharmacological Programming” in the First Trimester. Hypertension in Pregnancy. 21(2): 161–174.
- https://doi.org/10.1081/PRG 120013785
- Butters, L., S. Kennedy, and P. C. Rubin (1990). Atenolol in Essential Hypertension During Pregnancy. British. Med. J. 301(6752): 587–589. https://doi.org/10.1136/bmj.301.6752.587.
- Chobanian , A. V., G. L. Bakris, H. R. Black, W. C. Cushman, L. A. Green, J. L. Izzo, …. E. J. Roccella (2003) Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 42(6):1206-1252. doi: 10.1161/01.HYP.0000107251.49515.c2
- Fitzgerald, J. D., R. Ruffin, K. G. Smedstad, R. Roberts, and J. McAinsh (1978). Studies on the Pharmacokinetics and Pharmacodynamics of Atenolol in Man. Eur. J. Clin. Pharmacol. 13(2):81–89.
- Freyer, A. M. (2009). Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. Obstet. Med. 2(2): 89-89.
- Hind, N. M. and E. A. Sara (2014). "Management of Hypertensive Disorders in Pregnancy." Women's Health. 10(4): 385-404.
- Isla S. M., B. W. Ian and R. C. John (2005). Churchill's Pocketbook of Hypertension. Ist Ed. Churchill Livingstone London, 140p
- James, P. A., S. Oparil, B. L. Carter, W. C. Cushman, C. Dennison-Himmelfarb, J. Handler, … E. Ortiz (2014). 2014 Evidence-Based Guideline for the Management of High Blood Pressure in Adults: Report from the Panel Members Appointed to the Eighth Joint National Committee (JNC 8)2014 Guideline for Management of High Blood Pressure.JAMA.311(5):507–520. https://doi.org/10.1001/jama.2013.284427
- Kawamura, S., A. Hirohashi, T. Kato, and M. Yasuda (1990). Bone‐Staining Technique for Fetal Rat Specimens without Skinning and Removing Adipose Tissue. Congenital. Anomalies. 30: 93–95.
- https://doi.org/10.1111/j.17414520.1990.tb00498.x
- Lip, G. Y. H., M. Beevers, D. Churchill, L. M. Shaffer, and D. G. Beevers (1997). Effect of Atenolol on Birth Weight. Am. J. Cardio. 79(10): 1436–1438. https://doi.org/10.1016/S0002-9149(97)00163-XM.
- Lydakis, C., G. Y. H. Lip, M. Beevers, and D. G. Beevers (1999). Atenolol and Fetal Growth in Pregnancies Complicated by Hypertension. Am. J. Hypertension. 12(6): 541–547. https://doi.org/10.1016/s0895-7061(99)00031-x
- Mancia, G., S. Laurent, E. Agabiti-Rosei, M. Burnier, M. J. Caulfield, et al., 2009. Reappraisal of European guidelines on hypertension management: a European Society of Hypertension Task Force document. J. Hypertens. 27(11):2121-2158.
- McCormack, T., T. Krause, and N. O'Flynn, (2012). Management of hypertension in adults in primary care: NICE guideline. British. J. Gen. Practice. 62(596): 163-164.
- Montan, S., I. Ingemarsson, K. Marsal, and N. O. Sjoberg (1992). "Randomised controlled trial of atenolol and pindolol in human pregnancy: effects on fetal haemodynamics." BMJ. 304(6832): 946-949.
- Moussa, H. N., S. E. Arian, and B. M. Sibai (2014). Management of Hypertensive Disorders in Pregnancy. Women’s Health, 10(4): 385–404. https://doi.org/10.2217/WHE.14.32
- Räsänen, J. and P. Jouppila (1995). "Uterine and fetal hemodynamics and fetal cardiac function after atenolol and pindolol infusion. A randomized study." Euro. J. Obstet. Gyn. Reprod. Biol. 62(2): 195-201.
- Reynolds, B., L. Butters, J. Evans, T. Adams, and P. C. Rubin (1984). First year of life after the use of atenolol in pregnancy associated hypertension. Arch. Dis. Child. 59(11): 1061–1063.
- Ravindra Babu, P., H. P. L. Peter, M. Ankaiah, P. Hemanth Sairam, and M. Ramesh (2012). Positive Inotropic Actvity of Aqueous Extract of Pericarp of Punica Granatum on Isolated Frog’s Heart. Int. J. Pharm. and Pharmaceutic. Sci. 4(SUPPL.3): 95–98.
- Tabacova, S. C. A. Kimmel, K. Wall, and D. Hansen (2003). Atenolol Developmental Toxicity: Animal-to-Human Comparisons. Birth Defects Research Part A – Clin. Mol. Teratol. 67(3): 181–192. https://doi.org/10.1002/bdra.10011
- The atlas of mouse development, by M.H. Kaufman, Academic Press, San Diego, CA, 1992, 512 pp, https://doi.org/10.1002/mrd.1080370119
- The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. (2003). JAMA. 289(19):2560–72.
|





