COMPARISON OF SERUM ACUTE PHASE PROTEINS AND INFLAMMATORY CYTOKINES BETWEEN HEALTHY AND SUBCLINICALLY ENDOMETRITIC POSTPARTUM BUFFALOES
Salah¹,*, Nooruldeen Yaseen Khudhair1, Rasheed Y1 and Mohammad Yousif Mahmood1
¹Faculty of Veterinary Medicine, University Of Diyala, Baquba, 00964, Iraq.
Corresponding author’s E-mail: salah.Laser@yahoo.com , baba65640@gmail.com
ABSTRACT
The aim of the current study was to determine the levels of acute-phase proteins haptoglobin [Hp],serum amyloid A [SAA] and proinflammatory cytokinesinterleukin-6 [IL-6, and IL-8] in serum of buffaloes that developed subclinical endometritis (SCE) during the early postpartum period. Endometritis, which is one of the most common postpartum diseases in cattle and occurs several weeks postpartum, causes severe economic losses. A total of 72 buffaloes, aged 3 years to 12 years and at 15–24 days post-calving, were enrolled in this study. All the animals were checked by transrectal palpation, ultrasound examination to evaluate degree of uterine involution. Endometrial cytological samples were collected by using cytobrush to determine the proportions of endometrial cells and PMNs and later incidence of cytological endometritis in week 4 postpartum buffaloes. Blood samples (10 ml) were collected weekly from week 4 to week 7 after calving to measure levels of IL-6, IL-8 and acute-phase proteins: Hp and SAA in healthy and buffalo cows that developed cytological endometritis. The experimental group consisted of 12 buffaloes with subclinical endometritis and the control group of 12 buffaloes without cytological endometritis. Analyses in both groups of cows were carried out at weeks 4, 5, 6, and 7 postpartum (DPP). The levels of the following cytokines: IL-6, IL-8 and acute-phase proteins: Hp and SAA were determined using ELISA. Twelve of 72 clinically healthy buffalo cows (16.6%) had subclinical endometritis (≥ 8 % PMN cows). The levels of IL-6, IL-8, Hp, and SAA significantly (P < 0.05) were higher in buffalo cows with subclinical endometritis than healthy animals. The results indicate that the evaluation of the levels of cytokines and Hp in serum can be an important diagnostic indicator in buffalo cows that developed subclinical endometritis.
Keywords: Buffalo cows; Endometrial cytology; Endometritis; Postpartum uterus.
https://doi.org/10.36899/JAPS.2021.3.0257
Published online November 20, 2020
INTRODUCTION
Severe economic losses occur in cows when the uterus is exposed to several types of microbial infection during and after parturition (Sheldon et al., 2006). Many types of bacteria play different role in the complex etiology of subclinical endometritis (SCE) (Bicalho et al., 2010). Although the uterus is exposed to different types of microorganisms after calving, more than 70 % of postpartum cows can clear uterine contamination through immune mediated responses, but 17 to 37 % of these postpartum cows may develop clinical endometritis (CE), whereas 14 to 53 % of postpartum cows are exposed to SCE (Gilbert et al. 2005; Cheong et al. 2011; Madoz et al. 2014)
Cellular and humoral immunity play an important role in the clearance of uterine infectious agents. Inflammation of the endometrium develops complicated signaling processes including the early detection of microorganism components by innate immune cells by Toll-like receptors. The proinflammatory cytokines factors like TNF-α, IL-1β, and IL-6 and chemokines (IL-8) play an essential role to stimulate leukocytes like neutrophils diapedesis and acts as a chemoattractants and to promote white blood cells for phagocytosis (Singh et al., 2008). These inflammatory cytokines also help and stimulate in the secretion of different acute phase proteins (APPs), like haptoglobin (Hp), acid glycoprotein and serum amyloid A (SAA) (Tothova et al., 2008). The function of these APPs is to clear uterine contaminants through adjustment of other immune proteins and stimulation of phagocytosis. APPs also play other roles by providing protective function against any damaging impact of different enzymes secreted during the inflammation process.
APPs are secreted mainly by the liver, and their levels in the blood of cows increase during the first weeks after calving, in response to uterine diseases (Tothova et al., 2008). Although APPs are synthesized in other organs outside the liver, the possibility of the endometrium, to syntesize and secrete APPs is still not confirmed (Davies et al. 2008). However, another previous study by Chapwanya et al. (2013) had concluded the possibility of SAA secretion by uterine endometrium cells in cows. Haptoglobin (Hp) and serum amyloid A (SAA) are considered as essential positive biomarkers for exposure to pathogens (Tothova et al., 2014). Serum amyloid A is a lipoprotein substance secreted within 24–48 h after infection, as a protein of the first line of response and SAA production is dependent mainly on proinflammatory cytokines IL-1 and TNF-α (Tothova et al., 2014). Hp is considered as a protein of the second line of inflammatory response, whose production is controlled by the proinflammatory cytokine IL-6 and its high concentration may be a characteristic of long and or subacute inflammation (Tothova et al., 2014). Brodzki et al. (2015a) found a significant increase in the high concentrations of serum SAA in cows that suffer from endometritis as compared to healthy cows. Furthermore, the concentration of Hp was significantly greater in uterine washings and serum of cows that suffered from SCE. Such alteration in the level of APPs may indicate the existence of chronic inflammation of the uterus. The continued presence of an elevated concentration of Hp factor in the uterine washings and serum in cows suffered from SCE is very important from a clinical point of view. Hp is considered as a good indicator of inflammatory response of the uterus in cows. Previous studies have implicated proinflammato+ry cytokines and Hp in serum and uterus as an important diagnostic indicator of cows that had SCE (Brodzki et al., 2015a and 2015b).
Most of the previous studies on this subject was conducted on dairy postpartum cows and there is paucity of information in buffaloes. The objective of this study was to determination and compare the concentrations of IL-6, IL-8, Hp and SAA between healthy postpartum and SCE groups in postpartum buffalo cows.
MATERIALS AND METHODS
Animals:A total of 72 Iraqi Buffalo cows (Bubalus bubalis) at 15 days to 24 days post-calving period were obtained from many different private buffalo farms located in Diayla province, Iraq. All samples were collected from animals while buffalo cows remained at their farm of origin between Jan. 2019 to October 2019. The animals were aged 3–12 years, weighed 300–450 kg, and managed under free grazing. The cows were also fed according to field management after providing concentrated feed, which consisted of alfalfa, corn silage, beet pulp, cottonseed, soybean, corn, and barley. These animals were kept in an outdoor near the rivers for wallowing and milked twice daily. Individual animal data on calving history, lactation, breed, parity were recorded. The body condition score (BCS) of the cows was evaluated by using a 5-point scale (Ferguson et al., 1994). Pregnancy diagnosis for cows was achieved by using B-mode ultrasound attached with a linear probe of 5MHz frequency (Sonosite VET 180 Plus, Bothell, WA, USA) at 150 and 200 days after calving.
Animals physical examination: All practical examinations and scoring for clinical findings were done by the same veterinarian (author) for postpartum buffaloes of the current study. Genital tract discharge of all buffalo cows was checked through examination of vaginal secretions by using hands covered with clean disposable long gloves at 20 days to 30 days post-calving to identify abnormal discharges and excludeit in this study. All animals were checked by transrectal palpation and Ultrasound examination 20 to 30 days after calving to evaluate degree of uterine involution, symmetry of uterine horns, position of the uterus relative to the pelvic brim and fluid accumulation in the bovine uterine lumen.
Endometrial cytological: Endometrial cytological samples were collected from cows by using a sterile cytobrush Plus GT, Medscand Medical, Germany (Fig. 2) was modified for utilization in cows (Madoz et al., 2013). The handle was shortened to 2 cm and threaded to enable it to be inserted into a stainless steel rod (artificial insemination gun; 65 cm × 4 mm). The cytobrush and stainless steel rod combination were then inserted into a plastic sheath (Chemise Sanitaire, IMV Technologies, and France) to avoid vaginal contamination), and the device introduced into the vagina. Next, a sleeved arm was introduced into the rectum to facilitate passage of the instruments through the genital tract and os cervix. Once the device has passed through the cervix, the CB was exposed and turned (360º) to get cellular materials from the adjacent endometrium (body of uterus). Collected samples were then rolled 2-3 times on a clean glass slide.
The sample was rolled on the sterile microscopic slide (75mm×25mm) and stored in a transport medium (LABCHEM SDN.BHD, Malaysia) for bacteriological analysis. All slides were fixed with methanol for 30 min, transported to the laboratory within 3 h, stained with 5% Giemsa stain for 3 min, and dried. All of the slides were evaluated by counting 300 cells at 400× magnification (Leitz Labourlux-S, Wetzlar, Germany) to determine the percentage of neutrophils (PMN %). Endometrial threshold value ≥ 8% was used (Madoz et al., 2013; Ricci et al., 2015) to determine the SCE occurrence in the farms between 20 and 30 days postpartum(Fig. 3).
Blood samples:Blood samples (10 ml) were collected weekly from week 4 to week 7 after calving through jugular venupuncture into plain vacutainer tubes (Becton-Dickinson, NJ, USA). Blood samples were immediately placed in an ice box and transported to the laboratory within 3 h of collection. Serum was separated by centrifugation at 2500 rpm for 15 min and was transferred to 2ml microcentrifuge tubes and stored at -80°C until analysis.
Measurement of interleukins and acute phase proteins in blood serum: The concentrations of IL-6, IL-8, Haptoglobin and SAA measured by using an enzymatic method ELISA. The QAYEE® kit (Qayee-bio for life science, China) was used for IL-6, IL-8, Haptoglobin and SAA serum levels. Sensitivity was 7.8 pg /mL 31.2 pg /mL, for IL-6, IL-8 and 7.8 ng /mL, and 12 μg/mL for haptoglobin and SAA, respectively. The Intra- and interassay coefficients of variation were 4.8% and 10.7% for IL-6, and 5.6 % and 11.4% for IL-8, and 3.8% and 12.2% for Hp, and 5.4% and 13.4% for SAA.
Statistical analysis: All the statistical methods were performed by SPSS software (version 18.0, IBM SPSS Inc., Chicago: USA). All values are expressed as the mean ±standard error of the mean. The Shapiro–Wilk test was used to confirm the normal distribution of the traits examined. The results of the control and experimental groups were compared using the Student t-test to determine statistical significance. Statistically significant differences for the samples collected on w 4, w 5, w 6 and w 7 postpartum in the two groups were calculated using one-way ANOVA as well as Tukey and Duncan post hoc tests at the probability threshold P < 0.05.

Figure
1. Transverse ultrasound images of the uterine
horn)red
arrow), B-mode ultrasound with linear
Probe of 7.5 MHz frequency.

Figure
2. Cytobrush used to obtain endometrial cytological samples.
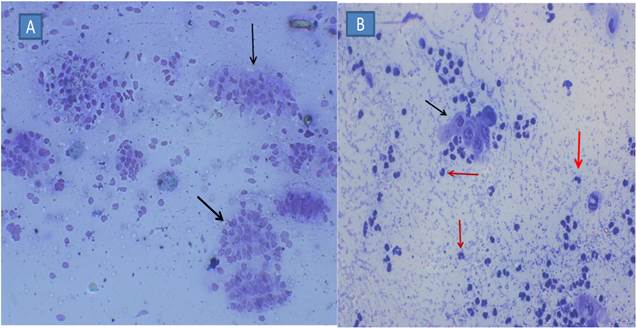
Figure
3. Cytology
smear obtained by CB from a healthy buffalo (A) and SCE buffalo (B), stained with
Giemsa. The black arrows show endometrial cells, and red pointed neutrophil
(400x).

Figure
4: Serum IL-6 levels Mean ±SEM in both healthy (n=12) and SCE (n=12) buffalo
* indicate
significant difference at p< 0.05 between healthy and SCE groups.
ᵃᵇ Means values among
weeks in same group with different superscripts indicate significant difference
at P < 0.05.

Figure
5: Serum IL-8 levels Mean ±SEM in both healthy (n=12) and SCE (n=12) buffalo
* indicate
significant difference at p< 0.05 between healthy and SCE groups.
ᵃᵇ Means values among
weeks in same group with different superscripts indicate significant difference
at P < 0.05.
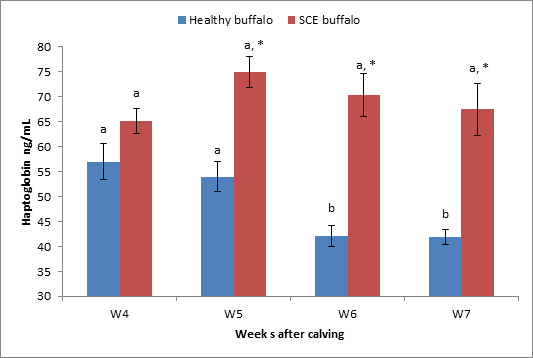
Figure
6: Serum Hp levels Mean ±SEM in both healthy (n=12) and SCE (n=12) buffalo
* indicate
significant difference at p< 0.05 between healthy and SCE groups.
ᵃᵇ Means
values among weeks in same group with different superscripts indicate
significant difference at P < 0.05.
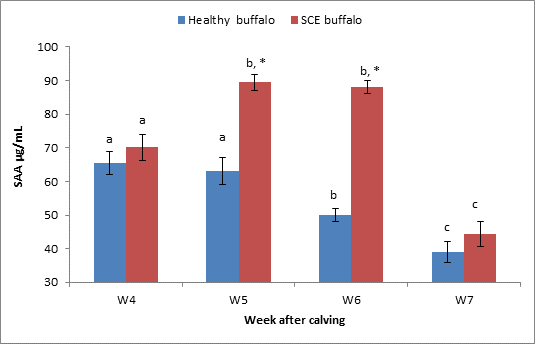
Figure
7. Serum SAA levels Mean ±SEM, in both healthy (n=12) and SCE (n=12) buffalo
groups.
* indicate
significant difference at p < 0.05 between healthy and SCE groups.
ᵃᵇͨ Means values among
weeks in same group with different superscripts indicate significant difference
at P < 0.05.
RESULTS
The overall occurrence of SCE in buffalo cows was 16.6 % (12/72) at week 4 postpartum. The results of endometrial cytology examination at week 4 (21-28 days postpartum) for buffalo cows showed that a 16.6 % (12/72) were positive to SCE (≥ 8 PMN %).
The study showed also that there was no significant (P > 0.05) difference in serum IL-6 levels between healthy group (12 animals) and buffalo cows having SCE at week 6 postpartum. However, it was significantly (P < 0.05) greater in SCE group on weeks 4, 5 and 7 among buffalo cows. The IL-6 level in healthy group was lower at week 7 (P < 0.05) than previous weeks 4, 5 and 6 postpartum while there was significant difference (P < 0.05) in serum IL-6 levels during 4-5 week and 6-7 weeks postpartum in buffalo cows having endometritis (Fig. 4).
The study showed that there was no significant (P > 0.05) difference in serum IL-8 levels between healthy group and buffaloes having SCE at week four postpartum. The level of IL-8 in infected group was greater (P < 0.05) at week 5-7 than in healthy group. The concentration of IL-8 had gradually decreased in buffaloes with SCE with the advance of the postpartum period. At week 7, there was a significantly lower concentration (P < 0.05) of IL-8 among healthy buffalo cows than at any other week postpartum (Fig. 5).
The study revealed that dairy SCE buffalo cows had greater concentrations of Hp compared with healthy group, which were significant (P < 0.05) at week 5-7 postpartum, but there was no significant difference along 4-7 postpartum SCE buffalo cows. There was a decrease in serum Hp level with a significant difference at week 6 and 7 in healthy buffalo cows when compared to week 4 and week 5 postpartum (Fig. 6).
The study showed an increasing serum level of SAA at week4 to 6 in SCE group compared to healthy ones. SAA level was decreased significantly with advance of postpartum period in both SCE and healthy groups (Fig. 7).
DISCUSSION
The present study showed that occurrence of SCE in buffalo cows was 16.6 % and this result was lower than previous studies, higher occurrence of endometritis 22.4%, 24.7%, and 25% got by (Moghaddam and, Mamoei, 2004; Sar et al., 1996, Ghanem et.al., 2002) in Iranian, Indian and Egyptian buffaloes respectively. On the other hand, this endometritis rate was higher bite than many studies in Iraq (12.3%) and Ethiopia (10.8%) (Azawi et al., 2008, and Abalti et. al., 2006). Another study using (≥ 8 PMN %) as threshold for cytological endometritis by Singh1 et al (2017) reported 22.6% in normal cyclic buffaloes in India. The occurrence of SCE in buffalo cows in the present study was 16.6%, which is greater than a report of 11.8% by Barlund et al. (2008), who used 8% PMN as the threshold during 28 to 41 postpartum in dairy cows. However, this occurrence was lower than in another study (Denis Robichaud and Dubuc, 2015) where it was reported 36% by using a threshold of 6% PMN at 28 to 42 days after calving. Thus far, no consensus has been established with regard to the effect of threshold value and time of uterine sampling on SCE diagnosis. SCE can be diagnosed using different cut-off values, such as PMN range of 5–18%with either cytobrush or low-volume lavage techniques (Kasmanickam et al., 2004; Gilbert et al., 2005; Barlund et al., 2008). Other studies depended on the thresholds of PMN % according to the effects on the reproductive performance (Cheong et al., 2011; McDougall et al., 2011). The low SCE prevalence may be attributed to differences in geographic area, environment, and the number of endometrial cells counted among the studies. A total of 300 cells were counted per slide in the present study, whereas 100 cells were counted in previous studies (Barlund et al., 2008).
The aim of this part of the study was to compare and evaluate the concentrations of proinflammatory cytokines: IL-6, chemokine IL-8, and APPs (Hp and SAA), in the serum of buffalo cows with and without SCE.
Our study showed increased levels of serum IL-6 and IL-8 during weeks 4-7 postpartum in buffalo cows with SCE compared with healthy animals. This result may be due to an increased immune response, especially in the uterus, and as a result, levels of IL6 and IL-8 in the serum of cows with SCE becomes increased. It is noteworthy that IL-6 is an essential pro-inflammatory cytokine, and plays a vital role in many aspects of the inflammation process like the induction of fever in animals, elevation of vascular permeability and stimulation APPs secretion by the liver (Van Snick, 1990).
One of the most common causes for the increased level of IL-6 is the continuous production by uterine endometrium cells, as a result of the activity of endometrium Toll-like receptors and the recognition of the pathogen that invade the endometrium in postpartum period (Turner et al., 2012). This reaction is considered the key activation factor to the uterine immune response, which leads to the attraction of leucocyte and enhancement of the activity of neutrophils and macrophages for phagocytosis (Singh et al., 2008).
Many previous studies reported infiltration of neutrophils to the endometrium of cows that suffered from SCE and this was due to exposure the uterus of these cows to many different pathogens microorganisms after calving (Kasimanickam et al., 2004; Sheldon et al., 2006; Brodzki et al., 2015a; 2015b).
The concentrations of IL-6 and IL-8 in healthy buffalo cows started to decrease gradually from week 4 until week 7 and this can be explained due to the absence of inflammatory reaction in these cows compared with cows had SCE and were characterized by high levels of proinflammatory agents.
Proinflammatory cytokine-like (IL-8), is also as considered as one of the major neutrophil chemokine, which was primitively known as a chemotactic agent produced by activated white blood cells that promote the migration of leukocytes like neutrophils and lymphocytes, have an essential function for inflammatory response and acute and chronic diseases (Tseng and Leibert, 2009). The concentrations of IL-8 in week 4 postpartum were low in buffalo cows with SCE compared with same cows in following weeks postpartum. This finding could have been related to induce stimulation by pathogenic microorganisms that begun after week 4 when the inflammation of endometrium of SCE cows have occurred. Sheldon et al. (2009) confirmed in a previous study that most of the bacterial contamination of the uterus after parturition is cleaned within three weeks after calving, and such cows are sterile during 6–8 weeks postpartum. The local immune response is not hight as because the level of uterine bacterial contamination is not high or virtually absent after birth. The concentrations of cytokines agents and APPs are also low, which was found in healthy cows while the cows that suffered SCE have a huge local uterine immune reaction and more increased proinflammatory mediators.
Our results agree with previous studies that confirm higher levels of proinflammatory cytokines (TNFa, interleukin-1b, IL-6, and IL-8) in serum of dairy cows with SCE compared with healthy cows (Brodzki et al., 2015a). Moreover, another study concluded that postpartum dairy cows with SCE expressed greater concentration of TNF-a, IL-6, and IL-8 mRNA than healthy dairy cows (Ghasemi et al., 2012). A previous study by Galvão et al. (2011) also reported, increasing pro-inflammatory cytokines (IL-1_, IL-6, and IL-8) in SCE cows only during weeks 5 and or 7 postpartum compared to control (healthy) cows. However, the present study used concentration of interleukins in serum of postpartum cows while the two previous studies depended on the express gene (Real-time RT-PCR) of these cytokines as a marker to compare between healthy and SCE dairy cows.
A study by Mossallam et al. (2015) that involved the investigation of IL8-expression in the uterus of healthy and endometritis in Egyptian buffalo cows reported a great increase in IL8 expression (26.6-fold) in endometritis compared with healthy buffalo. Moreover, the study showed that concentration of IL-6 was higher in most of the weeks of the study period and this indicates that these cytokines might be strongly related to the development of uterine inflammation in postpartum dairy cows (Brodzki et al., 2015a).
Despite the foregoing, our study has depended serum assessment to determine proinflammatory and did not use DNA and gene expression, most of the buffalo cows that had SCE showed increased the levels of IL-6 and IL-8 during most of the weeks of the study compared with healthy cows.
The current study also showed decreased concentrations of IL-6 and IL-8 in advance of the postpartum period in most cows with SCE and this finding may be due to the ability of the most of the cows with time to get rid of the pathogenic infection in the following weeks after uterine infection. In contrast, another study revealed that no significant difference exists in the levels of IL-6 and IL-8 in serum of healthy and cytological endometritis cows. However, differences in cytokines concentration were noticed in the uterine flush of endometritis cows (Kim et al., 2014). This finding may be explained due to the minor intensity of uterine infection in the previous study that causes local inflammatory reflex without increasing of serum cytokines. Also, one a study by The difference between results that have obtained by different studies may be due to the different methods that used, times of sampling and breed differences. Also, our study involved buffalo cows and due to the lack of studies about these interleukins in buffalo postpartum cows, this study depended on dairy cows as a guide to explain most of results findings.
Our results indicated increased serum SAA and Hp level during most weeks of the postpartum period for buffalo SCE cows compared with healthy groups and this agrees with other studies that have confirmed that APPs, especially Hp, and can be used as a marker to diagnosis uterine inflammatory reaction in dairy cows (Ceciliani et al., 2012). The use of Hp as an indicator for diagnosis of SCE is very useful as there were a few previous studies that included this matter (Brodzki et al., 2015a; 2015b).
The level of Hp was higher during 4-7 weeks postpartum in the serum of cows with SCE. These concentrations of APPs may suggest the persistence of chronic inflammation of the uterus from the early postpartum period to day 50 postpartum.
In contrast, Yasui et al. (2014), reported that a weak relationship between serum Hp and cytological endometritis, or with decreasing reproductive performance in postpartum cows between 40 and 60 days postpartum. The same study reported that NEB during the early postpartum period is associated with SCE and not necessarily is occurred with systemic inflammation reaction. The type of infection and intensity of microbial contamination after calving may affect the immune response, as well as the difference in the breed, age, and environment on the size and type of the immune response in these cows between different studies.
Our results confirmed the results of the most of previous studies about a significant relationship between IL-6 and the level of Hp during inflammation process, most the weeks of the study the both concentration of Hp and IL-6 was high in infected cows cpmpared with healthy cows (Tothova et al., 2014). Hp, in turn, it is a protein factor of the second line of reaction, whose production is regulated by the proinflammatory cytokines (IL-6), and its increased concentration is characteristic of long and less severe inflammatory processes (Tothova et al., 2014). Hp concentration was higher most the duration of the present study coinciding with the increasing of the IL-6, this confirms the strong relationship between them in SCE cows, also because increasing Hp is often associated with chronic infection in these cows.
Many previous studies concluded increasing Hp level at postpartum uterine infection in cows (Burke et al., 2010; Schneider et al., 2013) and this agrees with our findings in both beef and dairy SCE groups. The levels of Hp were increased from week 4 and had a significant difference during the following weeks (week 5-7). Also SAA levels were greater in SCE cows compared with healthy ones along postpartum week and this agrees with other previous studies (Brodzki et al., 2015a; 2015 b). Serum SAA in our findings was gradually decreased with the advance of the postpartum period in both healthy and SCE beef and dairy cows because it is related with initiated immune response (first line immune) and begin to decrease gradually.
Although the level of both Hp and SAA in SCE group was significantly greater than healthy cows, the studies confirmed that Hp is a better indicator of postpartum uterine disease than SAA (Chan et al., 2010). This result disagrees with another study which concluded that serum Hp increased in dairy cows with acute metritis, but not in chronic endometritis (Skinner et al., 1991 ). Our finding found IL-6, IL-8, and Hp was better markers for postpartum cows that suffer from SEC compared with healthy cows because these factors were increased most weeks of our study.
Besides inflammation, it must be noted that Hp and SAA concentrations are increased in sera of cows subjected to physical stress, such as transportation, exhaustion, starvation, or parturition, during which serum glucocorticoid concentrations increased. All cows of the present study were already checked to ensure these cows are free signs of inflammation and clinical disease to avoid possible errors in concentration of these proinflammatory and acute phase proteins.
Conclusion: The current study is considered the first report on the level of cytokines and APPs in the serum of buffalo cows that developed subclinical endometritis in the early postpartum period buffalo cows in Iraq. The current study shows increased levels of both proinflammatory and APPs in buffalo cows with subclinical endometritis compared with healthy cows during 4-7 weeks postpartum. It can clearly be seen that high, IL-6, IL-8, Hp and SAA concentrations in the postpartum period are negatively associated with cow’s health status. Our results indicated that the evaluation of the levels of IL-6, IL-8 and Hp in serum can be important diagnostic indicators of SCE in buffalo cows as long as these animals are without clinical diseases and not exposed to stress factors.
Acknowledgments: The study was funded by university of Diayla, Iraq. I would like to thank all my colleagues and all the workers in the cow herds to help me complete this study.
Author Contributions: All authors contributed effectively in this research, N. Salah designed and achieved experiments and wrote the paper; Y. Nooreddin analysed the data; M. Yassen, and Y. Mohammed supervised the project and revised the manuscript.
REFERENCES
- Abalti, , M. Bekana, M. Woldemeskel and F. Lobago (2006). Female genital tract abnormalities of Zebu cattle slaughtered at Bahir-Dar Town, north-west Ethiopia. Trop Anim Health Prod. 38: 505-510.
- Azawi, O. I, A. J. Ali and E. H. Lazim. (2008). Pathological and anatomical abnormalities affecting buffalo cows reproductive tracts in Mosul. Iraqi J Vet Sci 22: 59-67.
- Barlund, C. S., T. D. Carruthers, C. L. Waldner and C. W. Palmer (2008). A comparison of diagnostic techniques for postpartum endometritis in dairy cattle. Theriogenology69(6):714–723.
- Bicalho, R. C., V. S. Machado, M. L. S. Bicalho, R. O. Gilbert, A. G. V. Teixeira, L. S. Caixeta and R. V.V. Pereira (2010). Molecular and epidemiological characterization of bovine intrauterine Escherichia coli. Dairy Sci.93(12): 5818–5830.
- Brodzki, P., Kostro, K., Brodzki, A., Wawron, W., and Marczuk, J. (2015a). Inflammatory cytokines and acute-phase proteins concentrations in the peripheral blood and uterus of cows that developed endometritis during early postpartum. Theriogenology, 84(1), 11–18.
- Brodzki, P., K. Kostro, L. Krakowski and J. Marczuk (2015b). Inflammatory cytokine and acute phase protein concentrations in the peripheral blood and uterine washings of cows with subclinical endometritis in the late postpartum period. Vety. Res. Communications, 39(2), 143–149.
- Burke, C. R., S. Meier, S. McDougall, C. Compton, M. Mitchell and J. R. Roche (2010). Relationships between endometritis and metabolic state during the transition period in pasture-grazed dairy cows. Dairy Sci.93(11): 5363–5373.
- Chapwanya, A., K. G. Meade, M. L. Doherty, J. J. Callanan and C. OFarrelly (2013). Endometrial epithelial cells are potent producers of tracheal antimicrobial peptide and serum amyloid A3 gene expression in response to E. coli stimulation. Vety. Immunopathology 151(1): 157–162.
- Cheong, S. H., D. V. Nydam, K. N. Galvão, B. M. Crosier and R. O. Gilbert (2011). Cow-level and herd-level risk factors for subclinical endometritis in lactating Holstein cows. Dairy Sci.94(2): 762–770.
- Davies, D., K. G. Meade, S. Herath, P. D. Eckersall, D. Gonzalez, J. O. White and I. M. Sheldon (2008). Toll-like receptor and antimicrobial peptide expression in the bovine endometrium. Reprod. Biology Endo. 6(1): 53.
- Denis-Robichaud, J., and J. Dubuc (2015). Determination of optimal diagnostic criteria for purulent vaginal discharge and cytological endometritis in dairy cows. Dairy Sci.98(10): 6848–6855.
- Galvão, K. N., N. R. Santos, J. S. Galvão and R. O. Gilbert (2011). Association between endometritis and endometrial cytokine expression in postpartum Holstein cows. Theriogenology76(2): 290–299.
- Ghanem, M., A. H. Shalaby S. Sharawy and N. Saleh (2002). Factors leading to endometritis in Egypt with special reference to reproductive performance. J. Reprod. Sci., 48: 371-375.
- Ghasemi, F., P. Gonzalez-Cano, P. J. Griebel and C. Palmer (2012). Proinflammatory cytokine gene expression in endometrial cytobrush samples harvested from cows with and without subclinical endometritis. Theriogenology 78(7): 1538–1547.
- Gilbert, R. O., S. T. Shin, C. L. Guard, H. N. Erb and M. Frajblat (2005). Prevalence of endometritis and its effects on reproductive performance of dairy cows. Theriogenology 64(9): 1879–1888.
- Ferguson, J. D., D. T. Galligan and N. Thomsen (1994). Principal descriptors of body condition score in Holstein cows. J. Dairy Sci., 77(9), 2695–2703.
- Ishikawa, Y., K. Nakada, K. Hagiwara, R. Kirisawa, I. Hiroshi, M. Moriyoshi and Y. Sawamukai (2004). Changes in interleukin-6 concentration in peripheral blood of pre-and post-partum dairy cattle and its relationship to postpartum reproductive diseases. J. Vety .Med. Sci., 66(11): 1403–1408.
- Kasimanickam, R., T. F. Duffield, R. A. Foster, C. J. Gartley, K. E. Leslie, J. S. Walton and W. H. Johnson (2004). Endometrial cytology and ultrasonography for the detection of subclinical endometritis in postpartum dairy cows. Theriogenology 62(1): 9–23.
- Kim, I. H., H. G. Kang, J. K. Jeong, T. Y. Hur and Y. H. Jung (2014). Inflammatory cytokine concentrations in uterine flush and serum samples from dairy cows with clinical or subclinical endometritis. Theriogenology 82(3): 427–432.
- Madoz, L. V, M. J. Giuliodori, A. L. Migliorisi, M. Jaureguiberry and R. L. de la Sota (2014). Endometrial cytology, biopsy, and bacteriology for the diagnosis of subclinical endometritis in grazing dairy cows. Dairy Sci. 97(1): 195–201.
- McDougall, S., H. Hussein, D. Aberdein, K. Buckle, J. Roche, C. Burke and S. Meier (2011). Relationships between cytology, bacteriology and vaginal discharge scores and reproductive performance in dairy cattle. Theriogenology76(2): 229–240.
- Moghaddam, A. A. I., and M. Mamoei (2004). A survey on some of the reproductive and productive trails of the buffalo in Iran. 23rd world Buiatrics Cong. Qu & eacute, Canada 2004: 1910.
- Mossallam, A. A. A., S. M. El Nahas, E. R. Mahfouz and N. M. Osman (2015). Characterization of buffalo interleukin 8 (IL-8) and its expression in endometritis. J. Genetic Engin. Biotech., 13(1): 71–77.
- Ricci, A., S. Gallo, F. Molinaro, A. Dondo, S. Zoppi and L. Vincenti (2015). Evaluation of Subclinical Endometritis and Consequences on Fertility in Piedmontese Beef Cows. Rep.Domestic Anim., 50(1): 142–148.
- Sar, G. C., B. N. Mohunty S. K. H. Ray and D. N. Mohunty (1996). Endometrial biopsy in infertile cows. Indian J Anim Sc. 66: 1100-1105.
- Schneider, A., M. N. Corrêa and W. R. Butler (2013). Short communication: Acute phase proteins in Holstein cows diagnosed with uterine infection. Res. Sci., 95(1): 269–271.
- Sheldon, I. M., J. Cronin, L. Goetze, G. Donofrio and H. J. Schuberth (2009). Defining Postpartum Uterine Disease and the Mechanisms of Infection and Immunity in the Female Reproductive Tract in Cattle. Biology Repr. 81(6): 1025–1032.
- Sheldon, I. M., G. S. Lewis, S. LeBlanc and R. O. Gilbert (2006). Defining postpartum uterine disease in cattle. Theriogenology 65(8): 1516–1530.
- Singh, J., P. Singla, G. S. Dhaliwal, A. Kumar and H. S. Banga (2008). Histomorphological alterations in uterus of repeat breeding cows with subclinical endometritis following E. coli lipopolysaccharide and autologous serum therapy. Ind. J. Anim. Sci., 78(7): 710.
- Skinner, J. G., R. A. Brown and L. Roberts (1991). Bovine haptoglobin response in clinically defined field conditions. The Veterinary Record 128(7): 147–149.
- Tothova, C., O. Nagy and G. Kovac (2014). Acute phase proteins and their use in the diagnosis of diseases in ruminants: a review. Veterinarni Medicina 59(4): 163–180.
- Tóthová, C., O. Nagy, H. Seidel and G. Kováč (2012). Acute phase proteins in relation to various inflammatory diseases of calves. Comparative Clinical Pathology 21(5): 1037–1042.
- Tóthová, C. S., O. Nagy, H. Seidel, J. Konvičná, Z. Farkašová and G. Kováč (2008). Acute phase proteins and variables of protein metabolism in dairy cows during the pre-and postpartal period. Acta Veterinaria Brno 77(1): 51–57.
- Tseng-Rogenski, S., and M. Liebert (2009). Interleukin-8 is essential for normal urothelial cell survival. Amer. J. Physio. Renal Physio., 297(3): F816–F821.
- Turner, M. L., G. D. Healey and I. M. Sheldon (2012). Immunity and inflammation in the uterus. Reprod. in Domestic Anim., 47(s4): 402–409.
- Van Snick, J. (1990). Interleukin-6: an overview. Annual review of immunology8(1): 253-278.
|





