IMPACT OF EARLY EPHEMERAL AND TERMINAL DROUGHT ON THE GRAIN YIELD OF THE NAKED OAT (AVENA NUDA L.)
H. Batool1, 2, 3, A. Tahir2, X. Fang1* and T. Yasmin2*
1State Key Laboratory of Grassland Agro-ecosystems, School of Life Sciences, Lanzhou University, Lanzhou, 730000, China; 2Department of Biosciences, COMSATS University Islamabad, Islamabad Campus Pakistan; 3Botany Department, Sardar Bahadur Khan Women’s University Quetta, Quetta Campus, Balochistan, Pakistan
*Corresponding author’s email: tayyaba_yasmin@comsats.edu.pk, drtayyabayasmin@gmail.com, fangxw@lzu.edu.cn
ABSTRACT
Drought has become more frequent in the recent era due to climate change, which is responsible for reducing the global cereal production. In the present study, two naked oat cultivars; Dingyou7 (Dy7) and Yanmai (Ym) with contrasting drought tolerance were compared for their yield and growth adaptation under different watering schemes at two different stages of plant growth. The plants were grown in a completely randomized design (CRD) under a) well-watered (WW), b) water stressed (WS) conditions; an early ephemeral, c) terminal drought, d) and different field water capacities (FC) of 80%-90%, 60%, 40%, and 20% respectively. The root length (RL), leaf area (LA), leaf number (LN), flower number (FN) and grain yield in two growing seasons in spring to summer and mid-summer to autumn 2018- were measured for all the water treatments at Yuzhong County experiment station, Lanzhou University Gansu, China. The terminal drought and reduced FC were more detrimental to the grain yield as compared with the early ephemeral drought and higher FC as the terminal drought caused 42% and 48% whereas the early ephemeral drought caused 28% and 44% reduction in grain yield of the Dy7 and Ym respectively. All the growth parameters except RL had significant impact (p > 0.05) on the grain yield. This study shows the efficiency of the cultivar Dy7 under drought stress. The fewer number of leaves in the latest cultivar appeared as an important crop adaptation under drought stress to optimize yield. This feature holds potential for improvement of cereal crops yield under drought stress through genetic crop breeding.
Keywords: Naked Oat; Field Water Capacity; Water Stress; Grain Yield; Drought
https://doi.org/10.36899/JAPS.2021.3.0284
Published online November 11, 2020
INTRODUCTION
Oat is an important cereal crop throughout the world, which is the sixth highest amongst the global cereal production in tons (Statista, 2020). The hulless (naked) oat is acquiring greater attention of the grower and the consumer communities as it requires less water and nutrients for its growth although it provides immense health and nutritional benefits than other major cereals such as maize, rice and wheat (Zhao et al., 2017). The daily use of oat in diet can reduce the risk of heart diseases, it possess the unique class of antioxidants known as avenanthramides (AVAs). Oat grains contain sufficient amount of proteins, which makes them a staple diet for humans and cattle (Ibrahim et al., 2020). Currently, world grain production has decreased due to increasing abiotic and biotic stress environments, amongst these, drought plays a significant role. The frequency and severity of drought events in different parts of the world has increased due to climate change, (Gu et al., 2020) and in turn has caused a notable decline in the crop yield. The threats to oat cultivation specifically can be defeated by reinforcing the oat genotypes against climate change through next generation crop breeding system and manipulation of genome (Isidro-Sánchez et al., 2020). Efficiency of oat varieties to grow in drought stress is strongly linked to the abiotic factors such as the soil type and fertility levels and the temperature. However, the genetic makeup and biochemical signaling in the particular oat variety enables them to cope with the dry environments (Canales et al., 2019).
The production of viable flowers and seeds is notably affected in plants by drought stress and different cultivars may show contrasting sensitivity to drought stress in different growth stages. The terminal drought causes a major decline in the yield of cereal crops such as winter wheat whereas, the vegetative stage drought comparatively causes a negligible decline in the winter wheat crop yield (Yu et al., 2018). The vegetative stage drought has comparatively less detrimental effect on the crop yield, the maize crops showed a decline in grain yield both under vegetative stage and under terminal stage drought however the decline was greater for the drought at the terminal reproductive growth stage (Mi et al., 2018).
The present study aimed to explore a recent naked oat cultivar Dy7 introduced in 2008 in comparison with 70 years old Ym which differ in their tolerance to drought as used in previous literature (Wang et al., 2017). Therefore, this study was designed to compare the yield and growth components of the Avena nuda L. cultivars under drought stress applied at different stages of growth to determine which growth stage and cultivar is more sensitive to drought stress. The present study also emphasized to understand which vegetative growth component may have detrimental or beneficial effect under drought stress on the crop yield.
MATERIALS AND METHODS
Experimental material: In this study two naked oat cultivars the drought-sensitive Yanmai (Ym), and drought-resistant Dingyou7 (Dy7) were selected. These seeds were provided by the Institute of Crop Germplasm Resources from the Chinese Academy of Agricultural Sciences (Beijing, China) and the Dingxi Academy of Agricultural Science (Dingxi, Gansu, China).
Growth conditions and experimental design: The plants were grown in pots in controlled conditions at 25 ± 3 °C air temperature and 16 h with 200-300 μmol supplemented light in a completely randomized design (CRD). The pots were reshuffled weekly to minimize the position effect. Pots with 230 mm width and 250 mm height were filled with 7 Kg of topsoil and vermiculite mixture at (3:1 ratio) having 36.56% field water capacity (FC). Each pot was supplied with ample nutrition mainly comprising of Potassium (K), Nitrogen (N) and Phosphorus (P). In April 2018, and June 2018 the pots were inoculated with seven seeds per pot spaced approximately 3 inches from all sides, to avoid further thinning. There were 3 replicate pots for each water treatment and the control plants.
Calculation of field water capacity FC: For calculation of FC, the soil and vermiculite mixture was soaked with water and allowed to drain for 24-72 hrs. to obtain saturated soil. The weight of the saturated soil and vermiculite mixture with the container were measured as the initial weight. Then, the soil and vermiculite mixture were dried in the oven at 105 °C for 48 hours and final weight was noted. FC was calculated as the difference between weights of the saturated soil and the oven-dried soil, the weight of empty container was subtracted from both the initial and the final weight (Ogbaga et al., 2014). This procedure was repeated three times for accurate measurement.
Water treatments: The plants were kept well-watered (WW) initially, after two weeks, four different treatments were applied i.e., 90-80%, 60%, 40%, and 20% FC respectively. The pots were weighed regularly and the amount of lost water was added to maintain the required FC. Supplementary pots were kept well-watered for the first 45 days after sowing (DAS) and after that, an early transitory drought was given for 20-25 days. Furthermore, five replicate pots were kept well-watered until the commencement of flowering around 65-70 DAS and then a terminal drought was given to the plants for 20-25 days. There were five pots of control (C) plants that were kept well-watered for 110-115 DAS from germination to full plant maturity, each pot contained seven plants. The plants were recovered from both the terminal and an early ephemeral drought by rewatering. Experimental plan is summarized in fig 1.
Measurement of growth and yield components: Plant height and root length were measured in cm at 35 DAS, 70 DAS and 105 DAS respectively for different water treatments by removing the soil from the roots and washing the roots. Then the root length was measured with a ruler. The leaf area was measured in cm2 with the help of a scanner using ImageJ software. Briefly, a mature (fully extended) leaf was scanned and the leaf image was demarcated to measure the leaf area directly through the leaf area calculation option in ImageJ software (Package version: 1.52r-1, Wayne Rasband, wayne@codon.nih.gov). The procedure was replicated in triplets for all the measurements. In the month of July 2018 and in second growing season in the month of October 2018 at full maturity when the plants were 115 days old the plants were harvested. Then the mean number of grain yield per plant was calculated for both the cultivars and for each water treatment including the WW control plants as described previously (Dong et al., 2017). Briefly, four plants were randomly selected from each replicate pot for each water treatment including the C plants.
Statistical analysis: All the recorded variables including LA, LN, PH, RL, FN and grain number were tested by applying a two-way analysis of variance (ANOVA) based on the Fisher method (Fisher, 1921). Then, Duncan’s multiple range test (DMRT) was further applied for each of these variables and grain number to test the differences among the means of the treatments (Duncan, 1955). All the recorded data were initially entered into spread sheet Excel (Microsoft®, Redmond, WA, USA) using its special tool kit application and then further validated on SPSS® 26.0 Faculty Pack (IBM Inc., Chicago, Illinois, USA). Here, 𝑝 value ≤ 0.05 was considered statistically significant.
RESULTS
Single leaf area: Decreasing field water capacity caused a significant (p ≤ 0.05) decrease in the single leaf area in both the cultivars but Dy7 leaves routinely retained much larger leaf area as compared with Ym (Fig. 2). The leaf area in well-watered control plants of Dy7 was 47.8 cm2 while it was 37.4 cm2 in Ym. In water-stressed plants that were grown at an extreme drought condition of 20%, FC the leaf area decreased largely in the case of Ym plants i.e. 18 cm2 while Dy7 leaves could maintain leaf area to 31.2 cm2. In the present study, a significant leaf area reduction was recorded at 35 DAS in Dy7 and Ym WS plants grown under 60%, 40%, and 20% FC compared to the WW control plants grown in 80-90% FC.
Number of leaves per plant: A significant (p ≤ 0.05) difference was recorded in leaf number per plant at 35 DAS, 70 DAS, and 105 DAS respectively. Overall, Ym as compared with Dy7 produced greater numbers of leaves in all the treatments. After 35 DAS, the calculated mean leaf number in Dy7 and Ym at 80-90% FC was 3 and 3.2 respectively, while it decreased to 2.4 in both cultivars at 20% FC. After 70 DAS the calculated mean leaf number at 80-90% FC in Dy7 and Ym were 15.2 n and 16.2, while this number was reduced to 9.8 and 10.4 for both the cultivars respectively at 20% FC. The calculated mean leaf number at 80-90% FC after 105 DAS for Dy7 and Ym were 25 and 25.2 respectively while at 20% FC only 17.8 and 18.2 for both cultivars respectively (Table 1).
Plant height: A significant difference (p > 0.05) was recorded in plant height for various water treatments. The Dy7 plants exhibited taller plants in different water treatments (Table 1). The calculated mean height of plants after 35 DAS was 24.8 cm and 23 cm at 80-90% FC in Dy7 and Ym plants respectively. At the same age, plant height at 20% FC was 16.4 cm and 14.2 cm in Dy7 and Ym, respectively (Table 1). The mean medium plant height recorded after 70 DAS was 54 cm for Dy7 plants at 80-90% FC while it was 52 cm for Ym plants at the same FC and age. A significant decrease in mean height was noted at 20% FC, after 70 DAS Dy7 plants exhibited a height of 34.4 cm, and 30.8 cm was the height of Ym plants (Table 1). The maximum height recorded was exhibited by the Dy7 plants at the age of 105 DAS and 80-90% FC i.e. 72.8 cm while the Ym plants at the same age and water treatment exhibited a height of 64.6 cm only. The height reduced with decreasing water contents of the soil. At 20% FC which represent an extreme drought condition the calculated mean plant height was 52.6 cm and 43.2 cm for Dy7 and Ym respectively 105 DAS (Table 1).
Root length: The mean root length was 10 cm and 9.6 cm after 35 DAS in the plants of Dy7 and Ym at 80-90% FC. This length was reduced to 9.2 cm and 8.4 cm in plants of Dy7 and Ym that were grown in water stressed condition of 20% FC at the same age. The mean root length was calculated as 14 cm and 14 cm in Dy7 and Ym respectively in 70 DAS grown in well watered conditions with a field water capacity of 80-90%. The root length was 12.8 cm and 12.6 cm in the same age plants of Dy7 and Ym respectively grown under a severe drought having an FC of 40% while at 20 % FC it was 12.8 cm and 13 cm in Dy7 and Ym plants respectively. At 80-90% FC and 105 DAS, the mean root length was calculated as 22.4 cm and 23 cm in Dy7 and Ym plants respectively while the same age plants of Ym with an FC of 20% exhibited longer roots i.e. 23.2 cm while the Dy7 plants exhibited a mean root length of 20.2 cm (Table 1).
Number of flowers: A significant reduction (p ≤ 0.05) in the number of flowers was noticed in the WS plants of Dy7 and Ym. The cultivar Dy7 produced comparatively more viable number of flower than Ym. The mean number of viable flowers in WW control plants of Dy7 and Ym was 376.6 and 332 respectively. It was also evident that both cultivars showed a reduction in the number of flowers under terminal drought. The mean number of flowers for the terminal drought was 232.2 and 188.8 for Dy7 and Ym respectively (Fig. 3A) while for the early transitory drought the mean number of flowers was 264.2 and 200.4 for Dy7 and Ym respectively (Fig. 3B). In addition, a significant reduction was recorded in the mean flower number per plant of both cultivars grown in reduced field water capacity. Nevertheless, the mean number of flowers per plant produced by the water stressed plant of Ym was limited as compared with WS plants of Dy7; the water stressed Dy7 plants on the other hand also produced a reduced mean number of flowers per plant as compared with the well watered control plants of Dy7. The mean number of flowers in both cultivars reduced with a reduction in Field water capacity and the least mean number of flowers per plant was recorded for both cultivars in 20% FC (Fig. 3C).
Grain yield: The grain number is identified as the most important factor that contributes and determines the final yield of oat crops (Smith et al., 2012). The WS plants of Ym yielded less grain than Dy7. The well watered (WW) spikelets of Dy7 and Ym produced grains at the rate of 3 grains per spikelet while water stressed (WS) spikelets of Ym mostly formed only 2 mean number of grains per spikelet. The WW plant of Dy7 and Ym produced 323.6 and 279.4 grains per plant respectively, the mean grain number borne by the plants given a terminal drought was 186 and 143.8 in Dy7 and Ym plants respectively (Fig. 4A), whereas, the mean grain number in an early transitory drought was 231.4 and 154.6 in Dy7 and Ym respectively (Fig. 4B). Moreover, both cultivars exhibited a significant decline in mean grain number when grown in different field water capacities FC. The least mean number of grains was recorded at 20% FC, which was 152.4 and 100.8 for Dy7 and Ym respectively (Fig. 4C)
DISCUSSION
Crop plants generally encounter drought during different growth stages and oats specifically may be sensitive to drought because they are grown in poor soils where the other grain crops may not survive or their yields might be impaired. The present study investigated the effect of drought at two different growth stages of naked oat cultivars, namely the early ephemeral drought and drought at a terminal stage after the flowering had already started. The Avena nuda L. cultivars were also compared for their growth and yield when grown under different field water capacities during the drought stress and under normal irrigation in control plants. The cultivar Dy7 showed better growth and yield responses under reduced field water capacities and under terminal and early transitory drought in comparison with the Ym plants. The drought stress can reduce normal growth, plant height, leaf area and biomass in different field crops (Zheng et al., 2016). Similar to other growth components, the leaf area shows sensitivity to available water content in the soil and even a mild drought can harm leaf expansion by interfering with the normal cell enlargement and growth (Munns, 2011). Plants usually maintain sufficient moisture inside tissue by reducing leaf surface in response to the reduced soil water contents in order to prevent water loss from stomata (Zandalinas et al., 2018). Our findings are similar with Hütsch et al. (2015) who reported a reduction in the leaf area of maize plants due to drought as early as 34 - 40 DAS. In the present study, the leaf area was much reduced for the cultivar Ym that showed a reduction in all the physiological activities such as stomatal conductance and photosynthesis (data not shown).
Environmental stress can alter the plant morphology such as reducing number, surface area and size of the leaves. These changes in the plant morphology were noticed in the present study for the two Avena nuda L. cultivars. In agreement with our findings, previous studies have reported the decline in leaf number and leaf area under drought condition for wheat by Daryanto, Wang and Jacinthe (2016) and for maize and sorghum by Sato et al. (2020). Similar mechanism can be seen in crop plants that avoid loss of cell turgidity by reducing the surface area of the plants by reducing the number of leaves, leaf area and plant height as reported by Anjum et al. (2017). Unlike other growth responses, a reduced number of leaves may be beneficial for the plants because of the growing concentration of CO2 in the atmosphere that is expected to grow further in the coming future. The present study confirms the idea that crop plants with reduced number of leaves are actually better adapted to the drought and may produce close to normal yield in comparison with the plants that produce more leaves in the present scenario of global climate change (Srinivasan, Kumar and Long 2017). In the present study, Dy7 produced lesser number of leaves in all the watering schemes than the cultivar Ym while Dy7 could maintain higher yield.
Desiccation does not allow plants to maintain normal development and growth. The plants generally adopt strategies to rescue themselves from dehydration by three major behavioral adaptations (a) closing the stomata, (b) minimizing evapotranspiration and (c) reducing exchange of gases through the stomata. All these events compromise photosynthetic activity resulting in a stunted plant and shoot growth (Arifuzzaman et al., 2020). Canales et al. (2019) have recognized root length as an important morphological feature during drought condition. They compared two European oat varieties Flega and Patones the later showed better root length in seedling and terminal drought stress. Our results showed that water deficit induced drastic changes in the root length of Ym plants whereas; Dy7 exhibited a consistency for different water treatments and growth stages. Likewise Fang et al. (2017) have also reported an increase in root length in water stressed environments in the modern wheat (Titicum aestivum L.) cultivars. Increased root length under water stress is also associated with the concentration of abscisic acid (ABA) in leaves and other organs of the plant. Higher ABA concentrations increase the turgidity of guard cells, that in turn close the stomatal aperture and minimize the transpiration to retain the water contents inside the cells to minimize further root growth. Dong et al. (2020) have reported an increased concentration of ABA in drought stressed Arabidopsis thaliana L. In the present study Dy7 plants exhibited higher leaf ABA concentration (data not shown) and normal root growth while Ym had longer roots 105 DAS for water stressed plants and the leaf ABA concentration was lower than Dy7 plants.
We observed a decrease in the flower number under drought conditions especially the terminal drought caused a significant decrease in the total number of viable flowers produced by these cultivars. These observations are consistent with previously published studies (Sehgal et al., 2018) that under drought stress flower number may be decreased and have worst effect on the pollen viability and on occasions pistil can lose its function which account for reduced yield in the drought sensitive cultivars. The grain number is identified as the most important factor that contributes and determines the final yield of oat crops (Smith and Hamel, 2018). Abid et al. (2018) have reported grain yield losses due to drought nonetheless, the grain number and grain filling were higher in Dy7 spikelet as compared to the Ym undoubtedly due to better net photosynthetic rates, evapotranspiration and gaseous exchange in Dy7 (data not shown here), whereas the reduced number of leaves per plant caused a decrease in the total leaf area which benefits the final grain yield. Contrary to the terminal drought, the grain number was better in the oat cultivars that were subjected to a transitory water stress at the vegetative growth stage. Most probably, soil drying and changes in the hormonal balance of the plants are some of the factors responsible for reduced grain filling (Zong et al., 2020). Our findings are also consistent with Prasad et al. (2017), who reported a decrease in the final grain yield due to drought and other stress alone or in combination. The size of the seed was greater in early transitory water stress for Dy7 plants which maintained assimilative activities and better grain size and number compared with the Ym plants.
It is presumed in the current study that genetic and physiological features of Dy7 might have contributed towards its better performance under drought stress. Terminal drought has a negative impact on food translocation, because during grain filling the food reserves should be transported to the grains, as the endosperm requires food reserves to grow properly. The terminal drought during this time directs the food translocation towards the roots so they can grow longer in quest of water, such as in case of Ym plants was recorded in the present study, subsequently affecting reproductive organs and decline in seed numbers (Morales et al., 2020). The reduced yield is also attributed to pollen sterility and reduced pistil function because these functions are dependent on water availability. Aslam et al. (2015) have reported the reduced water in soil adversely effects the pollen tube elongation, pollen viability and pollen germination especially during the terminal stage when the pollens are produced, because all of these factors contribute towards reduced grain yield. Pushpavalli et al. (2015) have also reported a linear correlation between the soil water status that can directly affect the seed size and seed yield.
Conclusion: Dy7 maintained its yield under severe drought experimental conditions with 20% and 40% FC and moderate drought of 60% FC as compared to older Avena nuda L. variety Ym. Dy7 is a promising crop for soils with limited moisture availability both at vegetative and terminal stage drought stress. Cereal crop plants like Dy7 that can optimize their water use and yield in limited water resources are highly recommended for future agricultural practices, as a rise in drought frequency, magnitude and changes in raining patterns are expected throughout the world.
Acknowledgments: This study was financially supported by Sardar Bahadur Khan Women’s University Quetta, Balochistan, Pakistan under faculty development Split Ph.D. program (FDP) to the first author is highly acknowledged. The Ministry of Education, State Key Laboratory of Grassland Agro-ecosystems, School of Life Sciences, Lanzhou University, Lanzhou, 730000, China is greatly acknowledged for providing the laboratory facilities and experimental station where the study was accomplished.
Declaration of interest: All authors declare no conflict of interest in the accomplishment of this study.
Authors Contribution: XF, TY and HB designed and developed the methodology, AT analyzed statistical data, HB got funding acquisition, performed formal analysis and wrote the manuscript. All the authors have seen and approved the final version of the article for publication.
Fig. 1: Experimental plan schedule of sampling and data collection for different parameters

Fig. 2 Single Leaf area (cm2) (mean ± SD) of Dy7 and Ym plants following different water treatments. n = 5 five replicate pots per treatment and seven plants per pot. treatments: different field water capacities i.e. 80-90%, 60%, 40%, and 20% respectively. The different letters represent significant difference at (p ≤ 0.05)
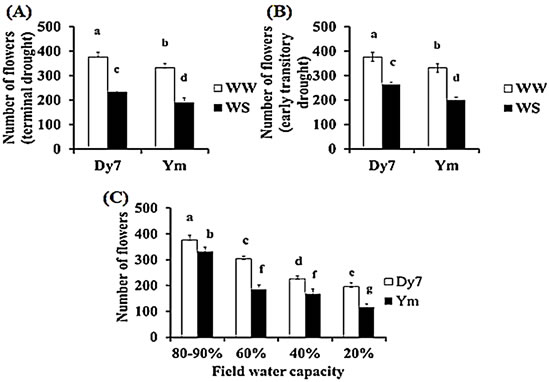
Fig. 3 (A) Flower number (mean ± SD) for Dy7 and Ym plant grown under different water treatments n = 5 five replicate pots per treatment and seven plants per pot treatments: terminal drought (65 -70 DAS) when flowering had already started and WW control (B) flower number (mean + SD) for Dy7 and Ym plants. n = 5 five replicate pots per treatment and seven plants per pot. treatments: early ephemeral drought (40 DAS) continued for 20-25 days and WW control (C) flower number (mean + SD) for Dy7 and Ym plants following different water treatments n = 5 five replicate pots per treatment and seven plants per pot treatments: different field water capacities of 80-90%, 60%, 40%, and 20% respectively. The different letters represent significant difference (p ≤ 0.05)
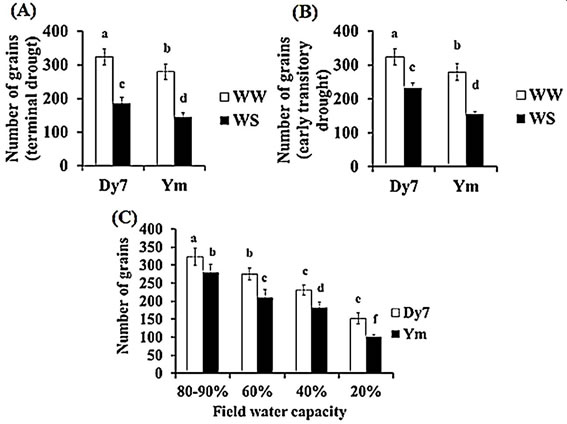
Fig. 4 (A) Grain number (mean ± SD) for Dy7 and Ym plants grown under different water treatments. n = 5 five replicate pots per treatment and seven plants per pot treatments: terminal drought (65-70) DAS and WW control (B) grain number (mean ± SD) for Dy7 and Ym plants grown under different water treatments. n = 5 five replicate pots for each cultivar with seven plants per pot treatments: early transitory drought (WS) (35-40 DAS) and WW control (C) number of grain (mean ± SD) for Dy7 and Ym plants grown under different water treatments. n = 5 five replicate pots for each cultivar with seven plants per pot treatments: different field water capacities of 80-90%, 60%, 40%, and 20% respectively. The different letters represent significant difference (p ≤ 0.05)
Table 1: Mean plant height (cm), root length (cm) and number of leaves for Dy7 and Ym plants grown under different water treatments.
|
Treatments
|
Plant Height (cm)
|
Root length
|
Number of leaves
|
|
FC (%)
|
Dy7
|
Ym
|
Dy7
|
Ym
|
Dy7
|
Ym
|
|
35 Days after sowing
|
|
80-90
60
40
20
|
23a
19.8b
17.8b
14.2c
|
24.8a
21.6b
19.8b
16.4c
|
10
9.2
9.2
9.2
|
9.6
9
8.6
8.4
|
3
3
2.4
2.4
|
3.2
2.8
2.8
2.4
|
|
75 Days after sowing
|
|
80-90
|
52a
|
54a
|
14
|
14
|
15.2a
|
16.2a
|
|
60
|
40.8b
|
42.8b
|
13.2
|
13.2
|
13.8b
|
14a
|
|
40
|
35.2c
|
39bc
|
12.8
|
12.6
|
11bc
|
13.6b
|
|
20
|
30.8d
|
34.4d
|
12.8
|
13
|
9.8c
|
10.4b
|
|
105 Days after sowing
|
|
80-90
|
64.6a
|
72.8a
|
22.4
|
23
|
25a
|
25.2a
|
|
60
|
56.4b
|
60.4b
|
20.2
|
20.4
|
21.4b
|
22.4b
|
|
40
20
|
53.2b
43.2c
|
60b
52.6c
|
19.4
20.2
|
20.4
23.2
|
20.6bc
17.8c
|
22b
18.2d
|
Treatments: Different field water capacities i.e. 80-90%, 60%, 40%, and 20%. The different letters represent significant variance amongst the different means (p ≤ 0.05) of a parameter and the cultivars. The values lacking any letters or same letter represent non-significant variance (p ≤ 0.05).
REFERENCES
- Abid, M., Z. Tian, R. Zahoor, S.T. Ata-Ul-Karim, C. Daryl, J.L. Snider and T. Dai (2018). Chapter two—pre-drought priming: a key drought tolerance engine in support of grain development in wheat. In: Sparks DL (ed) Advances in agronomy, vol 152. Academic Press, Cambridge, 51-85 pp.
- Anjum, S.A., U. Ashraf, A. Zohaib, M. Tanveer, M. Naeem, I. Ali, T. Tabassum, and U. Nazir (2017). Growth and development responses of crop plants under drought stress: a review'. Zemdirbyste. 104(3): 267-276.
- Arifuzzaman, M., S. Barman, S. Hayder, M.A.K. Azad, M.T.S. Turin, M.A. Amzad and M.S. Masuda (2020). Screening of bread wheat (Triticum aestivum) genotypes under drought stress conditions using multivariate analysis. Cereal Res. Commun. 48:301-308.
- Aslam, M., M.S.I. Zamir, S.A. Anjum, I. Khan, and M. Tanveer (2015). An investigation into morphological and physiological approaches to screen maize (Zea mays) hybrids for drought tolerance. Cereal Res. Commun. 43(1): 41-51.
- Canales, F.J., K.A Nagel, C. Müller, N. Rispail and E. Prats (2019). Deciphering Root Architectural Traits Involved to Cope With Water Deficit in Oat. Front. Plant Sci. 10: 1558.
- Canales, F.J., G. Montilla-Bascón, N. Rispail, and E. Prats (2019). Salicylic acid regulates polyamine biosynthesis during drought responses in oat. Plant Signal. Behav. 14(10): e1651183–e1651184.
- Daryanto, S., L. Wang and P.A. Jacinthe (2016). Global Synthesis of Drought Effects on Maize and Wheat Production. PlOS ONE. 11(5): e0156362.
- Dong, B., X. Zheng, H. Liu, J.A. Able, H. Yang and H. Zhao (2017). Effects of drought stress on pollen sterility, grain yield, abscisic acid and protective enzymes in two winter wheat cultivars. Front. Plant Sci. 8: 1008.
- Dong, H., X. Ma, P. Zhang, H. Wang, X. Li, J. Liu, L. Bai and Y. Song (2020). Characterization of Arabidopsis thalianaRoot-Related Mutants Reveals ABA Regulation of Plant Development and Drought Resistance. J. Plant Growth Regul. 39:1393-1401.
- Duncan, D.B. (1955). Multiple range and multiple F tests. Biometrics. 11: 1-42.
- Fang, Y., Y. Du, J. Wang, Wu, S. Qiao, B. Xu, S. Zhang, K.H.M. Siddique and Y. Chen (2017). Moderate Drought Stress Affected Root Growth and Grain Yield in Old, Modern and Newly Released Cultivars of Winter Wheat. Front. Plant Sci. 8: 672.
- Fisher, R.A. (1921). Studies in Crop Variation. I. An examination of the yield of dressed grain from Broadbalk. J. Agric. Sci. 11(2): 107-135.
- Gu, L., C. Jie, Y. Jiabo, C. Sylvia, H.W. Sullivan, G. Shenglian, Z. Liping and K. Jong-Suk (2020). Projected increases in magnitude and socioeconomic exposure of global droughts in and warmer climates. Hydrol. Earth Syst. Sci. 241: 451-472.
- Statista (2020). World grain production by type. Retreived from https://www.statista.com/statistics/263977/world-grain-production-by-type/
- Hütsch, B.W., S. Jung, and S. Schubert (2015). Comparison of salt and drought-stress effects on maize growth and yield formation with regard to acid invertase activity in the kernels. J. Agron. Crop Sci. 201: 353-367.
- Ibrahim, M.S., A. Ahmed, A. Sohail and M.J. Asad, (2020). Nutritional and functional characterization of different oat (Avena sativa) cultivars. Int J. Food Prop. 23(1): 1373-1385.
- Isidro-Sánchez, J., E. Prats, C. Howarth, T. Langdon, and G. Montilla-Bascón (2020). Genomic Approaches for Climate Resilience Breeding in Oats. In: Kole C. (eds) Genomic Designing of Climate-Smart Cereal Crops. Springer, Cham. 133-169 pp.
- Mi, N., F. Cai, Y. Zhang, R. Ji, S. Zhang, and Y. Wang (2018). Differential responses of maize yield to drought at vegetative and reproductive stages. Plant Soil Environ. 64: 260-267.
- Morales, F., M. Ancín, D. Fakhet, J. González-Torralba, L. Angie, Gámez , A. Seminario, D. Soba, S.B. Mariem, M. Garriga and I. Aranjuelo (2020). Photosynthetic metabolism under stressful growth conditions as a bases for crop breeding and yield improvement. Plants. 9(1): 88.
- Munns, R. (2011). Plant adaptations to salt and water stress: differences and commonalities. Adv. Bot. Res. 57: 1-32.
- Ogbaga, C., P. Stepien and G. Johnson (2014). Sorghum (Sorghum bicolor) varieties adopt strongly contrasting strategies in response to drought. Physiol. Plant. 152: 389-401.
- Prasad, P.V.V., R. Bheemanahalli, and S.K. Jagdish (2017). Field crops and the fear of heat stress-opportunities, challenges, and future directions. Field. Crops. Res. 200: 114-121.
- Pushpavalli, R., M. Zaman-Allah, N.C. Turner, R. Baddam, M.V. Rao and V. Vadez (2015). Higher flower and seed number leads to higher yield under water stress conditions imposed during reproduction in chickpea. Funct. Plant. Biol. 42: 162-174.
- Sato, G.J., M.K. Joshua, C. Ngongondo, F. Chipungu, C. Malidadi and M. Monjerezi (2020). Evaluation of Different Tillage Systems for Improved Agricultural Production in Drought-Prone Areas of Malawi. In: Matondo, J.I., B.F. Alemaw, W.J.P. Sandwidi (eds) Climate Variability and Change in Africa. Sustainable Development Goals Series. Springer, Cham 157-167 pp.
- Sehgal, A., K. Sita, K.H.M. Siddique, R. Kumar, S. Bhogireddy, R.K. Varshney, R.B. Hanumantha, R.M. Nair, P.V.V. Prasad and H. Nayyar (2018). Drought or/and Heat-Stress Effects on Seed Filling in Food Crops: Impacts on Functional Biochemistry, Seed Yields, and Nutritional Quality. Front. Plant Sci. 48: 1705.
- Smith, D.L. and C. Hamel (2012). Crop yield: Physiology and Processes Crop Yield. Springer-Verlag, Berlin, Heidelberg. 64 p.
- Srinivasan, V., P. Kumar and S. Long (2017). Decreasing, not increasing, leaf area will raise crop yields under global atmospheric change. Global Change Biol. 23: 1626-1635.
- Wang, T., Y.L. Du, H. Jin, N.C. Turner, B. Wang, C. Zhang, T. Cui and F.M. Li (2017). Recently released genotypes of naked oat (Avena nuda) out-yield early releases under water-limited conditions by greater reproductive allocation and desiccation tolerance. Field Crops Res. 204: 169-179.
- Zandalinas, S.I., R. Mittler, D. Balfagón, V. Arbona and A. Gómez-Cadenas (2018). Plant adaptations to the combination of drought and high temperatures. J. Plant Physiol. 162: 2-12.
- Yu, H., Q. Zhang, P. Sun and C. Song (2018). Impact of Droughts on Winter Wheat Yield in Different Growth Stages during 2001–2016 in Eastern China. J. Disaster Risk Sc.9:376-391.
- Zhao, N., N. Fu, D. Li, L.J. Wang and X.D. Chen (2017). Study on mechanical properties for shearing breakage of oat kernel. Int. J. Food Eng. 14(2): 1-9.
- Zheng, M., Y. Tao, S. Hussain, Q. Jiang, S. Peng, J. Huang, K. Cui and L. Nie (2016). Seed priming in dry direct-seeded rice: consequences for emergence, seedling growth and associated metabolic events under drought stress. Plant Growth Regul.78: 167-178.
- Zong, N., H. Wang, Z. Li, M. Li, X. Li, J Pang, Y. Fan and J. Zhao (2020). Maize NCP1 negatively regulates drought and ABA responses through interacting with and inhibiting the activity of transcription factor ABP9. Plant Mol. Biol. 102: 339-357.
|





