MICROSATELLITE MARKERS BASED
ASSESSMENT OF RUST RESISTANCE GENES IN PAKISTANI BREAD WHEAT GENOTYPES
N. Akhtar1*, A. Kiran1, Sajid-ur-Rehman2, M. Kausar1, M. Khan1, U. Saleem1 and T. Mahmood3
1
Department of Plant Breeding and Genetics, College of Agriculture, University of Sargodha, Sargodha, 40100, Pakistan
2
Agriculture Biotechnology Research
Institute, Ayub Agriculture Research Institute, Faisalabad, 38000, Pakistan
3
Department of Plant Breeding and Genetics, PMAS Arid Agriculture University, Rawalpindi, 46000, Pakistan
*
Corresponding Author's email: naeem.siraj@uos.edu.pk , naeem.uca@gmail.com
ABSTRACT
Development of rust resistant wheat cultivars to meet ever-increasing wheat production targets for national food security in climate change scenario is currently at high priority in Pakistan. Focusing on this, here we report the screening of 40 wheat genotypes including advanced breeding lines and commercial
varieties through PCR based DNA markers to detect genes conferring resistance against different rust
races. In this study 22 SSR/STS markers were used which included nine leaf rust (Lr19, Lr22a, Lr27, Lr28, Lr34, Lr35, Lr37, Lr46 and Lr67), eight stripe rust (Yr5, Yr15, Yr17, Yr18, Yr26, Yr29, Yr30 and Yr46) and seven stem rust resistance genes (Sr2, Sr22, Sr24, Sr25, Sr26, Sr38 and Sr39). The results
revealed that allelic frequency for rust resistance genes (Lr, Yr and Sr) was maximum (70.4%) in three advanced wheat lines namely V-9452, V-11153 and NNG-3. However, minimum
frequency (51.9%) was found in 25 genotypes. Three advanced lines NNG-3,
V-9432 and V-11153 had
maximum number of rust
resistance genes among all
the reported wheat genotypes which indicated narrow
genetic base of Pakistani
wheat cultivars for rust
resistance. Therefore, it is suggested that newly
discovered rust resistance genes especially Lr and Yr should be used along with existing genes for effective and durable rust resistance in new wheat varieties.
Key words:
Gene, heat map, DNA
markers, PCR, SSR.
https://doi.org/10.36899/JAPS.2021.3.0285
Published online November 11, 2020
INTRODUCTION
Wheat (Triticum aestivum L.) is widely grown and consumed food crop worldwide. It gives about 20%
of calories consumed by the world population. Its
importance is increasing with
increasing global population (Tadesse et al., 2013; Akhtar et al., 2018). It is also an important cereal crop of Pakistan due to its consumption as staple food of
more than 220 million
populations. In Pakistan, bread wheat is cultivated on 8740 thousand hectares having production of 25.195 million tons (Government of Pakistan, 2019). Although Pakistan has attained self-sufficiency in
wheat production but per
hectare yield is still lower than other wheat producing countries (FAO, 2018).
Therefore, advanced technologies need to be adopted to
save wheat crop from biotic and abiotic stresses and to enhance wheat production to fulfill the food requirements of large
population.
It is observed that in Pakistan rusts reduce wheat yield nearly 10% every year in later growth stages. But in early growth stages, yield losses may have reached up to 20% (Ejaz et al., 2012; Singh et al., 2016). Wheat
rusts caused by Puccinia spp. are among the major production
constraints worldwide (Qamar et al., 2014; Bhardwaj et al., 2019). Leaf rust (Lr) attack on wheat occurs more frequently than stripe rust (Yr) or stem rust (Sr).
Pakistan suffered from leaf
rust epidemics in 1972-73 when the disease severity level was 30-50% while in 1976-78, disease severity
increased up to 50-80% (Khan, 1997). Similarly, in 1995, 2005 and 2019 stripe rust and leaf rust
epidemics have caused significant yield losses (Qamar et al., 2014 and
Gessese, 2019). The emergence
of new races of stem rust (Sr) has brought a major challenge of world wheat production
(Gessese, 2019) The prime
objective of wheat breeders is to develop new wheat varieties with optimum levels of disease resistance (
McCallum
et al
., 2016). The wheat germplasm has a large set of resistance genes and more than 50 wheat rust
resistant genes for each of the three rust species have been reported by different wheat researchers
(Pretorius et al., 2012; Pal et al., 2015; Gessese, 2019).
Previously, conventional breeding
techniques were used for
development of new rust
resistant wheat genotypes through gene recombination but
recently, marker assisted selection, by far, the most
efficient and broadly used tool for the monitoring of Pst pathotypes and
threats of Ug99-complex races
(Bhardwaj et al., 2019; Gessese, 2019). Microsatellites (SSR/STS) as reported in the literature are one of the most suitable molecular markers for the detection of rust resistance genes in hexaploid wheat (Singh et al., 2016). At present, approximately
2,800 SSRs have been reported and genetically mapped in wheat genome to investigate wheat rust resistant genes.
This study was designed to identify rust resistant genes in candidate wheat varieties and existing commercial
cultivars by using DNA based markers tightly linked to rust resistance genes in bread wheat.
MATERIALS AND METHODS
Healthy seeds of 40 wheat genotypes (30 advanced lines and 10 commercial varieties) were sown in the
experimental area of Agricultural Biotechnology Research Institute (ABRI), Ayub Agriculture
Research Institute, Faisalabad, Pakistan in Alpha-Lattice design with two replications (Table 1). At tillering stage, 2-3 fresh leaves of selected plants from each genotype were collected and stored at -20oC. A
modified CTAB method was used to extract DNA of
studied wheat genotypes (Rahman et al., 2002).
The quality of extracted DNA was analyzed by gel electrophoresis using 0.8 % agarose gel. The concentration of total genomic DNA of 40 wheat lines/varieties was determined by an automated NanoDrop-1000 3.3.1
apparatus. Quantified DNA was
diluted to the working
concentration of about 30
ng/µl.
A total of 22 Simple Sequence Repeats (SSR) and Simple Tendom Sequence (STS) markers were used in the experiment to identify genes that conferred
resistance to various rust
races in wheat genotypes (Table 2) along with 20
gene differentials to confirm
the presence of resistant gene of interest in studied wheat genotypes. DNA
amplification was done in a thermo-cycler machine using
volume of 20 μl per sample having 2 μl 10X Taq buffer, 2 μl MgCl2 (25mM), 1 μl each of
dATP, dCTP, dTTP and dGTP (2.5 mM), 1.5 μl primer (30 ng/ μl), 2 μl of genomic DNA (30 ng/μl), 0.2 μl (5unit/μl) Taq DNA Polymerase and 8.3 μl ddH2O.
Amplification reactions were programmed for 40 cycles. Amplification products were electrophoresed
in 3g/l agarose gel. DNA ladder of 1000bp with known molecular weight bands was loaded along with PCR amplified DNA products on gel to determine the size/molecular weight of the polymorphic DNA fragments. Then amplified products were photographed using Syngene Gel Documentation System. Polyacrylamide Gel Electrophoresis (PAGE) was also performed for those samples which had small DNA fragments to study them precisely. Presence of rust resistant gene with adequate band size was scored as positive (+) while susceptible gene was scored as negative (-) as shown in Tables
6,7 & 8.
Estimation of allelic frequency (%) and allelic diversity were determined by R. and Power Maker V.0.3 software package. The
polygenetic analysis with reference to rust resistant genes was determined by using R-software version 3.0.2. All amplified loci based on SSR/STS banding pattern were recorded as present (1) and absent (0) in
the 40 genotypes. The bivariate 1-0 was used to
construct heat map dendrogram
by using cluster method between group linkage and genetic distance was determined
through Squared Euclidean Distance.
RESULTS
A total of 22 molecular markers (SSR/STS) were used in this study to
determine the presence/absence of rust resistance genes (Lr,Yr & Sr) in 40
wheat genotypes (Table 2).
Xgwm533, Xwmc44, Xgwm11, Lr28,
csLV34, Sr26#43, BF145935, Xcfd23, Xcfd71, Xbarc352, cfa2123,
SCS253 and Xgwm296 provided
reproducible results whereas csSr2, Sr39#22r, Sr39F2, STS7/8, STS9/10, BARC71, Xgwm437,
Ventriup LN2 and SCS265 failed to amplify fragments for Sr24, Lr37 and Yr5
due to some experimental error. Gene differentials (20) were also used to confirm band size of rust
resistant genes in studied
wheat genotypes. Regarding leaf rust (Lr) resistance genes, nine genes were investigated. Among these, five leaf rust (Lr) resistance genes Lr.19. Lr.22a, Lr.28, Lr.35 and Lr.67 genes were identified in all studied wheat genotypes while Lr.24 and Lr.37 were not
found. Whereas, Lr.34 and Lr.46 genes were present in 10-B9346 and NR-310 wheat genotypes, respectively (Table 3). Regarding eight studied stripe rust resistance
conferring genes, only Yr.15,
Yr.26 Yr.30 and Yr.46 were observed in all 40
wheat genotypes while Yr.5 and Yr.17 were absent in all genotypes. Stripe rust resistance genes Yr.18 and Yr. 29 were also
identified in limited number of wheat genotypes i.e. 10B9346 and 10B2003 genotypes,
respectively (Table 4). Stem
rust resistance genes were
examined in current wheat
genotypes by using seven Sr. markers and it was
observed that only Sr.2, Sr.22, Sr.25 and Sr.39 were present in all 40
wheat genotypes to confer stem rust resistance while Sr. 24, Sr.26 and Sr.
38 were lacking in all genotypes (Table 5).
Phenotypic evaluation of wheat
genotypes for leaf, stripe and stem rusts is presented in Table 9.
Allelic frequency of rust
resistance genes (Lr, Yr and Sr) was observed in studied wheat genotypes and found maximum frequency
(70.4%) in 3 advance lines namely V-9452, V-11153 and NNG-3. However minimum
frequency (51.9%) was estimated in twenty-five genotypes in which 10 were commercial varieties and other 15 were advanced wheat lines (Fig 1).
The genetic association among
advanced wheat breeding lines
for rust resistance genes was determined by developing heat map based on the
SSR/STS banding patterns. Heat
map indicated two groups;
Group-A contained three clusters indicating wheat genotypes
along Y-axis, whereas, Group-B
indicated clusters of rust
resistance genes (along with
their primers) on X-axis (Fig 2). Red colour in
map indicated presence of rust resistance genes in respective genotypes. However, yellow
colour indicated absence of
required genes. Cluster-A comprised of two leaves. Leaf-1 contained two genes Sr25 and Lr19 while leaf-2 had 11 genes (Lr19, Lr22a, Lr28, Lr67, Yr49, Sr2, Yr30, Sr39, Sr22 and Yr15), which indicated the existence of rust resistance genes in 40 wheat genotypes. Cluster-B
discriminated five rust resistance genes (Lr34, Lr46, Yr18, Yr29 and Sr57) in
red and yellow patches and cluster-C indicated five genes (Yr5, Sr24, Lr37, Yr17 and Sr38) in yellow patch for absence of resistance genes of rust. In group-B the cluster A' comprised of two leaves. Leaf 1 contains three genotypes (NNG-3, V-9452 and V-11153) with maximum allelic frequency of rust resistance genes (70.4%),
whereas leaf 2 contains eight genotypes (AARI-11, Ufaq-2002, TW86014, V-10355, 09B9110,
V-11161, NR-310 and 10B2003)
with 63% allelic frequency.
Cluster B' comprised of two leaves 4 and 5.
Leaf 4 contained the largest number of 24 genotypes with allelic frequency of 51.9%. However, leaf 5 contains three genotypes
(NR-378, V-11156 and TW96018)
showing 59.3% allelic frequency. Cluster C' comprised of Leaf 3 contains only one genotype (Seher-06) with 59.3% allelic frequency.
All this characterization of
leaf rust, stripe rust and stem rust resistant genes represented by + and - signs in wheat
genotypes showed in tables 6,7 and 8, respectively.
Sr.25 is one of the
stem rust resistance gene which exhibits strong linkage with Lr.19 in wheat for
adult plant resistance. In this study, the primer BF145935 amplified 198 bp and 180bp DNA fragments of Sr.25 gene in all
the studied wheat genotypes
which conferred stem rust
resistance at two fragment
lengths (Fig 3). DNA fragment length of 330 bp
linked with primer Lr28
indicated the presence of leaf rust resistance gene Lr.28 in all studied wheat genotypes (Fig 4). The primer Xgwm11 amplified 213bp DNA fragment length for Yr. 15 gene in most of the studied wheat genotypes which indicated the occurrence of stripe rust resistance gene. The banding pattern on agarose gel only for 10 wheat
genotypes. The results of
remaining gels are not reported here (Fig 5).
Table 1. Name of wheat varieties and advanced
breeding lines as supplementary material.
|
S. No.
|
Name of wheat
genotypes
|
Source
|
S. No.
|
Name of wheat
genotypes
|
Source
|
|
1.
|
NR-399
|
NARC, Islamabad
|
21.
|
V-10031
|
WRI Faisalabad
|
|
2.
|
V-9452
|
UAF Faisalabad
|
22.
|
Shafaq-06
|
WRI Faisalabad
|
|
3.
|
V-11153
|
WRI Faisalabad
|
23.
|
SH-2002
|
WRI Faisalabad
|
|
4.
|
V-11154
|
WRI Faisalabad
|
24.
|
V-10104
|
WRI Faisalabad
|
|
5.
|
NNG-3
|
NIBGE Faisalabad
|
25.
|
TW96018
|
AZRI Bhakkar
|
|
6.
|
V-11156
|
WRI Faisalabad
|
26.
|
V-10110
|
WRI Faisalabad
|
|
7.
|
NR-310
|
NARC, Faisalabad
|
27.
|
V-11166
|
WRI Faisalabad
|
|
8.
|
V-11160
|
WRI Faisalabad
|
28.
|
NR-400
|
NARC, Islamabad
|
|
9.
|
V-10B2003
|
RARI Bahwalpur
|
29.
|
V-TW86014
|
AZRI Bhakkar
|
|
10.
|
V-11161
|
WRI Faisalabad
|
30.
|
NR-399
|
NARC, Islamabad
|
|
11.
|
V-10-B9346
|
RARI Bahwalpur
|
31.
|
NR-378
|
NARC, Islamabad
|
|
12.
|
V-11164
|
WRI Faisalabad
|
32.
|
Pb-11
|
WRI Faisalabad
|
|
13.
|
V-09B9110
|
RARI Bahwalpur
|
33.
|
Galaxy-13
|
WRI Faisalabad
|
|
14.
|
V-10355
|
WRI Faisalabad
|
34.
|
Ufaq-2002
|
WRI Faisalabad
|
|
15.
|
V-09-BT043
|
ABRI Faisalabad
|
35.
|
AARI-11
|
WRI Faisalabad
|
|
16.
|
V-10217
|
WRI Faisalabad
|
36.
|
Sehar-06
|
WRI Faisalabad
|
|
17.
|
V-10-BT002
|
ABRI Faisalabad
|
37.
|
Inq-91
|
WRI Faisalabad
|
|
18.
|
V-09B9172
|
RARI Bahwalpur
|
38.
|
Millat-11
|
WRI Faisalabad
|
|
19.
|
V-10025
|
WRI Faisalabad
|
39.
|
Fsd-08
|
WRI Faisalabad
|
|
20.
|
V-10193
|
WRI Faisalabad
|
40.
|
Lasani-08
|
WRI Faisalabad
|
Table 2. List of SSR/STS markers demonstrating in
the experiment.
|
Sr. #
|
Primer Name
|
Linked gene
|
Location
|
Size (bp)
|
Reference
|
|
1
|
Xgwm 296
|
Lr22a
|
2DS
|
121/131
|
Hiebert et al., 2007
|
|
|
2
|
Xwmc 44
|
Lr46/Yr29
|
1BL
|
242
|
Suenaga et al. (2001)
|
|
|
3
|
Xgwm 437
|
Lr19
|
7DL
|
130
|
(
http://maswheat.ucdavis.edu
)
|
|
|
4
|
X-barc 352
|
Lr34/Yr18
|
4D.7DS
|
250
|
(
http://maswheat.ucdavis.edu
)
|
|
|
5
|
Xgwm-11
|
Yr15/Yr26
|
1B,1BS
|
213
|
(
http://maswheat.ucdavis.edu
)
|
|
|
6
|
Xgwm-533
|
Sr2
|
3B
|
120
|
Spielmeyer et al., (2003)
|
|
|
7
|
Cfa2123
|
Sr22
|
7A
|
245/260
|
Hiebert et al., 2010
|
|
|
8
|
csSr2
|
Sr2/Yr30
|
3B
|
378
|
(
http://maswheat.ucdavis.edu
)
|
|
|
9
|
Lr28
|
Lr28
|
4AL
|
330
|
(http://www.graingenes.com)
|
|
|
10
|
BF145935
|
Sr25/Lr19
|
7D.7A
|
180/198
|
Mago et al.,
2011
|
|
|
11
|
STS(7/8)
|
Yr5
|
1B
|
500
|
Campbell et al., 2009
|
|
|
12
|
Sr26#43
|
Sr26
|
6AS.6AL
|
206
|
Liu et al.,
2010
|
|
|
13
|
csLv34
|
Lr34/Yr18
|
7DS
|
150
|
(
http://maswheat.ucdavis.edu
)
|
|
|
14
|
Sr39#22r
|
Sr39/Lr35
|
2B
|
487
|
Mago et al.,
2011
|
|
|
15
|
Xcfd71
|
Lr67/Yr26
|
4DL
|
214
|
Hiebert et al., 2010
|
|
|
16
|
Xcfd23
|
Lr67
|
4DL
|
211
|
Hiebert et al., 2010
|
|
|
17
|
BARC71
|
Sr24/Lr24
|
3DL
|
85/103
|
(
http://maswheat.ucdavis.edu
)
|
|
|
18
|
STS-9/10
|
Yr5/Yr17
|
2BL
|
439
|
(
http://maswheat.ucdavis.edu
)
|
|
|
19
|
SCS253
|
Lr19
|
7D.7A
|
736
|
(
http://maswheat.ucdavis.edu
)
|
|
|
20
|
Ventriup LN2
|
Lr37/Sr38
|
2AS
|
259
|
(
http://maswheat.ucdavis.edu
)
|
|
|
21
|
SCS265
|
Lr19
|
7D.7Ag
|
512
|
(
http://maswheat.ucdavis.edu
)
|
|
|
22
|
Sr39F2
|
Sr39
|
2BS
|
900
|
(
http://maswheat.ucdavis.edu
)
|
|
|
|
|
|
|
|
|
|
|
Table 3. Wheat genotypes
showing presence/absence of leaf rust (Lr) resistance of genes.
|
Sr. #
|
Primers
|
Wheat genotypes
|
Lr genes
|
|
1
|
SCS265
|
All genotypes were
positive for this marker.
|
(Lr19) +ve
|
|
2
|
Xgwm 296
|
All genotypes were
positive for this marker.
|
(Lr22a) +ve
|
|
3
|
BARC71
|
All genotypes were
negative for this marker.
|
(Lr24) -ve
|
|
4
|
Lr28
|
All genotypes were
positive for this marker.
|
(Lr28)+ve
|
|
5
|
Xbarc-352Lr34
|
9452, 11153, NNG-3,
NR-310, 10B2003, 11161,
09B9110, 10355, TW86014,
Ufaq-2002, AARI-11 were
positive for this marker.
|
(Lr34) +ve
|
|
6
|
Sr39#22r
|
All genotypes were
positive for this marker.
|
(Lr35) +ve
|
|
7
|
VentriupLN2
|
All genotypes were
negative for this marker.
|
(Lr37) -ve
|
|
8
|
Xwmc-44
|
9452, 11153, NNG-3,
NR-378, 11156, TW96018,
Sehar-06 were positive for this marker.
|
(Lr46) +ve
|
|
9
|
Xcfd 23
|
All genotypes were
positive for this marker.
|
(Lr67)+ve
|
+ = Presence & - = Absence of leaf
rust resistance genes
Table 4. Wheat genotypes
showing presence/absence of stripe rust (Yr) resistance
genes.
|
Sr. #
|
Primers
|
Wheat genotypes
|
Yr genes
|
|
1
|
STS(7-8)
|
All genotypes were
negative for this
marker.
|
(Yr5) -ve
|
|
2
|
Xgwm-11
|
All genotypes were
positive for this
marker.
|
(Yr15 ) +ve
|
|
3
|
STS9/10
|
All genotypes were
negative for this
marker.
|
(Yr17) -ve
|
|
4
|
csLv34
|
9452, 11153, NNG-3,
NR-310, 10B2003, 10355, 11161, TW86014,
Ufaq-2002, AARI-11, 09B9110 were positive for this marker.
|
(Yr18) +ve
|
|
5
|
Xgwm-11
|
All genotypes were
positive for this
marker.
|
(Yr26) + ve
|
|
6
|
Xwmc-44
|
9452, 11153, NNG-3,
NR-310, 11156, ,
10355, TW96018, NR-378, Sehar-06
|
(Yr29) +ve
|
|
7
|
Xgwm-533
|
All genotypes were
positive for this
marker.
|
(Yr30) +ve
|
|
8
|
Xcfd71
|
All genotypes were
positive for this
marker.
|
(Yr46) +ve
|
+ = Presence & - = Absence of stripe rust resistance genes
Table 5. Wheat genotypes
showing presence/absence of stem rust (Sr) resistance
genes.
|
Sr. #
|
Primers
|
Wheat genotypes
|
Sr genes
|
|
1
|
Xgwm-533
|
All genotypes were
positive for this
marker.
|
(Sr2) +ve
|
|
2
|
cfa2123
|
All genotypes were
positive for this
marker.
|
(Sr22) +ve
|
|
3
|
Sr24
|
All genotypes were
negative for this
marker.
|
(Sr24) -ve
|
|
4
|
BF145935
|
All genotypes were
positive for this
marker.
|
(Sr25) +ve
|
|
5
|
Sr26#43
|
All genotypes were
negative for this
marker.
|
(Sr26) -ve
|
|
6
|
Ventriup/LN2
|
All genotypes were
negative for this
marker.
|
(Sr38) -ve
|
|
7
|
Sr39#22r
|
All genotypes were
positive for this
marker.
|
(Sr39) +ve
|
+ = Presence & - = Absence of stem
rust resistance genes
Table 6. Characterization of Leaf Rust Resistant Genes (Lr) in Wheat Genotypes.
|
Sr. #
|
Genotypes
|
Lr19
|
Lr22a
|
Lr24
|
Lr28
|
Lr34
|
Lr35
|
Lr37
|
Lr46
|
Lr67
|
|
1.
|
NR-399
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
2.
|
V-9452
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
+
|
+
|
|
3.
|
V-11153
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
+
|
+
|
|
4.
|
V-11154
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
5.
|
NNG-3
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
+
|
+
|
|
6.
|
V-11156
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
+
|
+
|
|
7.
|
NR-310
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
8.
|
V-11160
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
9.
|
10B2003
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
10.
|
V-11161
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
11.
|
10-B9346
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
12.
|
V-11164
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
13.
|
09B9110
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
14.
|
V-10355
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
15.
|
09-BT043
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
16.
|
V-10217
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
17.
|
10-BT002
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
18.
|
09B9172
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
19.
|
V-10025
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
20.
|
V-10193
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
21.
|
V-10031
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
22.
|
Shafaq-06
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
23.
|
SH-2002
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
24.
|
V-10104
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
25.
|
TW96018
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
+
|
+
|
|
26.
|
V-10110
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
27.
|
V-11166
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
28.
|
NR-400
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
29.
|
TW86014
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
30.
|
KANZO
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
31.
|
NR-378
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
+
|
+
|
|
32.
|
Pb-11
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
33.
|
V-07096
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
34.
|
Ufaq-2002
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
35.
|
AARI-11
|
+
|
+
|
-
|
+
|
+
|
+
|
-
|
-
|
+
|
|
36.
|
Seher-06
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
+
|
+
|
|
37.
|
Inq-91
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
38.
|
Millat-11
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
39.
|
Fsd-08
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
|
40.
|
Lasani-08
|
+
|
+
|
-
|
+
|
-
|
+
|
-
|
-
|
+
|
*
+ = Presence of required gene-, - = Absence of required gene
Table 7. Characterization of Stripe Rust Resistant
Genes (Yr) in Wheat
Genotypes.
|
Sr. #
|
Genotypes
|
Yr5
|
Yr15
|
Yr17
|
Yr18
|
Yr26
|
Yr29
|
Yr30
|
Yr46
|
|
1.
|
NR-399
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
2.
|
V-9452
|
-
|
+
|
-
|
+
|
+
|
+
|
+
|
+
|
|
3.
|
V-11153
|
-
|
+
|
-
|
+
|
+
|
+
|
+
|
+
|
|
4.
|
V-11154
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
5.
|
NNG-3
|
-
|
+
|
-
|
+
|
+
|
+
|
+
|
+
|
|
6.
|
V-11156
|
-
|
+
|
-
|
-
|
+
|
+
|
+
|
+
|
|
7.
|
NR-310
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
8.
|
V-11160
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
9.
|
10B2003
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
10.
|
V-11161
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
11.
|
10-B9346
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
12.
|
V-11164
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
13.
|
09B9110
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
14.
|
V-10355
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
15.
|
09-BT043
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
16.
|
V-10217
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
17.
|
10-BT002
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
18.
|
09B9172
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
19.
|
V-10025
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
20.
|
V-10193
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
21.
|
V-10031
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
22.
|
Shafaq-06
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
23.
|
SH-2002
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
24.
|
V-10104
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
25.
|
TW96018
|
-
|
+
|
-
|
-
|
+
|
+
|
+
|
+
|
|
26.
|
V-10110
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
27.
|
V-11166
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
28.
|
NR-400
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
29.
|
TW86014
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
30.
|
KANZO
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
31.
|
NR-378
|
-
|
+
|
-
|
-
|
+
|
+
|
+
|
+
|
|
32.
|
Pb-11
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
33.
|
V-07096
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
34.
|
Ufaq-2002
|
-
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
35.
|
AARI-11
|
|
+
|
-
|
+
|
+
|
-
|
+
|
+
|
|
36.
|
Seher-06
|
-
|
+
|
-
|
-
|
+
|
+
|
+
|
+
|
|
37.
|
Inq-91
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
38.
|
Millat-11
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
39.
|
Fsd-08
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
|
40.
|
Lasani-08
|
-
|
+
|
-
|
-
|
+
|
-
|
+
|
+
|
*
+ = Presence of required gene, - = Absence of required gene
Table 8. Characterization of Stem Rust Resistant Genes (Sr) in Wheat Genotypes.
|
Sr. #
|
Genotypes
|
Sr2
|
Sr22
|
Sr24
|
Sr25
|
Sr26
|
Sr38
|
Sr39
|
|
1.
|
NR-399
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
2.
|
V-9452
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
3.
|
V-11153
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
4.
|
V-11154
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
5.
|
NNG-3
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
6.
|
V-11156
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
7.
|
NR-310
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
8.
|
V-11160
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
9.
|
10B2003
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
10.
|
V-11161
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
11.
|
10-B9346
|
+
|
+
|
-
|
-
|
-
|
-
|
+
|
|
12.
|
V-11164
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
13.
|
09B9110
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
14.
|
V-10355
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
15.
|
09-BT043
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
16.
|
V-10217
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
17.
|
10-BT002
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
18.
|
09B9172
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
19.
|
V-10025
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
20.
|
V-10193
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
21.
|
V-10031
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
22.
|
Shafaq-06
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
23.
|
SH-2002
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
24.
|
V-10104
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
25.
|
TW96018
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
26.
|
V-10110
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
27.
|
V-11166
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
28.
|
NR-400
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
29.
|
TW86014
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
30.
|
KANZO
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
31.
|
NR-378
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
32.
|
Pb-11
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
33.
|
V-07096
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
34.
|
Ufaq-2002
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
35.
|
AARI-11
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
36.
|
Seher-06
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
37.
|
Inq-91
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
38.
|
Millat-11
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
39.
|
Fsd-08
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
|
40.
|
Lasani-08
|
+
|
+
|
-
|
+
|
-
|
-
|
+
|
*
+ = Presence of required gene, - = Absence of required gene
Table 9. Phenotypic expression of wheat genotypes for rust (Lr, Yr and Sr) resistance.
|
Sr #
|
Genotype
|
Leaf Rust (Lr)
Resistance Genes
|
Stripe Rust (Yr)
Resistance Genes
|
Stem Rust (Sr)
Resistance Genes
|
|
1
|
NR-399
|
|
MR
|
|
|
|
|
MR
|
|
|
MR
|
|
|
|
2
|
V-9452
|
|
R
|
|
|
|
|
R
|
|
|
R
|
|
|
|
3
|
V-11153
|
|
R
|
|
|
|
|
R
|
|
|
R
|
|
|
|
4
|
11154
|
|
MR
|
|
|
|
|
MR
|
|
|
MR
|
|
|
|
5
|
NNG-3
|
|
R
|
|
|
|
|
R
|
|
|
R
|
|
|
|
6
|
V-11156
|
|
MR
|
|
|
|
|
MR
|
|
|
MR
|
|
|
|
7
|
NR-310
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
8
|
V-11160
|
|
MS
|
|
|
|
|
MS
|
|
|
MS
|
|
|
|
9
|
10B2003
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
10
|
V-11161
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
11
|
10-B9346
|
|
MS
|
|
|
|
|
MR
|
|
|
MR
|
|
|
|
12
|
V-11164
|
|
S
|
|
|
|
|
MS
|
|
|
MS
|
|
|
|
13
|
09B9110
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
14
|
V-10355
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
15
|
09-BT043
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
16
|
V-10217
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
17
|
10-BT002
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
18
|
09B9172
|
|
S
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
19
|
V-10025
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
20
|
V-10193
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
21
|
V-10031
|
|
MS
|
|
|
|
|
MR
|
|
|
MS
|
|
|
|
22
|
Shafaq-06
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
23
|
SH-2002
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
24
|
V-10104
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
25
|
TW96018
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
26
|
V-10110
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
27
|
V-11166
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
28
|
NR-400
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
29
|
TW86014
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
30
|
KANZO
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
31
|
NR-378
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
32
|
Pb-11
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
33
|
V07096
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
34
|
Ufaq-02
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
35
|
AARI-11
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
36
|
Seher-06
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
37
|
Inq-91
|
|
MS
|
|
|
|
|
MR
|
|
|
MR
|
|
|
|
38
|
Millet-11
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
39
|
Fsd-08
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
|
40
|
Lasani-08
|
|
MS
|
|
|
|
|
MS
|
|
|
MR
|
|
|
Note:
R: resistant, MR: moderately
resistant, MS: moderately susceptible and S: susceptible.

Figure 1. Allelic frequency of rust resistance genes in studied bread wheat genotypes.
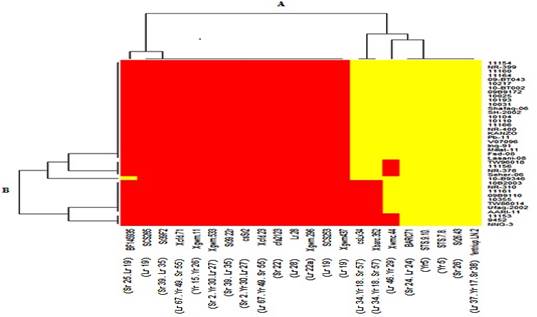
Figure 2. Heat map dandrogram showing genotype clusters along Y-axis and gene clusters (with name of primer) along X-axis. Red color in map indicated presence of rust resistance genes in respective genotypes. However, yellow color indicated absence of required genes.

Figure 3. Wheat genotypes showing resistance against stem rust (Sr.25/Lr.19) by primer BF145935.
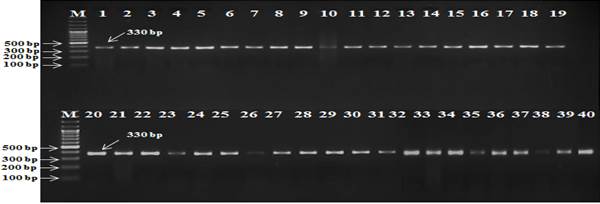
Figure 4. Wheat genotypes showing resistance against leaf rust (Lr.28) by primer Lr28.
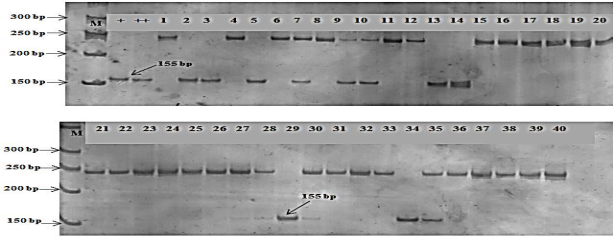
Figure 5. Wheat genotypes showing resistance against stripe rust (Yr. 18) by primer csLv34
DISCUSSION
The proposed study was aimed to screen advanced
lines/commercial varieties of bread wheat developed by various wheat breeding institutes of Pakistan using molecular
markers tightly linked with the genes conferring resistance to different rust races. Plant breeders broadly categorized
rust resistance genes into
seedling as well as adult plant resistance (APR) genes. The APR genes reveals durable, slow rusting,
non-specific and horizontal resistance which are effective against rust epidemics (Jeffrey et al., 2014). Basnet
et al., 2013 reported that at seedling stage rust resistance genes reveal the phenotypes of major effect and with varying infection types whereas most of the APR genes are
limited in effect with varying disease severity levels. In this study various durable, slow rusting/non-specific rust resistance gene (Lr34/Yr18,
Lr46/Yr29, Sr2/Yr30, Lr67/Yr49) have been observed. Based on the results, it was observed that only five out of nine reported Lr genes (Lr.19. Lr.22a, Lr.28, Lr.35 and Lr.67) were identified in all present wheat genotypes, while Lr.34 and Lr.46 genes were detected in 11 and 7 wheat genotypes respectively, which conferred
leaf rust resistance in studied wheat genotypes. This
indicated that a satisfactory
number of leaf rust genes were not present in present Pakistani wheat
genotypes and it was strongly suggested to broaden the wheat genetic back ground to cope with future rust epidemics (Qamar et al., 2014;
McCallum
et al
., 2016, Bhardwaj et al., 2019; Gessese, 2019). Though in some cases, the results were not as per expectation as Lr. 24 did not show any band on
agarose gel from the amplified DNA of studied wheat genotypes but previously these genes were observed in some bread wheat genotypes (Ejaz et al., 2012). This may be due to either improper amplification
of PCR products by the primers or absence of these genes in studied wheat genotypes.
A total of eight stripe rust (Yr) resistance genes were observed and found four Yr resistance genes (Yr.15, Yr.26, Yr.30 & Yr.46) in all studied wheat genotypes except Yr.5 and Yr.17 which did not show the bands
on agarose gel. This might be the failure of
STS primers to amplify the PCR products. Yr.18 and Yr.29 were also observed in different wheat genotypes. This indicated that Pakistani wheat genotypes have enough resistance against stripe rust pathogens. Our results get support from the results of Ejaz et al., (2012); Yu et al., (2012); Parveen et al
., (2014); Wang et al
., (2015); Bhardwaj et al., 2019).
Stem rust (Sr) is an important disease of wheat throughout the world and resistance against Sr in newly developed wheat
genotypes is very important to combat foreseen threat of Ug99 epidemics in Pakistan. A total of seven
stem rust resistance genes
(Sr2, Sr22, Sr24, Sr25, Sr26, Sr38 and Sr39) were studied in 40 wheat genotypes using already
reported primers which amplified the DNA fragments in wheat. Among these four stem rust genes (Sr2, Sr22, Sr25 and Sr39) were found in all
studied wheat genotypes while
the remaining did not show bands on agarose gel. It reveals that although Pakistani wheat genotypes have enough resistance against stem rust and did not
identified in Pakistan yet but chances are there to spread in Pakistan through Iran and currently Ug99 resistant wheat genotypes may become susceptible to this virulent. It is an
unremitting straggle and should be continued in future to save wheat from
rusts. Furthermore, rust resistant genes (Lr, Yr, Sr)
as discovered world widely
should be introduced in our wheat breeding program to add in new breeding lines through hybridization.
Our results get support from the conclusions of Parveen et al., (2014);
Kosgey et al., (2015); Bhardwaj et al.,
(2019); Gessese, (2019). However, Ejaz et al., (2012) found contrary results and reported insufficient stem
resistance in Pakistani wheat
genotypes. This might be due to study of different stem rust genes in
different wheat genotypes.
Conclusion:
The results of the study revealed that Pakistani wheat genotypes have enough
resistance against stem rust
(Sr) pathogens yet but
satisfactory number of genes
are not available for leaf rust (Lr) and stripe rust (Yr). Therefore, it is strongly proposed that local wheat germplasm should be strengthen through
introduction of rusts resistant exotic genotypes developed by internationally to cope the future rust epidemics.
Furthermore, in current climate change scenario, it is the need of hour to add in maximum discovered rusts resistance genes in newly developing candidate
wheat lines to ensure food security in Pakistan.
Acknowledgement:
The research project was
conducted by the research fund provided by University of Sargodha under the program "Research and Development".
REFERENCES
- Akhtar, N., A. Waseem, T. Mehmood, S. Bano, A. Raza and Ahsan Aziz (2018). Gene action appraisal for seed yield and related traits in bread wheat. The J. Anim. & Plant Sci, 28: 1457-1465.
- Ashfaq, M., F. Zulfiqar, I. Sarwar, M.A. Quddus and I.A. Baig. (2011). Impact of climate change on wheat productivity in mixed cropping system of Punjab. Soil Environ. 30: 110-114.
- Basnet, B.R., P.R. Singh, S.A. Herrera-Foessel, A.M.H. Ibrahim, J. Huerta-Espino,V. Calvo-Salazar and J.C. Rudd. (2013). Genetic analysis of adult plant resistance to yellow rust and leaf rust in common spring wheat quaiu 3. Pl. Dis. 97: 728-736.
- Bhardwaj S. C. G.P. Singh., O, P. Gangwar., P, Prasad and S. Kumar. (2019). Status of Wheat Rust Research and Progress in Rust Management-Indian Context. Agron., 9:1-14.
- Doyle, J.J and J.L. Doyle. (1987). A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull. 19: 11-15.
- Ejaz, M., M. Iqbal, A. Shahzad, A.I. Atiq-ur-Rehman and G.M. Ali. (2012). Genetic variation for markers linked to stem rust resistance genes in Pakistani wheat varieties. Crop Sci. 52: 2038-2648.
- FAO. (2018). FAO statistical year book. Food and Agriculture organization (FAO) of the United Nations, Rome, Italy. Available at http://faostat3.fao.org/browse/Q/QC/E.
- Gessese, M. K. (2019) Description of Wheat Rusts and Their Virulence Variations Determined through Annual Pathotype Surveys and Controlled Multi-Pathotype Tests.Advan. Agri., 2019: 1-7.
- Government of Pakistan. (2019). Federal Bureau of Statistics. Economic Survey of Pakistan.
- Jeffrey, G.E., S.L. Evans, S. Wolfgang and N.D. Peter. (2014). The past, present and future of breeding rust resistant wheat. Fron. Pl. Sci. 5: 641-651.
- Khan, M.A. (1997). Evaluation of Multiple Regression Models based on Epidemiological Factors to predict Leaf Rust on Wheat. Pak. J. Agric. Sci. 34: 1-7.
- Kosgey, Z., J.O. Owuoche, M.A. Okiror and P.N. Njau. (2015). Inheritance of stem rust (Puccinia graminis Pers. F. Sp. Triticiericks and E. Hen) resistance in bread wheat (Triticum aestivum L.) lines to TTKST race. Int. J. Agron. Agric. Res.7: 1-13.
- McCallum, B.D., C.W. Hiebert, S. Cloutier, D. Bakkeren, S.B. Rosa, Humphreys DG, Marais GF, McCartney CA, Panwar V, Rampitsch C, Saville BJ, Wang X (2016). A review of wheat leaf rust research and the development of resistant cultivars in Canada. Canadian J Pl Path 38: 1-18.
- Njau, P.N. (2010). Identification and evaluation of sources of resistance to stem rust race Ug99 in wheat. Pl. Dis. 94: 413–419.
- Pal, D., S.C. Bhardwaj, D. Sharma, S. Kumari, M.V. Patial and P. Sharma. (2015). Assessment of Genetic Diversity and Validating Rust Resistance Gene Sources Using Molecular Markers in Wheat (Triticum aestivum L.). SABRAO J. Br. Genet. 47: 89-98.
- Parveen, Z., M. Iqbal, S. Rahman, M. Younis, M. Nawaz, S.H. Raza and M.Z. Iqbal. (2014). Rust resistance evaluation of advanced wheat (Triticum aestivum L.) genotypes using PCR-based DNA markers.Pak. J. Bot. 46: 251-257.
- Pretorius, Z.A., Y. Jin, C.M. Bender, L. Herselman, and R. Prins. (2012). Seedling resistance to stem rust race Ug99 and marker analysis for Sr2, Sr24 and Sr31 in South African wheat cultivars and lines. Euphy. 186:15-23.
- Qamar, M., S.D. Ahmad, M.S. Rabbani, Z.K. Shinwari and M. Iqbal. (2014). Determination of rust resistance genes in Pakistani bread wheats. Pak. J. Bot. 46: 613-617.
- Rahman, M., D. Hussain and Y. Zafar. (2002). Estimation of genetic divergence among elite cotton cultivars genotypes by DNA fingerprinting technology. Crop Sci. 42:2137-2144.
- Singh, R.P.; Singh, P.K.; Rutkoski, J.; Hodson, D.P.; Xinyao He; Jorgensen, L.N.; Hovmoller, M.S. and Huerta-Espino (2016) Disease impact on wheat yield potential and prospects of genetic control. J. Annual Review of Phytopathology 54: 303-322.
- Tadesse, W., S. Tawkaz, M.N. Inagaki, E. Picard and M. Baum. (2013). Methods and Applications of Doubled Haploid Technology in Wheat Breeding. ICARDA, Aleppo, Syria, pp 03.
- Wang, H., F. Qin, Q. Liu, L. Ruan, R. Wang, Z. Ma, X. Li, P. Cheng and H. Wang. (2015). Identification and disease index inversion of wheat stripe rust and wheat leaf rust based on hyperspectral data at canopy level. J Spect ID 651810, 10 pages.
- Yu, L.X., A. Morgounov, R. Wanyera, M. Keser, S.K.Singh and M. Sorrells. (2012). Identification of Ug99 stem rust resistance loci in winter wheat germplasm using genome-wide association analysis. Theo. Appl. Genet. 125: 749-758.
|





