EVALUATION OF ANTIMICROBIAL, ANTIOXIDANT AND ENZYME INHIBITION ROLES OF POLAR AND NON-POLAR EXTRACTS OF CLITORIA TERNATEA SEEDS
Z. Mushtaq1*, U. Khan1, N. Seher1, M. Shahid2, M. T. Shahzad1, A. A. Bhatti1 and T. Sikander1
1Bioactive Molecules Research lab (BMRL), Department of Biochemistry, 2Medicinal Biochemistry Lab, Department of Biochemistry, University of Agriculture Faisalabad, Pakistan. 38000
*Corresponding Author’s E-mail: zahidmushtaquaf@uaf.edu.pk
ABSTRACT
Clitoria ternatea is one of the medicinal plants of fabaceae family that have been traditionally useful in herbal medicine. In present study, the invitro antimicrobial, antioxidant and enzyme inhibition assay of polar and non-polar extracts/fractions of Clitoria ternatea seeds were evaluated. The antimicrobial activity was done by well diffusion assay while MIC was done by serial dilution method. Crude ethanolic extract showed maximum antibacterial activity against B. subtilus (17.6 mm ±0.17). Ethyl acetate extract showed maximum MIC at 0.156 μg/mL against E. coli. In addition, Crude methanolic extract showed maximum antifungal activity against Aspergillus parasiticus (16 mm ±0.27). The antioxidant activity was done by DPPH and FRAP assay. Crude methanolic extract showed maximum antioxidant activity by DPPH assay (86.26%) while crude methanolic extract exhibit maximum reducing power 370.19 % ± 20.50. Moreover, n-hexane extract possessed highest phenolic content (272.16 ± 17.96 GAE µg/g), crude methanolic extract possessed highest flavonoid content (275.5 ± 8.24 GAE µg/g). The highest enzyme inhibition assay was observed in crude methanolic extract (α-amylase), n-hexane extract and water-soluble residue exhibited 50% pepsin enzyme inhibition. The results concluded that most of the extracts/fractions obtained from Clitoria seeds showed significant biological activities and can act as promising source of natural therapeutic agents and can be used for future drug developmental studies.
Key words:Clitoria ternatea, α-amylase, therapeutic, antibacterial, antioxidant, fractions.
https://doi.org/10.36899/JAPS.2021.5.0342
Published online January 24, 2021
INTRODUCTION
Extensive use of antibiotics is the leading cause of increase in large number of resistant pathogens in all over the world. The resistance of antibiotics cannot be controlled because resistant pathogens grows rapidly and transfer their genetic material to other pathogens which makes them resistant to antibiotics (Mohammadi et al., 2019). They cause serious infectious diseases. The resistance towards available antibiotic drugs are becoming a major problem for researchers.
The World Health Organization (WHO) has estimated that more than 80% of the world’s population in developing countries depends primarily on herbal medicine for basic healthcare (Nair et al., 2005) as an alternative to synthetic drugs. Natural antimicrobial agents are essentially important in reducing global burden of infectious diseases. Different plants have been recognized as important sources of antimicrobial compounds that potentially act as therapeutic agent to control many infectious diseases with less side effects.
Researches working on natural products have significantly progressed over the last decades, mainly on plants corroborating their importance to the discovery of new biological and medicinal agents. The medicinal value of plants due to their phytochemicals particularly secondary metabolites and their combination with minerals and vitamins, become more beneficial for the cure of human diseases (Jayaraman et al., 2008; Mohammedi and Atik, 2011. They have different compositions with higher antimicrobial effects which is mainly related to its phenolic compounds. The higher the amount of phenolic content, the greater is its antimicrobial properties (Saeidi et al., 2019). Agents like carvacrol, eugenol and thymol are phytoconstituent which reduces the resistance of bacteria by affecting different parts of microorganism and also used as pesticides and pharmaceutical agents in traditional medicine (Walter and Marschesan, 2011).
Medicinal plants are used to treat many infectious diseases caused by bacteria and fungi. Pathogenic microbes such as Escherichia coli (E. coli), Staphylococcus aureus (S. aureus) and Bacillus subtilis (B. subtilus) are widely spread in environment responsible for high mortality and morbidity rates in human beings. S. aureus causes several puss forming infections and some serious infections such as meningitis, urinary tract infections, pneumonia, and mastitis. E. coli. and B.subtillus cause food poisoning (Sapkota et al., 2012). S. aureus, E. coli. and Pasteurella multocida are also harmful to our livestock causing hemorrhagic septicemia, diarrhea and dysentery problems in cattle etc (Hussain et al., 2011). Aspergillus species cause many diseases such as allergic and nasal sinus diseases (Albrecht et al., 2011). Fusarium species also cause a wide range of infections including allergic diseases and mycotoxicosis in animals and humans (Kaplancikli et al., 2013). Cheffouxanthone and 1,5-dihydroxyxanthoneare are antimicrobial compounds that are produced from Garcinia smeathmanii oliver, used to treat such infections (Kuete et al., 2011).
Reactive oxygen species are involved in the defensive action of cells of phagocytosis, apoptosis and detoxification in human body. But later it was found that excess production of ROS are not only harmful byproducts but are also indispensable component (Liu et al., 2013). Studies showed that over production of reactive oxygen species by certain environmental stress have been found to have adverse pathological effects in human body that induce several diseases such as cancer, diabetes, rheumatic arthritis, cardiovascular disease, Alzheimer’s disease and Parkinson disease (Sakanaka et al., 2005).High levels of ROS, inactivate antioxidant enzymes. In order to reduce the ROS, the natural and synthetic antioxidant compounds are used. Phytochemicals present in plants have wide range of bioactivities and are used in combating such challenges. Phenolic compounds are ubiquitous in plants and responsible for antioxidant activities. Plants contain soluble and insoluble phenolics in inner and outer wall. P-coumaric acid and ferulic acid are major cell wall phenolic acids (Stalika, 2007). Plants like Bersama engleriana Gurke produce bioactive compounds such as flavonoids, terpenes and saponins as natural antioxidant compounds.
Similarly, diseases like diabetes mellitus is a major endocrinal disorder affecting nearly 10% of the population all over the World. It is characterized by hyperglycemia, depleted insulin secretion and include disturbances of carbohydrate, protein and fat metabolisms. It was also predicted that by 2030, India, China and the United States will have the largest number of people with diabetes (Wild et al., 2004). It is known that this disease can be cured by inhibiting specific hydrolyzing enzymes like amylases. From this aspect, many insulin supplementry and oral hypoglycemic medications are used with more side effects. The need for alternative medicine and new approaches for managing such health challenges is becoming important. Plants are becoming popular source for natural bioactive compounds with antioxidant, antimicrobial, anticancer and enzyme inhibitiory (antidiabetic) activities etc (Ozcan, 2020).
Clitoria ternatea (CT) belonging to Fabaceae family, had been traditionally useful for different illnesses. The whole plant has been reported to have antidepressant, anxiolytic, anticonvulsant and anti-stress properties. The juice of flowers is reported to be used in insect bites and skin diseases. The root extracts are capable of curing whooping cough and goiter. Leaves are diuretic, laxative, emetic and antiperiodic. Stem is considered as a good brain tonic and is useful for throat and eye infections, skin diseases, ulcer, antidotal properties, and urinary troubles even in cattle (Mhaskar et al., 2010). Studies showed that Clitoria ternatea (CT) leaves are responsible for cytotoxic activity, antifungal, antimicrobial, antioxidant, nephroprotective (Durga et al., 2015) and anticancerous activities. Little work on seeds was found reported and we planned some activities with different extraction technique which was not practiced on it before especially from our indigenous flora. Keeping in mind the above discussion and gaps, the aim of this present study is to reveal its bioactive profile of polar and non-polar extracts or fractions of Clitoria ternatea (CT) seeds by in vitro antimicrobial, antioxidant and enzyme inhibition assays (amylase and pepsin).
MATERIALS AND METHODS
Plant material: Seeds of Clitoria ternatea (CT 25340) were collected from Botanical Garden, University of Agriculture Faisalabad during the month of February and identified through Department of Botany, University of Agriculture, Faisalabad. Seeds of Clitoria ternatea (CT) were washed with sterile distilled water, dried, ground into fine powder and preserved in a closed container at 25°C for further use.
Extract preparation and fractionation: The extractions were performed by using method of continuous stirring and Soxhlet extraction. In the first method 10 g seed powder of Clitoria ternatea was soaked in 250 mL of methanol and stirred on magnetic stirring for 16 hours and after filtration, the extraction was again repeated for next 8 hours till its repeated 3 times. The filtrate i.e. crude methanol extract (CME) thus obtained was concentrated by evaporation. After evaporation dry mass was weighed and used this extract for further fractionation in different organic and aqueous fractions (increasing order of polarity) using n-hexane, chloroform, ethyl acetate, water soluble residue and methanol soluble residue as shown in Figure No. 1 and these extracts/fractions or residues were all dried weighed and dissolved in DMSO (Li et al., 2014).
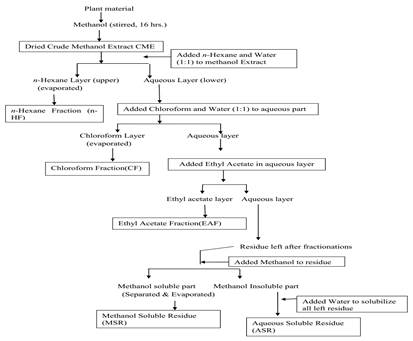
Figure 1. Schematic representation of the modified extraction method of Nisa et al., 2011 for different fractions from Clitoria ternatea seed.
The crude methanolic extract (CME) was partitioned successively by four solvent systems such as n-hexane, chloroform, ethyl acetate, aqueous fraction and final residue fractionated into methanol soluble fraction (MSR) and water-soluble fraction (WSR) in order of increasing polarity and each extraction was repeated three times. The evaporated dried powder from each extract was weighed and dissolved in DMSO to be used for biological assays (Nisa et al., 2011).
Soxhlet extraction: Ethanol extract of Clitoria ternatea seed was prepared through Soxhlet apparatus (Taur et al., 2011). 10g powdered seed material of Clitoria ternatea was placed in extraction chamber and distilled with 2/3rd ethanol filled for 16 hours. The extract was evaporated on water bath, weighed and dissolved in DMSO for further bioactivities.
Antibacterial Assay by well diffusion method: Antimicrobial activity of polar and non-polar extracts/fractions of Clitoria ternatea seeds was determined by well diffusion method (Candan et al., 2003). Nutrient agar 37 g/L was suspended in distilled water and adjusted pH 7 with 0.1N HCl/NaOH. The medium was sterilized by autoclaving at 121˚C for 15 min. Then this medium was transferred to sterilized petri plates. Then 100 µL of inoculums was added to each plate and mixed with media homogenously. It was then allowed to solidify. After this, wells of fixed diameter were cut from the solidified agar with the help of sterilized borer. 50-60 µL of each sample was poured into the wells and in positive control Ampicillin was poured. Plates were prepared under aseptic condition in laminar air flow cabinet. The Petri plates were then incubated at 37oC for 24 hours for the growth. The samples having antibacterial activity, inhibited the growth of bacterial and clear zones were formed around them. The diameters of inhibition zones were measured in millimeters with the help of vernier caliper. The results were compared with standard antimicrobial agent Ampicillin (Zaidan et al., 2005).
Bacterial strains:Escherichia coli (ATCC 35218), Staphylococcus aureus (ATCC 25923), Bacillus subtilis (ATCC 6633), Pasteurella multocida (ATCC43137) and Bacillus clausii(KP10, Isolate of BMRL lab). These strains were used to determine the antimicrobial activity of Clitoria ternatea (CT) seed extracts. We used them as were available strains in lab.
Minimum inhibitory concentration of bacteria: The Clitoria ternatea seed extracts were also evaluated through the minimum inhibition concentration (MIC) by the micro dilution method in culture broth (Sarker et al., 2007). The Clitoria ternatea seed extracts that showed inhibition zone in the well diffusion method were dissolved in 200 µL of DMSO and the solution added to 1800 µL of Muller–Hinton broth. Plates were prepared under aseptic condition in laminar air flow. A sterile 96 well plate was labeled. A volume of 100 µL of test material was pipette into the first row of the plate. To all other wells, 50 µL of broth was added. Serial dilutions were performed. Tips were discarded after each use such that each well had 50 µL of the test material in serially descending concentration. To each well 10 µL of resazurin indicator solution was added and finally, 10 µL of bacterial suspension (5x106 CFU/mL) to achieve a concentration of 5x105colony-forming unit CFU/mL. Each plate had a set of control: A column with a broad-spectrum antibiotic as positive control. A column with all solutions except test compound and a column with all solutions except bacterial solution adding 10 µL of broth instead. Each plate was wrapped and then incubated at 37 oC for 24 hours. The absorbance was measured at 500 nm. Any colour change from purple to pink or colourless was recorded as positive. The lowest concentration at which colour changes occur was taken as MIC value. The results were expressed in µg/mL (Snafi, 2016).
Antifungal assay by well diffusion method : Antifungal activity of each sample of Clitoria ternatea seed was determined by well diffusion method (Sikander et al., 2021). The Potato dextrose agar (PDA), 39 g/L was suspended in distilled water, sterilized growth medium was transferred to the sterilized Petri plates. The Petri plates were then inoculated with prepared fungal inoculum and with the help of sterilized borer, 6 mm diameter wells were made. Sample of Clitoria and standard antifungal agent Terbinafine (10 mg/mL) were poured into the wells and petri plates were incubated at 28oC for 48 hours. The extracts having antifungal activity exhibited clear zones around the sample wells. The zones of inhibition were measured in millimeters using Vernier caliper (Zaidan et al., 2005).
Determination of total phenol content (TPC): The content of total phenolics each sample of Clitoria ternatea seed was estimated by Folin Ciocalteau’s reagent (FCR) method (Vivek et al., 2013) with minor modifications. The readings of reaction mixtures were measured at 765 nm. A standard curve was plotted using different concentrations of gallic acid (standard, 50-250 μg/mL) and the TPC of extracts was expressed as μg gallic acid equivalents (GAE) from the graph.
Total phenolic content = mg of Gallic acid Equivalents per gram dried extract.
Total phenols were calculated by the following equation;
Concentration (μg) = Absorbance in nm – (value of y-intercept) Value of slope of standard curve
Determination of total flavonoid content (TFC): Aluminum chloride based colorimetric method was used for the flavonoid’s determination (Chang et al., 2002). 0.5 mL of each sample of Clitoria ternatea seed were separately mixed with 1.5 mL of methanol, 0.1 mL of 10% aluminum chloride, 0.1 mL of 1 M potassium acetate and 2.8 mL of distilled water. The reaction mixture was allowed to stand at room temperature for 30 min and the absorbance of the reaction mixture was measured at 415 nm. The calibration curve was prepared by using quercetin at concentrations of 50 to 250 μg/mL.
Total flavonoid content = mg of Quercetin Equivalents per gram dried extract.
Total flavonoids were calculated by the following equation;
Concentration (μg) = Absorbance in nm – (value of y-intercept) Value of slope of standard curve
In vitro Antioxidant Activity:
DPPH scavenging assay: The antioxidant activity of samples of Clitoria ternatea seeds were assessed by measuring their scavenging ability to 1, 1-diphenyl-2-picrylhydrazyl stable free radicals (DPPH). Previously reported DPPH assay was performed with slight modifications (Roopa et al., 2015). 5 μL of sample solution were added in 95 μL of methanolic solution of DPPH. After 30 minutes incubation in darkness at room temperature the absorbance was recorded at 517 nm. The experiment was performed for three times. Ascorbic acid was used as standard control. Inhibition of free radicals by DPPH was calculated in the following way:
I (%) = (A blank __ A sample / A blank) x 100
Where Ablank is the absorbance of the control reaction mixture excluding the test compounds, and Asample is the absorbance of the test compounds. Percentage scavenging was calculated as depicted in formula where I (%) is the percentage inhibition of DPPH free radical.
Ferric reducing antioxidant power (FREP) assay: The determination of total antioxidant activity (FREP assay) in the extract is a modified method of Benzie and strain (1996). The stock solutions included 300 mM acetate buffer (3.1 g C2H3NaO2.3H2O and 16 mL C2H4O2), pH 3.6, 10 mM 2, 4, 6-tripyridyl-s-triazine (TPTZ) solution in 40 mM HCL, and 20 mM FeCl3.6H2O solution. The fresh working solution was prepared by mixing 25 mL acetate buffer, 2.5 mL TPTZ, and 2.5 mL FeCl3.6H2O. The temperature of the solution was raised to 37 oC before use. Different extracts/fractions of Clitoria ternatea seed (200 µL) was allowed to react with 2800 µL of the FREP solution for 30 min in the dark condition. Reading of the colored product (ferrous tripyridyltriazine complex) were taken at 593 nm. The standard curve was linear between 200 and 1000 µM Fe (11)/g dry mass and compared with ascorbic acid (Henderson et al., 2015).
Enzyme inhibition assay
Alpha-Amylase Inhibitory Assay: This assay was carried out using a modified procedure of McCue and Shetty (2003). A total of extract (1.25–10 mg/mL) was placed in a tube and 250 𝜇L of 0.02M sodium phosphate buffer (pH 6.9) containing 𝛼-amylase solution (0.5 mg/mL) was added. This solution was preincubated at 25 oC for 10 min, after which 250 𝜇L of 1% starch solution in 0.02M sodium phosphate buffer (pH 6.9) was added at time intervals and then further incubated at 25 oC for 10 min. The reaction was terminated by adding 500 𝜇L of dinitro salicylic acid (DNS) reagent. The tubes were then incubated in boiling water for 5min and cooled at room temperature. The reaction mixture was diluted with 5 mL distilled water and the absorbance was measured at 540 nm using spectrophotometer. A control was prepared using the same procedure replacing the extract with distilled water. The 𝛼-amylase inhibitory activity was calculated as percentage inhibition;
%Inhibition = [Abs control – Abs extracts/Abs control] × 100.
Pepsin inhibition assay: Pepsin inhibition assay was performed by following the appropriate method described by Rege and Chowdhary (2014). Agar was prepared along with 1% (w/v) casein, skimmed milk, nutrient agar was poured in petri dishes. The plates were solidified for 30 min and holes (6 mm diameter) were created. The samples were poured into holes and the concentration of each extract was 200 µg/mL. These plates were incubated for overnight at 37°C and pure pepsin solution was used as positive control. Zone of inhibition was measured with the help of Vernier caliper.
Statistical Analysis: The data obtained was analyzed statistically by using analysis of variance technique. The results obtained were analyzed through one-way analysis of variance (ANOVA) (Montgomery et al., 2008) by using SPSS software version 22.
RESULTS
The yield of each extract after extractions, fractionation and residues solubility by method as described (Nisa et al., 2011) earlier in figure 1 is shown in table 5.
Antimicrobial activity of Clitoria ternatea seed extracts
Antibacterial activity: Antibacterial potential of Clitoria ternatea seed extracts were investigated by agar well diffusion method (Candan et al., 2003). In the present study, the antibacterial potential of Clitoria ternatea extracts/fractions/soluble residues were performed and results are presented in table 1. Antibacterial activity of Clitoria ternatea extracts were analyzed against four bacterial strains of which some were gram positive such as Staphylococcus aureus,Bacillus clausii and Bacillus subtilis and two were Gram negative such as Pasteurella multocida and Escherichia coli. Ampicillin (10 mg/mL) was used as positive control.
In this study, crude methanolic extract (CME) of Clitoria ternatea showed maximum ZOI against Bacillus clausii (13 mm ± 0.32). Crude ethanolic extract (CEE) showed maximum ZOI against Bacillus subtilis (17.6 mm ± 0.17) and Staphylococcus aureus (14 mm ± 0.45). n-Hexane fraction (n-HF) showed maximum ZOI against Pasturella multocida (13 mm ± 0.26) and E. coil (14 mm ± 0.45). Chloroform fraction (CF) also showed significant activity against Bacillus subtilis (11 mm ± 2.0). Methanol soluble residue (MSR) showed significant antibacterial activity against Staphylococcus aureus (10 mm ± 0.28) and Escherichia coli (11 mm ± 0.32). Aqueous soluble fraction and residues (AQF, ASR) showed significant antibacterial activity against Pasturella multocida (10 mm± 0.25, 9 mm ± 0.3) The study indicated that crude ethanolic extract (CEE) and crude methanolic extract (CME) showed significant antibacterial activity among all extracts.
Table 1. Antibacterial potential of Clitoria ternatea seed extracts
| |
Sample Name
|
Bacillus subtilis
|
Staphylococcus aureus
|
Pasteurella multocida
|
Escherichia coli
|
Bacillus clausii
|
|
Mean ZOI (mm)±S.E
|
Mean ZOI (mm)±S.E
|
Mean ZOI (mm)±S.E
|
Mean ZOI (mm)±S.E
|
Mean ZOI (mm)±S.E
|
|
1
|
Crud Methanol Extract (CME)
|
10.0C ±1.00
|
9A ± 0.26
|
12E ±0.20
|
10D±0.39
|
13.6A ±0.32
|
|
2
|
n-Hexane Fraction (n-HF)
|
12.6A ±1.52
|
8F ± 0.33
|
13D ± 0.26
|
14B± 0.45
|
11F ± 0.53
|
|
3
|
Chloroform Fraction (CF)
|
11.0D ±2.0
|
7H ± 0.25
|
10B ± 0.32
|
12A ± 0.41
|
10.0C ±1.00
|
|
4
|
Ethyl Acetate Fraction (EAF)
|
10.3E±1.1
|
8G ± 0.45
|
8F ± 0.45
|
9C ± 0.15
|
9C± 0.3
|
|
5
|
Aqueous Fraction (AQF)
|
12.3B ±1.2
|
8B ± 0.25
|
10A± 0.25
|
9G ± 0.24
|
7H ± 0.25
|
|
6
|
Water Soluble Fraction (WSR)
|
6F ± 0.251
|
9C ± 0.3
|
9C± 0.3
|
13E± 0.26
|
11I ± 1.5
|
|
7
|
Methanol Soluble Fraction (MSR)
|
9H ±0.90
|
10D ± 0.20
|
9H ± 0.264
|
11I ±0.32
|
12I ± 0.351
|
|
8
|
Crude Ethanol Extract (CEE)
|
17.6G ±0.17
|
14I ± 0.25
|
12I ± 0.351
|
12H ± 1.4
|
8G ± 0.45
|
|
9
|
Ciprofloxacin (+ive control)
|
18.12I± 0.31
|
13E± 0.30
|
14G ± 0.173
|
16F ± 2.1
|
17G ±0.72
|
mm = millimeter. ZOI = Zone of inhibition in millimeter, ; *Means sharing same alphabets are statistically non-significant.
Minimum inhibitory concentration (MIC)
Minimum inhibitory concentration of Clitoria ternatea seeds extracts against gram positive and gram negative bacteria was determined by serial dilution method. All extracts showed antibacterial activity at lowest concentration (0.078 µg/mL to 2.5 µg/mL). EAF showed lowest MIC value against three gram positive and one gram negative bacteria as compared to others. But they have higher MIC values than ciprofloxacin which is used as positive control. These results indicate that CME and EAF showed lowest MIC value (0.156 µg/mL) against S. aureus. EAF showed lowest MIC value (0.312 µg/mL and 0.78 µg/mL) against B. subtilis and P. multocida. AQF, MSF and CEE showed lowest MIC value (0.625 µg/mL) against S. aureus. The MIC values of all extracts are shown in Table 2 and figure 2.
Table 2. Minimum inhibitory concentration of C. ternatea seed extracts with respect to ciprofloxacin
| |
Sample Name
|
Bacillus subtilis
|
Staphylococcus aureus
|
Pasteurella multocida
|
E. coli
|
|
MIC (μg/μL)
|
MIC (μg/μL)
|
MIC (μg/μL)
|
MIC (μg/μL)
|
|
1
|
Crude Methanol Extract (CME)
|
1.25
|
0.156
|
0.156
|
0.312
|
|
2
|
n-Hexane Fraction (n-HF)
|
1.25
|
2.5
|
0.625
|
0.156
|
|
3
|
Chloroform Fraction (CF)
|
1.25
|
1.25
|
1.25
|
2.5
|
|
4
|
Ethyl Acetate Fraction (EAF)
|
0.625
|
1.25
|
0.078
|
0.321
|
|
5
|
Aqueous Fraction (AQF)
|
2.5
|
0.625
|
0.156
|
1.25
|
|
6
|
Water Soluble residue (WSR)
|
2.5
|
1.25
|
1.25
|
1.25
|
|
7
|
Methanol Soluble residue (MSR)
|
1.25
|
0.625
|
0.312
|
1.25
|
|
8
|
Crude Ethanol Extract (CEE)
|
1.25
|
0.625
|
1.25
|
0.312
|
|
9
|
Ciprofloxacin (+ive control)
|
0.019
|
0.078
|
0.321
|
0.009
|

(G) E.coli (CME-AQF) (H)E.coli (WSF-EAF)
Figure 2. Minimum inhibitory concentration of Clitoria ternatea seed extracts/fractions/residues: Each plate from A-H represents MIC values of bioactive samples against each tested strain of bacteria alongwith +ve and –ve controls described in protocols.
Antifungal assay: Antifungal activity of the Clitoria ternatea seed samples were determined through well diffusion method of Sikander et al., (2021). All extracts of Clitoria ternatea seed were subjected to antifungal activity against selected strains Fusarium solani, Aspergillus niger, Aspergillus parasiticus and Aspergilus ustus. Results of antifungal activity are shown in Figure 3 and documented in table 4. Terbinafine and Fluconazole were used as standard drug.
In our study, the Ethyl acetate fraction (EAF) showed maximum antifungal activity against Fusarium specie (13 mm ±0.16). Aqueous fraction (AQF), Chloroform extract (CF) and crude ethanol extract (CEE) showed maximum antifungal activity against Aspergillus niger (13 mm±0.16). Methanol soluble residue (MSR) showed maximum antifungal activity against Aspergillus parasticus (12 mm±0.17). Crude methanol extract (CME) showed maximum antifungal activity against Aspergillus ustus (16 mm ±0.27). Ethyl acetate fraction (EAF) showed significant activity against Fusarium solani and Aspergillus ustus (13 mm±0.16). Our study shows that all extracts possess antifungal activity but less than standard drug. CME showed maximum results among all extracts against Aspergillus ustus nearer to standard drug Fluconazole.
Table 3. Antifungal activity of Clitoria ternatea seed extracts against different fungal strains.
|
Sr
|
Extract/fractions
|
Fusarium solani
Mean ZOI (mm)±S.E
|
Aspergillus niger
Mean ZOI (mm)±S.E
|
Aspergillus parasiticus
Mean ZOI (mm)±S.E
|
Aspergilus ustus
Mean ZOI (mm)±S.E
|
|
1
|
CME
|
11C±0.17
|
8D±0.71
|
9D±1
|
16C±0.27
|
|
2
|
EAF
|
13B±0.16
|
7.2E±0.22
|
9.7E±0.22
|
13B±0.16
|
|
3
|
n-HF
|
12E±0.22
|
9D±0.19
|
7D±0.19
|
7.2E±0.25
|
|
4
|
CF
|
10CD±0.18
|
13B±0.16
|
10B±0.16
|
13F±0.16
|
|
5
|
AQF
|
5F±0.26
|
13B±0.16
|
11B±0.16
|
14E±0.45
|
|
6
|
WSR
|
9D±0.19
|
10CD±0.18
|
9CD±0.18
|
7B±0.23
|
|
7
|
MSR
|
9D±0.19
|
11C±0.17
|
12C±0.17
|
11G±0.36
|
|
8
|
CEE
|
10CD±0.18
|
13B±0.16
|
6B±0.16
|
7D±0.46
|
|
9
|
Fluconazole +ve
|
31A±0.10
|
26D ±0.96
|
18C±0.67
|
17H±0.9
|
|
10
|
Terbinafine +ve
|
31.7A±0.21
|
22F±0.54
|
16E±0.16
|
23C±0.18
|
mm = millimeter. ZOI = Zone of inhibition in millimeter,
*Means sharing same alphabets are statistically non-significant.
Total phenolic and flavonoids contents: Total phenolic and flavonoid contents were measured in the Clitoria ternatea seed extracts. Total phenolic contents have been expressed in terms of Gallic acid Equivalents µg/g plant. The total phenolic content ranged from 86.35 ± 7.45 to 272.16 ± 17.96 (GAE µg/g plant) (Table 4). Highest total phenolic content (272.16 ± 17.96 GAE µg/g plant) were recorded in n-hexane fraction (n-HF) and the least quantity (86.35 ± 7.45 GAE µg/g plant) was present by water soluble residue (WSR) of C. ternatea seed extracts. Moreover, crude methanol extract (CME) and crude ethanol extract (CEE) also presented higher total phenolic contents with 247.54 ± 5.16 and 225.81 ± 4.01 GAE µg/g respectively.
Table 4. Total phenolic and flavonoid contents in Clitoria ternatea seed extracts
|
Extract/fractions
|
Total Phenolic Contents
(GAE µg/g plant)
|
Total Flavonoid Contents
(µg/g)
|
|
Crude methanol extract (CME)
|
247.54 ± 5.16
|
224.79 ± 3.7
|
|
n-hexane fraction (n-HF)
|
272.16 ± 17.96
|
252.68 ± 2.23
|
|
Chloroform fraction (CF)
|
192.03 ± 4.20
|
187.17 ± 1.56
|
|
Aqueous fraction (AQF)
|
170.41 ± 8.60
|
208.27 ± 3.82
|
|
Methanol soluble residue (MSR)
|
170.41 ± 8.03
|
213.13 ± 8.21
|
|
Water soluble residue (WSR)
|
86.35 ± 7.45
|
125.13 ± 3.35
|
|
Crude ethanol extract (CEE)
|
225.81 ± 4.01
|
275.58 ± 8.24
|
|
Ethyl acetate fraction (EAF)
|
86.35 ± 1.30
|
257.5 ± 8.24
|
The total flavonoid contents of the plant extracts in terms of Catechin Equivalents varied from 125.16 ± 3.35 to 275.58 ± 8.24 CE µg/g plant (Table 4). Highest total flavonoid contents (275.5 ± 8.24 CE µg/g plant) were obtained in crude ethanol extract (CEE). While, lowest TFC were noted in water soluble residue (WSR) 125.16 ± 3.35 CE µg/g plant. n-hexane fraction (nHF) also presented a considerable TFC value of 252.68 ± 2.23 CE µg/g plant. Total flavonoid contents observed in crude methanol extract (CME) and ethyl acetate fraction (EAF) were 224.79 ± 3.7 and 257.5 ± 8.24 CE µg/g plant, respectively.
The phenolic contents of plant is totally dependent on the type of solvent. Solvent effects on intermolecular interaction, which interchange their functional group. Non polar solvents have better effects than polar ones, because lower charges penetrate into the less polar phenolic compounds i.e tocopherol. While polar ones are known to extract products such as proteins and carbohydrates also. They have lower electrostatic interaction, interact with compounds and interchange their functional groups which increases their efficiency (Venkatesan et al., 2019) and result in utilization of higher yield. The highest yield of Lebanese urtica dicota was observed by non-polar solvent.
DPPH free radical scavenging activity: The DPPH free radical scavenging activity of organic extracts of Clitoria ternatea seed were evaluated and results are shown in Table 5. The methanolic extracts of Clitoria ternatea showed highest antioxidant activity while ethyl acetate and chloroform showed significant results with minimum concentration (20 µg/mL). The methanolic extract of clitoria ternatea flowers showed significant antioxidant activity as reported by Patil and Patil, 2011.The methanol extract of Camellia sasanqus showed maximum antioxidant activity at concentration 18.3 µg/mL.
Table 5. Clitoria ternatea extracts possess free radical scavenging activity by DPPH
|
Sr#
|
Sample Name
|
Concentration(mg/mL)
|
%age antioxidant activity
|
|
1
|
Crude Methanol Extract (CME)
|
137
|
86.26
|
|
2
|
n- Hexane Fraction (nHF)
|
250
|
48.30
|
|
3
|
Chloroform Fraction (CF)
|
300
|
83.51
|
|
4
|
Ethyl Acetate Fraction (EAF)
|
700
|
85.35
|
|
5
|
Aqueous Fraction (AQF)
|
300
|
64.31
|
|
6
|
Water Soluble residue (WSR)
|
240
|
22.70
|
|
7
|
Methanol Soluble residue (MSR)
|
280
|
26.45
|
|
8
|
Crude Ethanol Extract (CEE)
|
600
|
43.48
|
|
9
|
Ascorbic acid
|
--
|
89.65
|
|
10
|
DPPH
|
--
|
0.712
|
Ferric Reducing Antioxidant Power (FRAP) assay: The Ferric Reducing Antioxidant Power (FRAP) assay is an extensively used assay for the assessment of the antioxidant compounds of the dietary polyphenols (Henderson et al., 2015). This method relies on the reduction of the Fe+3-TPTZ (2, 4, 6-tri (2-pyridyl) -1, 3, 5-triazine) complex by the antioxidants. The antioxidant potential of medicinal plants can be estimated from their ability to reduce TPTZ-Fe (III) complex to TPTZ-Fe (II) and the number of antioxidants can be estimated directly from the amount of reduced ion (Babu and Rao, 2013). All samples showed prominent antioxidant activity. CME showed maximum reducing power at 500 µg/mL while WSR showed minimum reducing power. The reducing power of all samples are shown in table 6
Table 6. FRAP values (Fe (II) mg/mL) of different extracts/fractions of Clitoria ternatea
|
Extracts/fractions
|
FRAP values (Fe (II) mg/g plant extract)
500µg/mL
|
|
CME
|
365.16 ± 19.95
|
|
EAF
|
346.71 ± 25.18
|
|
n-HF
|
365.83 ± 9.24
|
|
CF
|
329.26 ± 4.63
|
|
AQF
|
225.93 ± 20.50
|
|
WSR
|
156.60 ± 20.95
|
|
MSR
|
353.86 ± 7.89
|
|
CEE
|
370.19 ± 20.50
|
The polar and non-polar extracts of Clitoria ternatea seed showed considerable antioxidant effect ranged from 370.19 to 156.60 Fe (II) mg/g plant at 500 µg/mL All extracts showed prominent antioxidant activities presented by CME (365.16 ± 19.95), EAF (346.71 ± 25.18), n-HF (365.83 ± 9.24) and CEE (370.19 ± 20.50). The WSR exhibited lowest FRAP values with 156.60 ± 20.95 Fe (II) mg/g.
α-Amylase inhibition assay: Alpha-amylase inhibition of different samples of Clitoria ternatea seed were investigated by in vitro inhibitory assay. The results of different polar and nonpolar extracts of C. ternatea seeds are shown in Figure 3. These results demonstrated that all extracts showed alpha amylase inhibition ranged between 47% to 12% at highest concentration (100 μg/mL). WSR showed lowest amylase inhibition (12 ± 1.2 %) and (CEE) showed maximum amylase inhibition (47 ± 1.20 %) among all samples.
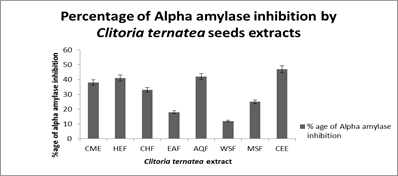
Fig. 3. Alpha amylase inhibition assay by different extracts of Clitoria ternatea seed
Pepsin inhibition assay: Pepsin enzyme inhibition activity of different extracts of Clitoria ternatea seed were investigated by casein agar well diffusion method. The results of pepsin inhibition of C. ternatea seed extracts are shown in table 7 and figure 4. These results demonstrated that all extracts showed little pepsin inhibition ranged between 9 % to 6 %. Water soluble residue (WSR) showed minimum zone of inhibition (6 ± 0.49 %) and crude methanol extract (CEE) and ethyl acetate fraction showed maximum inhibition (9 ± 1.2 %) among all extracts. Pepsin enzyme without sample was used as standard.
Table 7. Inhibition of pepsin enzyme by different extracts of Clitoria ternatea seeds
|
Extracts/fraction
|
ZOI (mm)
|
%age inhibition
|
|
CME
|
9 ± 1.7
|
25
|
|
nHF
|
6 ± 0.56
|
50
|
|
CF
|
8 ± 0.98
|
33.34
|
|
EAF
|
9 ± 1.2
|
25
|
|
AQF
|
8 ± 0.83
|
33.34
|
|
WSR
|
6 ± 0.49
|
50
|
|
MSR
|
7 ± 0.34
|
41.66
|
|
CEE
|
7 ± 0.35
|
41.66
|
|
Pepsin (+ive control)
|
12 ± 0.44
|
0.00
|

Figure 4. Inhibition of pepsin enzyme by C. ternatea seed extracts using casein agar well diffusion method. Crude methanol extract (CME) and ethyl acetate fraction (EAF) shows highest 9 mm inhibitory potential against pepsin where pepsin enzyme was used as a standard.
DISCUSSION
Studies on medicinal plants exhibit phytochemical constituents having antimicrobial, antioxidant and enzyme inhibitory activities have been on rise due to their therapeutic potential in various infectious and chronic disorders. Experimental studies have shown that prolonged oxidative stress caused by excess production of free radical molecules lead to certain metabolic disorders such as cardiovascular disease, cancer and diabetes (Zhang et al., 2014). Synthetic antibiotics are used to cure these diseases but they have high risk of adverse side effects. Medicinal plants have become a popular source of bioactive agents against pathogenic microbes. Genetic variability of microbes make them resistant pathogens and loose their antibiotic susceptibility (Govindappa et al., 2011).
Results of our finding confirmed that polar and non-polar extracts of Clitoria ternatea seeds possess antimicrobial, antioxidant and enzyme inhibitory activities. The crude methanolic and ethanolic extract is the most bioactive among all extracts having significant TPC and TFC values. The ethanolic and n-hexane extract of clitoria ternatea seed showed maximum phenolic content. WSF have least phenolic and flavonoid contents and became less bioactive among all extracts. Studies showed that the methanolic extract of Capnophyllum peregrinum have maximum phenolic and flavonoid content (Lefahi et al., 2018). The ethanolic extract of Limnophila aromatica showed maximum flavonoid and phenolic content as compared to other extracts. Water soluble extract/residue of Limnophila aromatica have least phenolic and flavonoid content which is significantly less bioactive than other solvents (Quy et al., 2014).
Phenolics and Flavonoids have been renowned as therapeutically active substances which regulate important physiological actions in human body. Research on Flavonoids from plant extracts has been tremendously increased because of their antimicrobial and antioxidant properties (Govindappa, 2011) with versatile health benefits, which have been reported in numerous studies (Kumar and Pandey, 2013). Antimicrobial activity of plant extracts is attributed due to the presence of numerous bioactive secondary metabolites known as polyphenols. Phenolic compounds, terpenoids and alkaloids are very important components for antimicrobial activity. For this reason, total phenolic content as well as flavonoid content are usually quantified alongwith bioassays. Our findings confirmed the relationship between the amount of phenolics, flavonoid and biological activities (Stankovik,2011).
In antimicrobial studies, the antibacterial activity was evaluated and found that crude ethanolic extract showed maximum inhibition against one gram positive bacteria B. subtilus and n-HF showed maximum inhibition against gram negative bacteria E. coli. Rahman et al. (2017) reported that ethanolic Clitoria leaf extract showed significant antibacterial activity against E. coli (9.8 mm). Zoysa et al. (2019) reported that Ethanol and n-hexane extracts of Epaltes divaricata gave maximum activity against S. aureus (16.3 mm and 13.7 mm). Ethanolic and n-hexane extracts of Vetiveria zizanioides also gave maximum activity against S. aureus (12.1 mm and 11.4 mm). Saeidi et al. (2019) reported that the mint extract showed antibacterial activity against gram negative bacteria E. coli (5.1 mm) and gram-positive bacteria S. aureus (3.1 mm) respectively.
The MIC was also determined against two gram positive and two gram negative bacteria to quantify least amounts required for the activity of polar and non-polar extracts. CME, nHF and EAF showed maximum growth inhibition at minimal concentration (0.156-0.312 µg/mL) against gram positive bacteria B. subtilis and P.multocida. While CME showed maximum growth inhibition against (0.156 µg/mL) one gram negative bacteria S. aureus. Hamayeili et al. (2019) reported that polar and non-polar extracts of sea anemone have MIC ranged between 0.62-100 µg/mL against gram positive and gram negative bacteria. Sen and Batra, (2012) reported that ethanolic and methanolic extract of Melia azedarach L. showed inhibition at 42.5 µg/mL and 39.6 µg/mL against E. coli, and 38.7 µg/mL and 42.4 µg/mL against S. aureus. Zoysa et al. (2019) reported that ethanol and hexane extracts of V. zizanioides gave MIC values of 2.4 mg/ml and 0.003 mg/mL against S. aureus. Comparison with reported literature showed that we have promising results.
For antifungal activity, CME and CEE extract showed maximum antifungal activity against Aspergillus niger and Aspergillus ustus. Snafi in 2019 discussed that crude methanolic extract of Clitoria ternatea possess antifungal activity against Aspergillus niger. Snafi, (2016) reported that methanolic extract of Clitoria ternatea leaf possess antifungal activity against Fusarium solani. By comparing our results, the concentration of phenolic and flavonoids content, and antimicrobial activity of plant extracts are not strongly correlated. This suggests that Clitoria ternate seed extracts are rich in bioactive compounds, not only solely phenols and flavonoids, but a variety of different bioactive compounds are also present in it that could be reason of bioactivities.
For antioxidant activity, we observed rapid and strong inhibition of free radicals by DPPH and FRAP assay. For DPPH assay, we found that CME showed maximum antioxidant activity. Water soluble extract/residue showed least antioxidant activity. Phytochemical constituents are responsible for scavenging free radicals and have positive correlation with antioxidant activity (Sahu et al., 2013). The methanolic extract of medicinal plant possess maximum phenolic contents and water soluble have least phenolic content. The increase in antioxidant potential may be due to increase in phenolic components such as flavonoids, phenolic acids and diterpenes (Mushtaq et al., 2017).From previous studies we observed that crude methanolic extract of C. intybus seeds possess maximum antioxidant activity (Mehmooda et al., 2012). The methanolic extract of T. arjuna possess high antioxidant potential due to presence of higher flavonoid content (Mandal et al., 2015).
For FRAP assay, CEE showed maximum free radical inhibition and possess higher flavonoid contents. n-HF and CME showed significant ferric reducing activity and water-soluble extract/residue have least ferric reducing activity. From previous studies found that crude ethanolic extract of Tehran propolis showed maximum ferric reducing ability (Mohammadzedah et al., 2007). The hexane extract of Garcinia madruno seed gave maximum ferric reducing activity (Ramirez et al., 2019). By comparing our results with previous reported data, we found that total phenolic and flavonoid content may be responsible for antioxidant activity but variety of different bioactive compounds are also needed. The phenolic compounds have redox properties, which allow them to act as reducing agents, hydrogen donors, and singlet oxygen quenchers. The redox potential of phenolic compounds played an important role in determining the antioxidant potential, and they can serve as promising preventive medication for various diseases (Pantavos et al., 2015).
Different mechanisms such as scavenging of free radicals, chelation of metal ions and inhibition of enzymes are involved in antioxidative properties (Mandal et al., 2015). The antioxidant level could be maintained by phytoconstituents present in medicinal plants, which claimed to act as powerful chain breaking antioxidants (Padmanabhan and Jangle, 2012). The Clitoria ternatea seed extracts could be beneficial to mankind by virtue of their effective antioxidant activity which may able to impart cardio-protective potential against various cardiovascular diseases (Snafi, 2016). Researchers reported that leaf extracts of Clitoriaternatea was traditionally useful in diabetes, Alzheimer disease and gastrointestinal disease treatment. The preliminary phytochemical screening showed that the plant contained tannins, phlobatannin, carbohydrates, saponins, triterpenoids, phenols, flavanoids, flavonol glycosides, proteins, alkaloids, anthraquinone, anthocyanins, cardiac glycosides, Stigmast-4-ene-3,6-dione, volatile oils and steroids. Phenolic compounds present in it, could be responsible for the interaction with key modulating enzymes in the digestive tract to initiate their activity. We observed enzyme inhibitory activities of polar and non-polar extract of C. ternatea seed extracts against alpha amylase and pepsin enzymes.
Alpha-amylase is a prominent enzyme found in the pancreatic juice and saliva which breaks down large insoluble starch molecules into soluble molecules. The mammalian glucosidase catalyzes the end product of starch into absorbable molecule (Manohar et al., 2002). Several inhibitors such as amylase inhibitor are known as starch blockers because they inhibit the breakdown, and slowdown the digestion of dietary starch which is being absorbed by the body and lowers the blood glucose level, and have beneficial effects on insulin resistance and glycemic index control in people with diabetes. Progressive metabolic disorder in 𝛽-cells of the pancreas or insulin resistance result in Type I and Type II diabetes (Heise et al., 2004). In our investigation, we found that crude ethanolic extract showed maximum amylase inhibition with maximum flavonoid content. Wang et al., 2010 reported that ethanolic extract of guava leaves showed alpha amylase inhibition with high flavonoid content. Belfeki et al., 2016 reported that the ethanolic extract of Coriandrum sativum showed maximum inhibition against alpha amylase 68.98 ± 0.4 %. This inhibitory activity could be correlated with the greatest amount of flavonoid content. From phytochemical screening, polyphenols have been reported to inhibit the activity of carbohydrate hydrolyzing enzymes like α-amylase. It was also reported that α-amylase inhibition is related to the presence of certain compounds such as tannins, phenols and flavonoids (Moein et al., 2017). From our previous reported data our finding suggested that flavonoid content could be responsible for alpha amylase inhibitory activity.
Similarly, pepsin is one the key enzyme which breaks down the small protein molecule into absorbable molecule and only produced in stomach. The inequality between acid and pepsin secretion, and defensive factors of gastric mucosa cause gastrointestinal disorder known as gastric ulcer. A number of substances have been known as pepsin inhibitors. The role of pepsin in ulcerogenesis are not well defined, and no single anti-ulcer drug could give complete healing without recrudescence. In our investigation, we found that crude methanolic and ethyl acetate extract showed maximum pepsin inhibition. Sofidiyaa et al., 2014 reported that ethyl acetate extract of Markhamia tomentosa possess pepsin inhibitory activity. Balan and his coworkers (2015) also reported that methanolic extract of Muntingia calabura leaves possess pepsin inhibition and useful against ulcer. Previous reports have dealt with the gastric protection or peptic ulcer treatment using plant extracts against gastric ulcer. Shahrokhi et al. (2015) reported that polyphenols inhibit pepsin secretion and increase mucin secretion in gastric ulcer. The presence of phytochemical constituents such as phenol and flavonoid may be responsible for pepsin inhibitory activity but merely some other bioactive compounds are also present in it.
In this research, plant part such as seed extracts of Clitoria ternatea possess phytochemicals such as phenols and flavonoids and its supposed to be responsible for bioactivities. This is the first report on antimicrobial, antioxidant and enzyme inhibition assay of polar and non-polar seed extracts of Clitoria ternatea with different described extraction/fractionations. Previous research data has not described well about the in vitro bioactive studies of seed extracts of this plant. The bioactivity of polar and non-polar seed extracts of Clitoria ternatea, their phenolics and flavonoid contents, and their bioactive role in the inhibition of α-amylase and pepsin enzymes has not been reported yet and this may be considered as novelty of current research.
On the basis of our studies it is suggested that Clitoria ternatea seed extracts like chloroform, ethyl acetate, methanol soluble residues and crude methanol extract, n- hexane, can be used against infectious diseases caused by different bacterial and fungal strains. Thus, can be used as source of therapeutic agents. Still there is a need of extensive studies in the field of medicinal plants and drug development through different plant extracts. In vivo studies may be helpful in determining the real potential usefulness of these Clitoria ternatea seed extracts for the treatment of infectious and metabolic disorders. Isolation of purified individual bioactive molecules is also required to investigate for drug developmental studies.
REFERENCES
- Albrecht, S. C., A. G. Barata, J. Grosshand, A. A. Teleman and T. B Dick (2011). In vivo mapping of hydrogen peroxide and oxidized glutathione reveals chemical and regional specificity of redox homeostasis. Cell. Metab. 14(6): 819-829.
- Babu, D. R., and G. N. Rao (2013). Antioxidant properties and electrochemical behavior of cultivated commercial Indian edible mushrooms. J. Food Sci. Technol. 50(2): 301-308.
- Balan, T., M. H. M. Sani, S. M. Ahmad, V. Suppaiah, N. Mohtarrudin and Z. A Zakaria (2015). Antioxidant and anti-inflammatory activities contribute to the prophylactic effect of semi-purified fractions obtained from the crude methanol extract of Muntingia calabura leaves against gastric ulceration in rats. J Ethnopharmacol. 164 (1):1–15.
- Belfeki, H., M. Mejri and M. Hassouna (2016). Antioxidant and α-amylase inhibitory activities of some tunisian aromatic plants. J. New. Sci. Agri. Biotech. 31(6): 1775-1782.
- Benzie, I. F., and J. J. Strain (1996). The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal. Biochem. 239(1): 70-76.
- Candan, F., M. Unlu, B. Tepe, D. Daferera, M. Polissiou, A. Sökmen and H. A. Akpulat (2003). Antioxidant and antimicrobial activity of the essential oil and methanol extracts of Achillea millefolium subsp. millefolium Afan. (Asteraceae). Journal of ethnopharmacology. 87(2): 215-220.
- Chang, C. C., M. H. Yang, H. M. Wen and J. C. Chern (2002). Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food. Drug. Anal. 10(3): 178-182.
- Durga, Ch. M. L., B. Mahitha, Madhav, T and Sushma, N. J (2015). Phytochemical screening and Ftir analysis of Clitoria ternatae leaves. Int. J. Sci. Eng. Res. 6(2): 287-290.
- Govindappa, M., N. Bharath, H. B. Shruthi, T. S. Sadananda and P. Sharanappa (2011). Antimicrobial, antioxidant and in vitro anti-inflammatory activity and phytochemical screening of Crotalaria pallida Aiton. Afr. J. Phar. Pharmacol. 5 (21): 2359-2371.
- Heise, T., L. Nosek, B. B. Rønn, L. Endahl, L. Heinemann, C. Kapitza and E. Draeger (2004). Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes. 53(6):1614-20.
- Hussain, I., M. R. Khattak, R. Ullah, Z. Muhammad, N. Khan, F. A. Khan, Z. Ullah and S. Haider (2011). Phytochemicals screening and antimicrobial activities of selected medicinal plants of Khyber Pakhtunkhwa, Pakistan. Afr. J. Pharm. 5 (6): 746-750.
- Henderson, G., F. Cox, S. Ganesh, A. Jonker, W. Young, G. R. C. Collarborators and P. H. Janssen (2015). Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci. Reports. 5(1):14567-14568.
- Hamayeili, H., M. Hassanshahian and M. A. Hesn (2019). The antibacterial and antibiofilm activity of sea anemone (Stichodactyla haddoni) against antibiotic-resistant bacteria and characterization of bioactive metabolites. Int Aquat Res. 11(1): 85–97.
- Jayaraman, S., M. S. Manoharan and S. Illanchezian (2008). In-vitro antimicrobial and antitumor activities of Stevia Rebaudiana (Asteraceae) leaf extracts. Trop. J. Pharm. Res. 7 (4): 1143-1149.
- Kaplancikli, Z. A., M. D. Altıntop., G. T. Zitouni, A. Ozdemir, R. Demirel., U. A. Mohsen and W. Hussein (2013). Synthesis and antifungal activity of new heterocyclic compounds. Cukurova Med. J. 38(1): 103-107.
- Kuete, V., B. Krusche, M. Youns, I. Voukeng, A. G. Fankam, S. Tankeo and T. Efferth (2011). Cytotoxicity of some Cameroonian spices and selected medicinal plant extracts. J. Ethnopharmacol. 134 (3): 803-812.
- Kumar, S., and A. K. Pandey (2013). Chemistry and biological activities of flavonoids: An overview. Scientific World Journal. .Article ID 162750: 1-16. https://doi.org/10.1155/2013/162750
- Lefahi, M., N. Zaabat, R. Ayad, E. H. Makhloufi, L. Djarri, M. Benahmed, H. Laouer, G. Nieto and S. Akkal (2018). In Vitro assessment of total phenolic and flavonoid contents, antioxidant and photoprotective activities of crude methanolic extract of aerial parts of Capnophyllum peregrinum (L.) lange (Apiaceae) growing in Algeria. Medicine. 5 (2): 1- 26.
- Li, J., B.E. Carlson and A. A. Lacis. (2014). Application of spectral analysis techniques in the inter-comparison of aerosol data, Part 4: Synthesized analysis of multisensor satellite and ground-based AOD measurements using combined maximum covariance analysis. Atmos. Meas. Tech.7(1): 2531-2549.
- Liu, J., L. Jia, J. Kan and C. H. Jin (2013). In vitro and in vivo antioxidant activity of ethanolic extract of white button mushroom (Agaricus bisporus). Food Chem. Toxicol. 51(1) : 310–316.
- Mandal, S., S. Upadhyay, V. P. Singh & R. Kapoor (2015). Enhanced production of steviol glycosides in mycorrhizal plants: A concerted effect of arbuscular mycorrhizal symbiosis on transcription of biosynthetic genes. Plant Physio Biochem. 89, 100-106
- Manohar, V., N. A. Talpur, B. W. Echard, S. Lieberman, H. G. Preuss (2002). Effects of a water-soluble extract of maitake mushroom on circulating glucose/insulin concentrations in KK mice. Diabetes. Obes. Metab. 4 (1): 43-48.
- McCue, P., and K. Shetty (2003). Role of carbohydrate-cleaving enzymes in phenolic antioxidant mobilization from whole soybean fermented with Rhizopus oligosporus. Food. Biotech. 17(1): 27-37.
- Mehmooda, A., M. Ishaqa, L. Zhao, S. Yaqoob, B. Safdara, M. Nadeeme and M. Munird, C. Wanga (2012). Impact of ultrasound and conventional extraction techniques on bioactive compounds and biological activities of blue butterfly pea flower (Clitoria ternatea L.). Ultrason. Sonochem. 51(1):12-19.
- Mhaskar, A. V., K. Prakash, K. S. Vishwakarma and V. L. Maheshwari (2010). Callus induction and antimicrobial activity of seed and callus extracts of Clitoria ternatae L. Curr. Trends Biotechnol. Pharm. 3(4): 561-567.
- Moein, M. M., A. E. Beqalli and M. B. Rahim (2017). Bioanalytical method development and validation: Critical concepts and strategies. J. chro. Ana. Tech. Biomed. life Sci. 1043:3-11.
- Mohammadi, M., F. Masoumipour, M. Hassanshahian, T and Jafarinasab (2019). Study the antibacterial and antibiofilm activity of Carum copticum against antibiotic-resistant bacteria in planktonic and biofilm forms. Microb. pathogenog 129 (02): 99-105.
- Mohammadzedah, S., M. Sharriatpannahi, M. Hamedi, Y. Amanzedah, S. E. S. Ibrahimi and S. N. Ostad (2007). Antioxidant power of Iranian propilis extract. Food. Chem. 103 (1): 729-733.
- Mohammedi, Z. and F. Atik (2011). Impact of solvent extraction type on total polyphenols content and biological activity from Tamarix aphylla (L.) Karst. Int. J. of Pharm. Bio Sci. 2 (4): 609-6.
- Montgomery C. P., B. S. Vavra and P. V. Adem (2008). The premiere of infectious diseases. J. Infec. Dise. 198 (11): 1725. https://doi.org/10.1086/593140
- Mushtaq, Z., T. Iqbal, N.Ahmed and A. Jamil (2017). Antioxidants from selected indigenous plants possessing cyclotides. Oxid. commun. 40(1-i):102–119.
- Nair, R.T., J. Kalariya and S. Chanda (2005). Antibacterial activity of some selected Indian medicinalflora. Tuky J. Biol. 29: 41-47.
- Nisa, S., Y. Bibi, A. Waheed, M. Zia, S. Sarwar, S. Ahmed and M. F. Chaudhary (2011). Evaluation of anticancer activity of Debregeasia Salicifolia extract against estrogen receptor positive cell line. African J. Biotech. 10(6): 990-995.
- Ozcan, K., (2020). Antioxidant, antibacterial and enzyme inhibition activity capacity of Doronicum Macrolepis (SYNT&FREN): An epidemic plant of turkey. Saudi. Pharma. J. 28(1): 95-100.
- Padmanabhan, P., and S. N. Jangle (2012). Evaluation of DPPH radical scavenging activity and reducing power of four selected medicinal plants and their combinations. Int. J. Pharma. Sci. Drug. Res. 4(2): 143-146.
- Pantavos , A., R. Ruiter, E. F Feskens, C. E Keyser, A. Hofman, B. H. Stricker, O. H Franco and J. C K. Jong (2015). Total dietary antioxidant capacity, individual antioxidant intake and breast cancer risk: the Rotterdam study. Int. J. Cancer.136 (9): 2178-2186.
- Patil A.P. and V.R. Patil (2011). Comparative evaluation of in vitro antioxidant activity of root of blue and white flowered varieties of Clitoria ternatea Linn. Int. J. Pharm. 7: 485-491.
- Quy, D. D., E. A. Artik, L. T. N. Phuong, H. H. Lein, E. S. Felycia, I. Suryadi and H. J. Yi (2014). Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromaticai. 22 (3): 296-302.
- Rahman, M. M., M. R. Shahriar, N. S. Meghla, T. Ishika, P. Chaudhary and M. Kamruzzaman (2017). Antimicrobial activity of some medicinal plant extracts against gram positive and gram-negative bacteria in Bangladesh. Asian. J. Med. Bio. Res. 3(4): 405-411.
- Ramirez, G. A., A. I. Garber, A. Lecoeuvre, T. D. Angelo, C. Geoffrey Wheat and B. N. Orcutt (2019). Ecology of subseafloor crustal biofilms. Front. Microbiol. Doi. 10.3389.fmicb.2019.01983.
- Rege, A.A., and A. S. Chowdhary (2014). Evaluation of Alpha amylase and glucosidase inhibitory activities of Rhizophora mucronate. IJPSR. 5 (6): 2320-5148.
- Roopa, G., M. C. Madhusudhan, K. Triveni, N. E. Mokaya, H. S. Prakash and N Geetha (2015). Evaluation of antioxidant properties of Salacia macrosperma leaf extracts. Int. J. Res. Stud. Sci. Eng. Technol. 2: 58-63.
- Saeidi, S., M. Mohsenbeygi, J. Roustakhiz, F. Javadian and M. Hassanshahian. (2019). Antimicrobial and antibiofilm effects of Mentha piperita and Zataria multoflora on pathogenic bacteria. J. Med. Bacteriol. 8(1): 37-44.
- Sahu, N. A., S. Hemalatha and K. Sairam (2013). HPTLC fingerprinting and in vitro antioxidant studies of argyreia speciosa sweet leaves and mesua ferrea linn. Flowers. Int. J. Res. Ayurveda Pharm. 4(4): 499-502.
- Sakanaka, S., Y. Tachibana and Y. Okada (2005). Preparation and antioxidant properties of extracts of Japanese persimmon tea (kakinoha-cha). Food Chem. 89: 569–575.
- Sen, A., and A. Batra (2012). Evaluation of antimicrobial activity of different solvent extracts of medicinal plant: Melia azedarach L. Int. J. Curr. Pharm. Res. 4(2): 67-73.
- Sikander, T., A.A. Bhatti, Z. Mushtaq, M. Shahid, F. Nighat and S. Anwar (2021) Evaluation of the bioactive role of indegenous Viola odorata leaf extracts. Pak. J. Bot., 53(2): DOI: http://dx.doi.org/10.30848/PJB2021-2(23)
- Sapkota, R., R. D. Nancy and D. S. Rawat (2012). Antibacterial effects of plants extract on human microbial pathogens & microbial limit tests. Int. J. Res. Pharm. Chem. 2(4): 926-936.
- Sarker, S. D., L. Nahar and Y. Kumarasamy (2007). Microtitre plate-based antibacterial assay incorporating resazurin as an indicator of cell growth, and its application in the in vitro antibacterial screening of phytochemicals. Methods. 42(4): 321-324.
- Shahrokhi, R. S., S. M. A. Zomorodian, A. Niazi and B. C. Okelly (2015). Improving sand with microbial-induced carbonate precipitation. Ground. Improv. 168 (3): 217-230
- Snafi, A. E. A., (2016). Pharmacological importance of Clitoria ternatea – A review. IOSR J. Pharm. 6(3): 68-83.
- Snafi, A. E. A., (2019). Iraqi medicinal plants with antifungal effect- A Review. IOSR J. Pharm. (IOSRPHR). 9 (7): 16-56.
- Sofidiyaa, M. O., E. Imeha, C. Ezeania, F. R. Aigbeb and A. J. Akindel. (2014). Antinociceptive and anti-inflammatory activities of ethanolic extract of Alafia barter. Rev. Bras. Farma. 24(1): 348-354.
- Stalika, C. D., (2007). Extraction, separation, and detection methods for phenolic acids and flavonoids. J. Sep Sci. 30(1): 3268-3295.
- Stankovik, S. M., (2011). Total phenolic content, flavonoid concentration and antioxidant activity of Marrubium peregrinum L. extracts. Kragujevac J. Sci. 33(1): 63-72.
- Taur, D.J., M.G. Waghmare, R.S. Bandal and R.Y. Patil (2011). Antinociceptive activity of Ricinus communis L. leaves. Asian Pac J. Trop. Biomed. 1(2): 139-141.
- Vivek, M.N., H.C. Swamy, M. Manasa, S. Pallavi, Y. Kambar, M.M. Asha and R. Onkarappa (2013). Antimicrobial and antioxidant activity of leaf and flower extract of Caesalpinia pulcherrima, Delonix regia and Peltaphorum ferrugineum. J. App. Pharma. Sci. 3 (8): 64-67.
- Venkatesan, T., Y-W. Choi and Y-K. Kim (2019). Impact of different extraction solvents on phenolic content and antioxidant potential of Pinus densiflora bark extract. BioMed. Res. Inter. https://doi.org/10.1155/2019/3520675
- Walter, M., and M. Marschesan (2011). Phenolic compounds and antioxidant activity of rice. Brazi. Arch. Biol Technol. 54 (2): 371-377.
- Wang, H., Y. J. Du and H. C. Song (2010). α-Glucosidase and α-amylase inhibitory activities of guava leaves. Food Chemistry. 123(1): 6-13.
- Wild, S., G. Roglic, A. Green, R. Sicree and H. King (2004). Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 27(5):1047-1053.
- Zaidan, M. R. S., A. N. Rain, A. R. Badrul, A. Afzan, N. Ahmad and I. Zakiyah (2005). In vitro screening of five local medicinal plants for antibacterial activity using disc diffusion method. Tropical biomedicine 22(2):165-170.
- Zhang, Y., Z. Luo, D. Wang, F. He and D. Li (2014). Phytochemical profiles and antioxidant and antimicrobial activities of the leaves of Zanthoxylum bungeanum. Sci. World. J. 115 (10): 1-13.
- Zoysa, M. H. N. D., H. Rathnayake, R. P. Hewawasam and W. M. D. G. B. Wijayaratne (2019). Determination of in vitro antimicrobial activity of five Srilankan medicinal plants against selected human pathogenic bacteria. Int. J. Microbiol. Article ID 7431439:Pages 1-8. https://doi.org/10.1155/2019/7431439
|





