ELUCIDATION OF NUTRITIONAL, PHYTOCHEMICAL AND PHARMACOLOGICAL ACTIVITIES OF TEUCRIUM POLIUM L GROWN IN LIBYA
R. Alghazeer1*, S. Elgahmasi2, E. Abdullah1, O. Ahtiwesh1, E. Althaluti3, G. Shamlan4, W. S. Alansari5 and A. A. Eskandrani6
1Biochemistry division, Chemistry Department, Faculty of Science, University of Tripoli, Tripoli, Libya.
2Department of Biochemistry, Faculty of Medicine, University of Tripoli, Tripoli, Libya; 3Department of Marine Chemistry and Physics, Marine Biology Research Center, Tajura-East of Tripoli, Tripoli, Libya; 4Department of Food Science and Nutrition, College of Food and agriculture Sciences, King Saud University, Riyadh 11362, Saudi Arabia; 5Biochemistry Department, Faculty of Science, University of Jeddah, Jeddah 21577, Saudi Arabia; 6Chemistry Department, Faculty of Science, Taibah University, Medina 30002, Saudi Arabia.
Corresponding Author’s E-mail: R.alghazeer@uot.edu.ly
ABSTRACT
Consumption of plant compounds play a crucial role in promoting health by improving the nutritional value of diet and preventing several chronic conditions such as cancer, cardiovascular, diabetes and neurological diseases. The current work aimed to analyse qualitatively and quantitatively the stem and leave extracts of Teucrium polium L.(Lamiaceae) grown in Libya for proximate, nutritional and phytochemical constituents as well as their biological properties. Methanolic extracts of leaves and stems were employed to evaluate their content of some of the primary and secondary compounds by several standard methods using a spectrophotometer. Additionally, the antioxidant properties of extracts were assessed using various in vitro systems including 1,1- diphenyl-2-picrylhydrazyl (DPPH), nitric oxide (NO) radicals scavenging ability, Lipid peroxidation activity, phosphomolybdenum reduction, and reducing power activity at different concentrations (6.25 to 100 mg/ml). Moreover, the cytotoxic effect of extracts was tested by measuring the Hemolytic activity against human red blood cells (hRBCs) while the anti-inflammatory was investigated by measuring the Inhibition of albumin denaturation. Results showed that the leaves had a higher concentration of carbohydrates, protein, fat, phenols, flavonoids, flavanols, tannins, catechins and coumarins, whereas the stems had a higher concentration of alkaloids. Correspondingly, the leaves displayed higher antioxidant properties (specifically, combatting lipid peroxidation) and a greater prevention of albumin denaturation than the stems. Both extracts inhibited hemolysis. Our findings provided valuable insight into the efficacy and applicability of plant compounds in the food and pharmaceutical industries.
Keywords:Teucrium polium; phytochemicals; antioxidant; hemolysis; anti-inflammatory
https://doi.org/10.36899/JAPS.2021.5.0345
Published online January 21, 2021
INTRODUCTION
There is increasing awareness that the consumption of medicinal plants is critical for improving or maintaining health, considering their vast array of nutritional components that have been shown to influence the biochemical activities in the human body (Gill et al., 2011; Sofowora et al., 2013; Kathirvel and Sujatha, 2016; Veiga et al., 2020). Phytochemicals, including phenolics, flavonoids, alkaloids, saponins, terpenoids and carotenoids, noteworthily, exhibit antimalarial, antitumor, antimicrobial, antidiabetic and antiulcerogenic properties (Nunes et al., 2012). Moreover, antioxidants detected in plants (Ko et al., 2015; Pavithra et al., 2016; Lourenço et al., 2019) have been shown to counter carcinogens (Rohman et al., 2010) and free radical scavengers in cellular systems (Kurutas, 2015). These substances trigger inflammation by generating reactive oxidative species, which often lead to tissue damage (Bishop, 2008; Aitken et al., 2009) and the development of chronic diseases, such as cancer and rheumatoid arthritis (Sugimoto et al., 2016). Emerging evidence has revealed that plant compounds may be used as a promising preventive measure or for the treatment of free radicals (Karawya et al., 2010; Boubekri et al., 2014; Sagnia et al., 2014; Jamshidi-Kia et al., 2020) as they inhibit inflammatory molecules such as prostaglandin E2, COX, 5-LOX, nitric oxide (NO), reduction in C-reactive protein (CRP), NFκB and various cytokines (Bajpai et al., 2014; Sagnia et al., 2014; Azab et al., 2016; Oguntibeju, 2018).
Teucrium (T) polium L. (Lamiaceae) is a wild flowering plant grown throughout Southwestern Asia, Europe and North Africa (Marzouk et al., 2016); it has been extensively utilized in traditional medicine for treating a wide range of pathological conditions (Stankovic et al., 2011; Jaradat, 2015; Hashemi et al., 2020). Previous laboratory phytochemical screenings of this plant have identified bioactive compounds such as glycosides (verbascoside and poliumoside); phenylethanoid; apigenin, 3’,6-dimethoxy apigenin, 4’,7-dimethoxy apigenin, rutin, flavonoids, tannins, terpenes and phenols (Sharififar et al., 2009; Bahramikia and Yazdanparast, 2012). These bioactive compounds exert cytotoxic, anticancer, antimutagenic, antioxidant and antibacterial effects on various cell lines (Capasso et al., 1984; Jurišić et al., 2003; Rajabalian, 2008; Khader et al., 2010; Shtukmaster et al., 2010; De Marino et al., 2012; El Atki et al., 2020). Of particular, longstanding interests are their ability to safeguard against the rupturing of red blood cells and hepatocytes by decreasing the amount of hydrogen peroxide- and Fe2+-induced lipid peroxidation, respectively (Suboh et al., 2004; Ljubuncic et al., 2005).
Currently, limited research has been conducted on T. polium L grown in Libya (Elmestiri, 2007; Abouzeed et al., 2013) and to our knowledge; no one has examined its specific antioxidant and anti-inflammatory profiles. Therefore, the purpose of this study was to identify bioactive compounds and assess the biological properties of samples grown in that country.
MATERIALS AND METHODS
Plant materials and alcoholic extraction: Fresh leaves and stems of T. polium were collected in August 2019 from Tarhwona, Libya. The collected plant was identified and authenticated by the Botany Department, Faculty of Science, University of Tripoli, Tripoli, Libya. The aerial parts (leaves and stems) of the plant were washed with tap water and air-dried at room temperature. The samples were ground into a powder, passed through a suitable mesh sieve and dried. The powdered plant parts were extracted with 95% methanol at 25°C for 48 h and after filtration, the samples were concentrated using a rotary evaporator (Heidolph, LaboRota 4000, Germany) under reduced pressure at 40°C. The residues were maintained at 20°C until analysis.
Chemicals: The chemicals, including 1,1'-diphenyl-2-picrylhydrazyl (DPPH), Folin–Ciocalteu phenol reagent, catechin, butylated hydroxyl anisole (BHA), coumarin, cortisone, phosphate buffered saline (PBS) and Anthron reagent were obtained from Sigma Chemical Company Ltd. (USA). Gallic acid, tannic acid, ascorbic acid, rutin, tannic acid, vitamin E (ɑ-tocopherol), β-carotene and bovine serum albumin (BSA) were obtained from Merck (Pvt.) Ltd. (Germany). Solvents and other reagents were of analytical grade.
Quantifying ash content: Ash content was determined according to Horwitz and Latimer (2007) and expressed as %w.
Quantifying total protein content: Total protein content was estimated according to the method described by Lowry et al. (1951). The amount of protein in 100 mg of extract was calculated by comparison with the standard curve for BSA.
Quantifying total carbohydrate content: Total carbohydrate content was estimated using Anthron reagent, as described by Hedge et al. (1962).
Extracting free fatty acids and quantifying total lipid content: Free fatty acids were first extracted according to the method described by Bligh and Dyer (1959). The free fatty acids in the lipid residue were calorimetrically estimated using a cupric acetate/pyridine reagent, as described by Lowry and Tinsley (1976).
Quantifying total ascorbic acid (vitamin C) content: Total ascorbic acid content was estimated according to the technique described by Ghate et al. (2013) and calculated on the basis of the calibration curve of L-ascorbic acid and expressed as mg of ascorbic acid equivalent per g of extract.
Quantifying total tocopherol (vitamin E) content: Total tocopherol content were determined using the method described by Wong et al. (1988), calculations were based on a standard curve of α-tocopherol (10–100 mg/mL in toluene) and it was expressed as mg of α-tocopherol equivalent per g of extract (mg α-tocopherol E/g).
Quantifying total carotenoid content: Total carotenoid content of crude extracts was determined according to Gentili and Caretti (2011) and expressed as mg of β-carotene equivalent per g of extract (mg β-carotene E/g).
Quantifying total phenolic content: Total phenolic content was analysed using the Folin–Ciocalteu colorimetric method by Singleton et al. (1999) and expressed as mg of gallic acid equivalent per g of extract (mg GAE/g).
Quantifying total flavonoid content. Total flavonoid content was estimated according to the method described by Zhishen et al. (1999) and expressed as mg of rutin (Sigma Chemical Company Ltd., USA) equivalent per g of extract (mg RE/g).
Quantifying total flavonol content: Total flavonol content was determined according to the procedure by Kumaran and Karunakaran (2007) and expressed as mg of rutin equivalent per g of dry weight (mg RE/g).
Quantifying total tannin content: Total tannin content was determined according to the method detailed by Julkunen-Tiitto (1985) and expressed as mg of tannic acid equivalent per g of dry weight (mg TAE/g).
Quantifying total alkaloid content: Total alkaloid content was determined according to the method described by Shamsa et al. (2008) and Sharief et al. (2014) and expressed as mg of atropine equivalent per g of extract (mg AE/g).
Quantifying total coumarin content: Total coumarin content was estimated following the standard methods by Rajat Buragohain (2015) and de carvalho Osório and Martins (2004) and expressed as mg of coumarin equivalent per g of extract (mg CE/g).
Quantifying total steroid content: Total steroid content was estimated according to Devanaboyina et al. (2013) and expressed as mg of cortisone equivalent per g of extract (mg QE/g).
Assessing in vitro biological activities
Assessing antioxidant actions: Investigations of in vitro antioxidant activity were according to different assays that examined their behaviour as radical scavengers or reducing agents.
Total antioxidant capacity was determined according to the procedure proposed by Prieto et al. (1999) at various concentrations (6.25–100 mg/mL).
The reducing power was determined according to the method detailed by Oyaizu (1986)) at various concentrations (6.25–100 mg/mL).
The free radical scavenging activity against 1,1-diphenyl-2-picrylhydrazine (DPPH) was evaluated as described by Wong et al. (2006).
The nitric oxide radical was measured spectrophotometrically according to Garratt (2012) at various concentrations (6.25–100 mg/mL).
Lipid peroxidation was evaluated according to Kuda et al. (2005), at various concentrations (6.25–100 mg/mL).
The scavenging activity of the DPPH and nitric oxide radicals as well as the inhibition of lipid peroxidation were calculated as percentages using the following equation (where AC was the absorbance of the control reaction and AS was the absorbance in the presence of the extracts)
(%) = [(𝐴C – 𝐴S)/𝐴C] × 100
Note: In all assays, ascorbic acid and butylated hydroxyl anisole (BHA) at various concentrations (6.25–100 mg/mL) were used as positive controls. The IC50 was calculated as the number of antioxidants required to inhibit 50% of the radical.
Assessing anti-inflammatory actions
Inhibiting albumin denaturation: This assay was performed according to the method detailed by Williams et al. (2008) at various concentrations (6.25–100 mg/ml) based on inhibition of albumin denaturation. Ascorbic acid and (BHA) were used as positive controls. The inhibition percentage of protein denaturation was calculated as follows (where D was the absorbance reading of the test sample and C was the absorbance reading without the test sample):
% Denaturation inhibition = (1 - D/C) ×100%.
Evaluating in vitro antihemolytic activity: Antihemolytic activity was assessed using human erythrocytes (O blood groups) as suggested by Takeshima et al. (2003) at various concentrations (6.25–100 mg/mL). A saline buffer was used as the negative control, and Triton X-100 was used as the positive control. Hemolysis percentage was determined as follows:
% Hemolysis = [(A sample - A buffer saline)/ (A Triton X-100 - A buffer saline)] × 100.
Statistical analyses: All the assays were repeated three times (n = 3), and values were expressed as mean ±Margin of Error (MOE). Statistical analysis was performed using SPSS (Statistical Program for Social Sciences) v.16 (SPSS Corporation, Chicago, IL). P values of ≤0.05 were considered statistically significant. Statistical significance was assessed using one-way ANOVA for the average over the time, followed by Tukey’s multiple comparisons test with a significance level set at p ≤ 0.05. Pearson correlation coefficient was determined between the antioxidant activities and primary, secondary metabolite contents.
RESULTS AND DISCUSSION
In vitro antioxidant activity: The antioxidant capacity of methanolic extract of leaves and stems was evaluated using several different methods based on two mechanisms, reducing capacity and free radical scavenging, to enable quick screening of compounds as tested samples that possess a reduced antioxidant impact in vitro will possibly show less effect in vivo (Nunes et al., 2012).
The phytochemical and biological properties of methanolic extracts of leaves and stems of T. polium grown in Libya were investigated. Specifically, we qualitatively and quantitatively evaluated their nutritive and non-nutritive (i.e. phytochemical) compounds as well as overall antioxidant, antihemolytic and anti-inflammatory activities.
Nutrients and phytochemical analyses: Phytochemical screening revealed the presence of different bioactive compounds and nutrients (Table 1). Phenols, tannins, glycosides, flavonoids, alkaloids, coumarins, terpenoids, steroids, resins, anthraquinones, emodins, proteins, carbohydrates and fats were found to be present in both leaf and stem extracts. The most abundant bioactive compounds were steroids, coumarins, terpenoids and resins (+++), followed by polyphenols, tannins, glycosides, flavonoids and fats (++), and then flavonols, proanthocyanidins, anthraquinones, emodins, glycosides, carbohydrates and proteins (+). Alkaloids and quinones were present in appreciable amount in the leaf extract (++) compared with that in the stem extract (+). Catechins were present only in the leaf extract (+). These findings are consistent with related previous phytochemical studies reporting the presence of tannins, diterpenoids (Piozzi et al., 2005), flavonoids (D’Abrosca et al., 2013), iridoids and teucardoside (Elmasri et al., 2015). Interestingly, our samples showed that saponins were absent, which is inconsistent with the results of (Elmasri et al., 2016), indicating that the composition of the samples is influenced by their geographic origin.
Table 1. Qualitative analysis of phytochemicals in leaves and stems of Teucrium polium.
|
S. No
|
Phytochemicals
|
Methanol extract
|
|
Leaf
|
Stem
|
|
1
|
Phenols
|
++
|
++
|
|
2
|
Tannins
|
++
|
++
|
|
3
|
Phlobatannins
|
̶
|
̶
|
|
4
|
Flavonoids
|
++
|
++
|
|
5
|
Flavonols
|
+
|
+
|
|
6
|
Anthocyanin
|
̶
|
̶
|
|
7
|
Anthocyanidins
|
̶
|
̶
|
|
8
|
Proanthocyanidins
|
+
|
+
|
|
9
|
Catechins
|
+
|
̶
|
|
10
|
Coumarins
|
+++
|
+++
|
|
11
|
Alkaloids
|
++
|
+
|
|
12
|
Saponins
|
̶
|
̶
|
|
13
|
Terpenoids
|
+++
|
+++
|
|
14
|
Steroids
|
+++
|
+++
|
|
15
|
Quinones
|
++
|
+
|
|
16
|
Anthraquinones & Emodins
|
+
|
+
|
|
17
|
Resins
|
+++
|
+++
|
|
18
|
Glycosides
|
+
|
+
|
|
19
|
Carbohydrates
|
+
|
+
|
|
20
|
Proteins
|
+
|
+
|
|
21
|
Fats/ Fixed oils
|
++
|
++
|
+++ Copiously present, ++ Moderately present, + Slightly present, - Absent
Nutrient contents: Apart from the potential use of medicinal plants as pharmacological agents, they can also serve as a source of food in countries wherein malnutrition is prevalent (Adesogan et al., 2020) owing to their rich nutrition. The nutrient contents of the leaves and stems tested in our study are summarised in Table 2. Ash content was greater in the leaves than in the stems (P ≤ 0.05). In general, the leaf was the major source of nutrition, and the amounts of these components, from greatest to least, are as follows: Vitamin C > carbohydrates > proteins > vitamin E > fats > carotenoids. These findings are inconsistent with those of previous research and it could be owing to differences in geographical regions, condition and structure of soil, environment, features, genetics, different parts of the plant and period of assessment (Imeh and Khokhar, 2002; Maqsood et al., 2020).
Phytochemical contents: The concentration of the various phytochemicals in the leaves and stems of Teucrium polium is presented in Table 3. Phenolic content. Phenols are widely found in medicinal plants and are proven to have strong antioxidative properties (Santos-Sánchez et al., 2019). In our study, the leaves had a significantly higher concentration of phenols than the stems (P ≤ 0.001). In previous studies, the phenolic content in leaves was found to be less than that found in this study (Stankovic et al., 2011; Stankovic et al., 2012).
Table 2.Nutritional contents (%) of leaves and stems of Teucrium polium.
|
S. No
|
Parameters (%)
|
Plant Part
|
P value
|
|
Leaf
|
Stem
|
|
1
|
Ash
|
16.62 ± 0.32
|
10.37 ± 0.22
|
0.05
|
|
2
|
Moisture
|
ND
|
ND
|
-
|
|
3
|
Carbohydrates
|
6.91 ± 0.53a
|
6.41 ± 0.50a
|
0.183
|
|
4
|
Proteins
|
1.58 ± 0.17b
|
0.25 ± 0.10a
|
0.0001
|
|
5
|
Fats
|
0.12 ± 0.02
|
0.05 ± 0.01
|
0.0001
|
|
6
|
Vitamin C
|
25.53 ± 0.62a
|
20.36 ± 1.83b
|
0.006
|
|
7
|
Vitamin E
|
0.26 ± 0.11a
|
0.20 ± 0.10a
|
0.468
|
|
8
|
Carotenoids
|
0.03 ± 0.01a
|
0.02 ± 0.01a
|
0.124
|
Data are expressed as means ± MOE (n=3). Values with different superscripts are significantly different.
Table 3. Phytochemical content of leaves and stems of Teucriumpolium.
|
S. No
|
Phytochemical
|
Unit
|
Plant Part
|
P value
|
|
Leaf
|
Stem
|
|
1
|
Phenols
|
mg GAE/g
|
2579.72 ± 81.31
|
1632.57 ± 77.19
|
0.0001*
|
|
2
|
Tannins
|
mg TAE/g
|
1.26 ± 0.05
|
0.58 ± 0.12
|
0.001*
|
|
3
|
Flavonoids
|
mg RE/g
|
52.63 ± 4.79
|
25.97 ± 0.33
|
0.008*
|
|
4
|
Flavonols
|
mg RE/g
|
4.32 ± 0.28
|
1.72 ± 0.35
|
0.0001*
|
|
5
|
Catechins
|
mg CaE/g
|
1.36 ± 0.28
|
-
|
-
|
|
6
|
Coumarins
|
mg CoE/g
|
45.68 ± 3.95
|
26.49 ± 4.48
|
0.006*
|
|
7
|
Alkaloids
|
mg AE/g
|
0.52 ± 0.26
|
1.46 ± 0.23
|
0.003*
|
|
8
|
Steroids
|
mg QE/g
|
107.84 ± 2.58
|
105.21 ± 3.01
|
0.236
|
Data are expressed as means ± MOE (n=3). Gallic acid, TA = Tannic acid, R = Rutin, Ca = Catechin; Co= coumarins A= atropine; Q: cortisone *significantly P ≤ 0.0.
Tannin content: Tannins exhibit many antioxidant, antimicrobial and anti-inflammatory properties (de Sousa Araújo et al., 2008; Amabeoku, 2009; Corrales et al., 2009; Hashemi et al., 2020). In our study, the concentration of tannins in leaves was significantly higher than that in stems (0.001). Notably, the content of tannins was the lowest compared with that of other identified phytochemical compounds (Table 3).
Flavonoid and flavonol contents: Extensive research has demonstrated a good correlation between flavonoid amounts and the degree of bioactivity in plants (Cakir et al., 2003; Al-Shalabi et al., 2020). In our study, the flavonoid content of the leaves was significantly higher than that of the stems (P ≤ 0.008). These findings are consistent with the results obtained by Bendjabeur et al. (2018), although the content of flavonoids in their leaves was almost twice as high as the amount detected in this study. Similarly, regarding the content of flavonols, it was markedly higher in the leaves than in the stems (P ≤ 0.0001) (Table 3).
Catechin content: Catechins have proven to be effective in preventing lipid oxidation, and therefore may be particularly useful in pharmaceutical products and extending the shelf life in food products (Wong et al., 2006). In our study, catechins were detected only in the leaves. In a related study, catechins were detected in the aerial parts of the plant. However, the amounts were lower than found in our study (Saif-Elnasr et al., 2019), presumably owing to estimations using HPLC (Table 3).
Coumarin content: Coumarins have been used to treat diseases owing to their low levels of toxins and side effects (Wang et al., 2009) and their anticoagulant, antioxidant, antimicrobial, anticancer, antidiabetic, analgesic and anti-inflammatory properties (Bansal et al., 2013; Borges Bubols et al., 2013; Matos et al., 2013; Xia et al., 2013). In this study, the content of coumarins in the leaves was significantly higher than that of the stems (P ≤ 0.006) (Table 3). This finding was lower than that reported by Purnavab et al. (2015).
Alkaloid content: Alkaloids possess antioxidant, antibacterial, anti-plasmodial, anticancer and anti-inflammatory properties (Alghazeer et al., 2013; Thawabteh et al., 2019). In our results, the level of alkaloids was significantly higher in the stems than in the leaves (P ≤ 0.001) (Table 3).
Steroid content: Steroid compounds have been isolated from natural sources and their bioactivities have been investigated (Ulubelen et al., 2000). The amount of steroids in leaves was greater than that in stems (Table 3).
Reducing power and total antioxidant activity: Examining the reducing power and total antioxidant activity are simple techniques that are used for preliminary estimation of a sample’s ability to combat free radicals. Previous studies have demonstrated a positive relationship between phenolic and flavonoid contents and antioxidant activity in many plants (Oktay et al., 2003; Sharififar et al., 2009). Dose response curves of the reducing powers and antioxidant activity of the leaves and stems of Teucriumpolium are presented in Figure 1a & b. It is noteworthy that the reducing power and total antioxidant activity were dependent on the concentration of extracts, wherein the highest effects were observed at the highest concentration (100 mg/mL). Interestingly, the reducing power for our extracts was lower than that observed for positive controls (Ascorbic acid and BHA). No significant differences were observed between our present findings and those of a study by (Stankovic et al., 2012).

Fig. 1. Reducing power and total antioxidant activity of leaves and stems of Teucrium polium. Data are expressed as mean ± MOE (n = 3): (A) Reducing power, (B) Total antioxidant activity. L: leaves, S: stems, BHA: Butylated hydroxyanisole, Asc: Ascorbic acid.
Free radical scavenging and anti-lipid peroxidation activities: Free radical scavenging activity using DPPH and NO radicals are presented in Figure 2a & b. Our results showed that the percentage of radical scavenging activity increased with increasing concentrations of the extracts. Maximum DPPH radical scavenging ability observed in the leaves and 28% in the stems. Across all concentrations, the leaves showed significantly higher DPPH scavenging activity than the stems (P ≤ 0.05). Specifically, the IC50 values for the DPPH scavenging activities, from greatest to least, are as follows: Asc > BHA > leaves > stems. Interestingly, the DPPH scavenging activity of the extracts was significantly lower than those of BHA and ascorbic acid (P ≤ 0.05) (Table 4). The same pattern of results was obtained using NO radicals, although our extracts showed a stronger effect against NO radicals than DPPH radicals. Both extracts showed minimal radical scavenging activity compared with positive controls. These findings are in line with those of previous studies (De Marino et al., 2012; Elmasri et al., 2017).
The anti-lipid peroxidation activity is shown in Figure 2c. The obtained results are similar to those of the DPPH and NO radicals scavenging activities, wherein the inhibition percentage of lipid peroxidation was dependent on the concentration of the extracts, and the leaves were better at inhibiting lipid peroxidation than the stems. Maximum inhibition of lipid peroxidation was exhibited by the leaves (Table 4). These results are consistent with those of related investigations (Panovska and Kulevanova, 2005; Krishnaiah et al., 2011; Vladimir-Knežević et al., 2014).
The NO and DPPH free radical scavenging activities as well as the anti-lipid peroxidation activity of the samples are also expressed as the inhibitory concentration IC50 in Table 4. IC50 values for the DPPH scavenging activities, from greatest to least, are as follows: Asc > BHA > leaves > stems. A similar pattern was observed for the NO scavenging and anti-lipid peroxidation activities. In comparison with ascorbic acid or BHA, the activity of the extracts was significantly lower (P ≤ 0.05). According to (Phongpaichit et al., 2007), extracts with IC50 values ranging between 10 and 50 mg/mL were considered to possess strong antioxidant activity, thereby confirming that our stems and leaves are powerful scavengers of free radicals.
Table 4. IC50 values for NO and DPPH free radical scavenging activities as well as the anti-lipid peroxidation activity of leaves and stems of Teucriumpolium.
|
Samples
|
IC50 (mg/mL)
|
|
NO˙
|
DPPH˙
|
Anti-LP
|
|
Leaf
|
9.375d
|
6.25c
|
12.5b
|
|
Stem
|
12.5c
|
12.5d
|
15.625c
|
|
BHA
|
4.5b
|
3.125b
|
12.5b
|
|
Asc
|
0.15625a
|
0.78125a
|
6.25a
|
Data are expressed as actual mean (n = 3): (A) DPPH (1,1-diphenyl-2-picrylhydrazine) radical scavenging activity, (B) NO (nitric oxide) radical scavenging activity, (C) lipid peroxidation. L: leaves, S: stems, BHA: Butylated hydroxy anisole, Asc: Ascorbic acid. Values with different superscripts are significantly different.
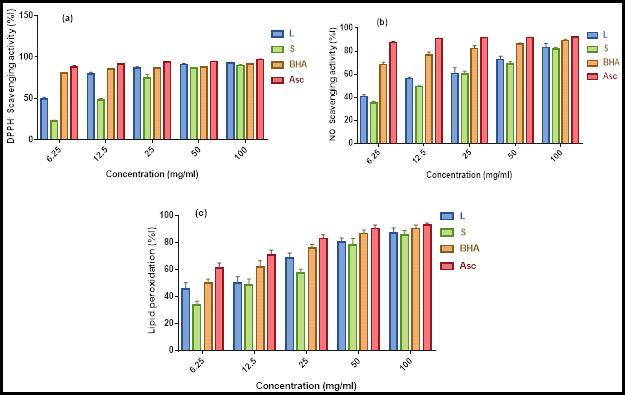
Fig. 2. Free radical scavenging and anti-lipid peroxidation activities of leaves and stems of Teucrium polium. Data are expressed as mean ± MOE (n = 3): (A) DPPH (1,1-diphenyl-2-picrylhydrazine) radical scavenging activity, (B) NO (nitric oxide) radical scavenging activity, (C) Lipid peroxidation activity. L: leaves, S: stems, BHA: Butylated hydroxyanisole, Asc: Ascorbic acid.
Antihemolytic activities: Erythrocyte membranes, due to the presence of polyunsaturated fatty acids, are the main target of free radicals (Fibach E, 2014). In an investigation examining hemolytic anaemia, researchers found that oxidative stress led to the induction of hemolysis (Fibach and Rachmilewitz, 2008). However, numerous studies have shown that plant compounds such as polyphenols and flavonoids are capable of safeguarding against such stress (Asgary et al., 2005; Kalaivani et al., 2011; Naqinezhad et al., 2012). Table 5 shows that the hemolysis induced by the leaves and stems in our study on red blood cells occurred in a concentration-dependent manner. Overall, the leaves showed a lower hemolytic effect than the stems, although the difference was not significant. In comparison with Triton X-100 (known to cause impressive red blood cell by membrane swelling and subsequent hemolysis), the extracts here displayed significantly lower hemolysis, indicating a low toxicity (Amrani et al., 2006). These results are consistent with those of (Suboh et al., 2004).
Table 5. Antihemolytic activity of leaves and stems of Teucrium polium.
|
Concentration (mg/ml)
|
Hemolysis activity (%)
|
| |
Leaf
|
Stem
|
|
6.25
|
|
1.67 ± 0.03a
|
2.13 ± 0.08a
|
|
12.5
|
|
2.00 ± 0.09a
|
2.40 ± 0.09a
|
|
25
|
|
2.33 ± 0.76a
|
3.13 ± 0.09a
|
|
50
|
|
3.01 ± 0.15a
|
4.27 ± 0.12a
|
|
100
|
|
4.86 ± 0.52a
|
5.13 ± 0.19a
|
|
0.1% Triton X-100
|
99.33 ± 1.30c
|
|
|
|
8.5 % Buffer saline
|
0.04 ± 0.03b
|
|
|
Data are expressed as means ± MOE (n=3). Values with different superscripts are significantly different (P ≤ 0.01)
Inhibition of albumin denaturation: It has been reported that protein denaturation is associated with inflammatory diseases such as arthritis; therefore, identifying substances that inhibit this denaturation are of great interest (Reddy et al., 2014). Figure 3 shows that the leaves and stems are both effective at inhibiting heat-induced albumin denaturation in a concentration-dependent manner. Of interest, the leaves were more potent than the stems (P ≤ 0.05), and no significant differences were observed with regard to the positive controls. These findings are consistent with previous observations (Mehrabani et al., 2009; Shah et al., 2012).
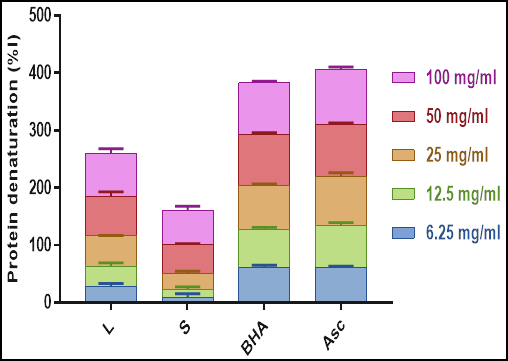
Fig. 3. Inhibition of albumin denaturation by leaves and stems of Teucrium polium. Data are expressed as means ± MOE (n=3). L: leaves; S: stems; BHA: Butylated hydroxyanisole; Asc: Ascorbic acid.
Correlations among nutrients, phytochemicals and the biological activities: Table 6 shows correlations among nutrients, phytochemicals and the biological activities. For the leaves, DPPH˙ activity was positively correlated with phenols and coumarins (P ≤ 0.05). A positive correlation was found between NO˙ activity and fat content (P ≤ 0.024). The reducing power in the leaves was positively correlated with the concentrations of carbohydrates, proteins and flavonoids (P ≤ 0.05). Anti-protein denaturation was positively correlated with tannins (P ≤ 0.986). For the stems, DPPH activity was negatively correlated with carotenes (P ≤ 0.02) and fat content (P ≤ 0.007). The total antioxidant capacity (TAC) was negatively correlated with tannins (P ≤ 0.05) and coumarins (P ≤ 0.006). The present findings are in agreement with those of previous studies (Felhi et al., 2016; Gan et al., 2017; Sayyad and Farahmandfar, 2017; Petropoulos et al., 2018). Bioactivity assays indicate that nutrients and phytochemicals exhibit redox properties, which allow them to act as reducing agents.
Table 6. Correlations between nutrients, phytochemicals and the biological activities in leaves and stems of Teucrium polium.
| |
DPPH˙
|
NO˙
|
Anti-PD
|
Anti-LP
|
TAC
|
RP
|
|
Leaf Extract
|
|
Carbohydrates
|
0.59
|
0.133
|
−0.978
|
−0.947
|
0.054
|
0.997*
|
|
Proteins
|
0.623
|
0.092
|
−0.968
|
−0.933
|
0.095
|
0.999*
|
|
Fats
|
−0.695
|
0.999*
|
−0.374
|
−0.478
|
−0.975
|
0.094
|
|
Vitamin C
|
−0.954
|
0.896
|
0.115
|
0.000
|
−0.963
|
−0.392
|
|
Vitamin E
|
−0.989
|
0.816
|
0.268
|
0.156
|
−0.910
|
−0.531
|
|
Carotenes
|
−0.760
|
0.098
|
0.904
|
0.849
|
−0.281
|
−0.988
|
|
Polyphenols
|
0.999*
|
−0.686
|
−0.453
|
−0.348
|
0.809
|
0.688
|
|
Tannins
|
−0.378
|
−0.368
|
0.999*
|
0.997
|
0.189
|
−0.949
|
|
Flavonoids
|
0.667
|
0.034
|
−0.952
|
−0.911
|
0.152
|
1.000*
|
|
Flavonols
|
0.326
|
−0.889
|
0.731
|
0.804
|
0.789
|
−0.506
|
|
Catechins
|
−0.851
|
0.251
|
0.826
|
0.756
|
−0.427
|
−0.952
|
|
Alkaloids
|
−0.553
|
−0.178
|
0.986
|
0.961
|
−0.009
|
−0.993
|
|
Coumarins
|
0.997*
|
−0.667
|
−0.476
|
−0.372
|
0.794
|
0.707
|
|
Stem Extract
|
|
Carbohydrates
|
−0.755
|
−0.312
|
0.264
|
0.921
|
0.934
|
−0.792
|
|
Proteins
|
−0.562
|
0.98
|
0.926
|
0.277
|
−0.466
|
−0.512
|
|
Fats
|
−1.000**
|
0.378
|
0.826
|
0.954
|
0.479
|
−0.999*
|
|
Vitamin C
|
−0.248
|
0.989
|
0.744
|
−0.065
|
−0.739
|
−0.190
|
|
Vitamin E
|
0.668
|
0.427
|
−0.143
|
−0.866
|
−0.971
|
0.711
|
|
Carotenes
|
−0.999*
|
0.417
|
0.85
|
0.94
|
0.441
|
−0.996
|
|
Polyphenols
|
0.586
|
−0.974
|
−0.937
|
−0.305
|
0.44
|
0.537
|
|
Tannins
|
0.536
|
0.571
|
0.023
|
−0.771
|
−0.997*
|
0.585
|
|
Flavonoids
|
0.668
|
0.427
|
−0.143
|
−0.866
|
−0.971
|
0.711
|
|
Flavonols
|
−0.289
|
0.994
|
0.772
|
−0.023
|
−0.710
|
−0.232
|
|
Alkaloids
|
0.034
|
−0.935
|
−0.583
|
0.277
|
0.866
|
−0.024
|
|
Coumarin
|
0.462
|
0.639
|
0.108
|
−0.714
|
−1.000**
|
0.513
|
DPPH˙: 1,1-diphenyl-2-picrylhydrazine radical activity, NO˙: nitric oxide radical activity, Anti-PD: anti-protein denaturation, Anti-LP: anti-lipid peroxidation, TAC: total antioxidant activity, RP: reducing power
*Correlation is significant at the 0.05 level (2-tailed), **Correlation is significant at the 0.01 level (2-tailed)
Conclusion: The nutritive and phytochemical compositions of Teucrium polium leaves extract are quite remarkable in contrast to their content in stems extract. Leaves extracts showed considered amount of carbohydrates, vitamin C, polyphenols, flavonoids, flavonols, and coumarins. In addition, the in vitro antioxidant efficacy of the extracts showed a dose-dependent effect, since decreasing concentrations of the extract had decreasing reducing and scavenging free radicals’ abilities as well as decreasing hemolysis and protein denaturation activities. Moreover, the results showed that noticeable correlations between the concentration of some nutrients, phytochemicals and the biological activities. Our findings provide valuable insight into the efficacy and applicability of Teucrium polium compounds in the food and pharmaceutical industries.
REFERENCES
- Abouzeed, Y.M., A. Elfahem, F. Zgheeland and M.O. Ahmed (2013). Antibacterial in-vitro activities of selected medicinal plants against methicillin resistant staphylococcus aureus from Libyan environment. Environ. Anal. Toxicol. 3(7):1-3.
- Adesogan, A.T., A.H. Havelaar, S.L. McKune, M. Eilittä and G.E. Dahl (2020). Animal source foods: Sustainability problem or malnutrition and sustainability solution? Perspective matters. Glob. Food Sec. 25, 100325. https://doi.org/10.1016/j.gfs.2019.100325.
- Aitken, S.L., E.L. Karcher, P. Rezamand, J.C. Gandy, M.J. Vandehaar, A.V. Capuco and L.M. Sordillo (2009). Evaluation of antioxidant and proinflammatory gene expression in bovine mammary tissue during the periparturient period. J. Dairy Sci. 92(2): 589-598. https://doi.org/10.3168/jds.2008-1551.
- Alghazeer, R., F. Whida, E. Abduelrhman, F. Gammoudi and M. Naili (2013). In vitro antibacterial activity of alkaloid extracts from green, red and brown macroalgae from western coast of Libya. Afr. J. Biotechnol. 12(51): 7086-7091.
- Al-Shalabi, E., M. Alkhaldi and S. Sunoqrot (2020). Development and evaluation of polymeric nanocapsules for cirsiliol isolated from Jordanian Teucrium polium L. as a potential anticancer nanomedicine. J. Drug Deliv. Sci. Technol. 56, 101544. https://doi.org/10.1016/j.jddst.2020.101544.
- Amabeoku, G.J. (2009). Antidiarrhoeal activity of Geranium incanum Burm. f. (Geraniaceae) leaf aqueous extract in mice. J. Ethnopharmacol. 123(1): 190-193. https://doi.org/10.1016/j.jep.2009.02.015.
- Amrani, S., H. Harnafi, N.E.H. Bouanani, M. Aziz, H.S. Caid, S. Manfredini, E. Besco, M. Napolitano and E. Bravo (2006). Hypolipidaemic activity of aqueous ocimum basilicum extract in acute hyperlipidaemia induced by triton WR‐1339 in rats and its antioxidant property. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives, 20(12): 1040-1045. https://doi.org/10.1002/ptr.1961.
- Asgary, S., G. Naderi and N. Askari (2005). Protective effect of flavonoids against red blood cell hemolysis by free radicals. Exp. Clin. Cardiol. 10(2): 88.
- Azab, A., A. Nassar and A.N. Azab (2016). Anti-inflammatory activity of natural products. Molecules, 21(10): 1321. https://doi.org/10.3390/molecules21101321.
- Bahramikia, S., and R. Yazdanparast (2012). Phytochemistry and medicinal properties of Teucrium polium L. (Lamiaceae). Phytother. Res. 26(11): 1581-1593. https://doi.org/10.1002/ptr.4617.
- Bajpai, S., R. Pathak and T. Hussain (2014). Anti-inflammatory activity of ethnobotanical plants used as traditional medicine: A review. Res. Rev. J. Bot. Sci. 3: 8-18.
- Bansal, Y., P. Sethi and G. Bansal. (2013). Coumarin: a potential nucleus for anti-inflammatory molecules. Med. Chem. Res. 22(7): 3049-3060. https://doi.org/10.1007/s00044-012-0321-6.
- Bendjabeur, S., O. Benchabane, C. Bensouici, M. Hazzit, A. Baaliouamer and A. Bitam (2018). Antioxidant and anticholinesterase activity of essential oils and ethanol extracts of Thymus algeriensis and Teucrium polium from Algeria. J. Food Meas. Charact. 12(4): 2278-2288. https://doi.org/10.1007/s11694-018-9845-x.
- Bishop, A. (2008). Role of oxygen in wound healing. J. wound care, 17(9): 399-402. https://doi.org/10.12968/jowc.2008.17.9.30937.
- Bligh, E.G., and W.J. Dyer (1959). A rapid method of total lipid extraction and purification. Canadian J. Biochem. Physiol. 37(8), 911-917. https://doi.org/10.1139/o59-099.
- Borges Bubols, G., D. Da rocha Vianna, A. Medina-remon, G. Von poser, R. Maria lamuela-Raventos, V. Lucia Eifler-lima and S. Cristina Garcia (2013). The antioxidant activity of coumarins and flavonoids. Mini. Rev. Med. Chem. 13(3): 318-334. https://doi.org/10.2174/138955713804999775.
- Boubekri, N., Z. Belloum, R. Boukaabache, A. Amrani, N. Kahoul, W. Hamama, D. Zama, O. Boumaza, H. Bouriche and F. Benayache (2014). In vivo anti-inflammatory and in vitro antioxidant activities of Genista quadriflora Munby extracts. Der Pharm. Lett. 6(1): 1-7.
- Cakir, A., A. Mavi, A. Yıldırım, M.E. Duru, M. Harmandar and C. Kazaz (2003). Isolation and characterization of antioxidant phenolic compounds from the aerial parts of Hypericum hyssopifolium L. by activity-guided fractionation. J. Ethnopharmacol. 87(1): 73-83. https://doi.org/10.1016/S0378-8741 (03)00112-0.
- Capasso, F., R. De fusco, M.P. Fasulo, M. Lembo, N. Mascolo and A. Menghini (1984). Antipyretic and antibacterial actions of Teucrium polium (L.). Pharmacol. Res. Commun. 16(1): 21-29. https://doi.org/10.1016/S0031-6989 (84)80101-0.
- Corrales, M., J.H. Han and B. Tauscher (2009). Antimicrobial properties of grape seed extracts and their effectiveness after incorporation into pea starch films. J. Food Sci. Technol. 44(2): 425-433. https://doi.org/10.1111/j.1365-2621.2008.01790.x.
- D’Abrosca, B., S. Pacifico, M. Scognamiglio, G. D’angelo, S. Galasso, P. Monaco and A. Fiorentino (2013). A new acylated flavone glycoside with antioxidant and radical scavenging activities from Teucrium polium leaves. Nat. Prod. Res. 27(4-5): 356-363. https://doi.org/10.1080/14786419.2012.695367.
- de Carvalho Osório, A., and J.L.S. Martins (2004) Determinação de cumarina em extrato fluido e tintura de guaco por espectrofotometria derivada de primeira ordem. Rev. Bras. Cienc. Farm. 40(4): 481-486. DOI: https://doi.org/10.1590/S1516-93322004000400005
- De Marino, S., C. Festa, F. Zollo, F. Incollingo, G. Raimo, G. Evangelista and M. Iorizzi (2012). Antioxidant activity of phenolic and phenylethanoid glycosides from Teucrium polium L. Food chem. 133(1): 21-28. https://doi.org/10.1016/j.foodchem.2011.12.054.
- De sousa Araújo, T.A., N.L. Alencar, E.L.C. De amorim and U.P. De albuquerque (2008). A new approach to study medicinal plants with tannins and flavonoids contents from the local knowledge. J. Ethnopharmacol. 120(1): 72-80. https://doi.org/10.1016/j.jep.2008.07.032.
- Devanaboyina, N., N. Ramalakshmi, B. Satyanarayana, P. Sudeepthi, K. Hemachakradhar and N. Pavankumar Raju (2013). Preliminary phytochemical screening, quantitative estimation and evaluation of antimicrobial activity of Alstonia macrophylla stem bark. Int. J. Sci. Invent. today, 2(1): 31-9.
- El Atki, Y., I. Aouam, F. El Kamari, A. Taroq, B. Lyoussi, B. Oumokhtar and A. Abdellaoui (2020). Phytochemistry, antioxidant and antibacterial activities of two Moroccan Teucrium polium L. subspecies: Preventive approach against nosocomial infections. Arab. J. Chem. 13(2), 3866-3874. https://doi.org/10.1016/j.arabjc.2019.04.001.
- Elmasri, W.A., M.E.F. Hegazy, Y. Mechref and P.W. Paré (2016). Structure-antioxidant and anti-tumor activity of Teucrium polium phytochemicals. Phytochem. Lett. 15: 81-87. https://doi.org/10.1016/j.phytol.2015.11.007.
- Elmasri, W.A., R. Zhu, W. Peng, M. Al-hariri, F. Kobeissy, P. Tran, A.N. Hamood, M.F. Hegazy, P.W. Paré and Y. Mechref (2017). Multitargeted flavonoid inhibition of the pathogenic bacterium Staphylococcus aureus: A Proteomic characterization. J. Proteome Res. 16(7): 2579-2586. https://doi.org/10.1021/acs.jproteome.7b00137.
- Elmasri, W.A., T. Yang, P. Tran, M.E.F. Hegazy, A.N. Hamood, Y. Mechref and P.W. Paré (2015). Teucrium polium phenylethanol and iridoid glycoside characterization and flavonoid inhibition of biofilm-forming Staphylococcus aureus. J. Nat. Prod. 78(1), 2-9. https://doi.org/10.1021/np5004092.
- Elmestiri, F.M. (2007). Evaluation of selected Libyan medicinal plant extracts for their antioxidant and anticholinesterase activities. (Doctoral dissertation, Newcastle University).
- Felhi, S., N. Baccouch, H.B. Salah, S. Smaoui, N. Allouche, N. Gharsallah and A. Kadri (2016). Nutritional constituents, phytochemical profiles, in vitro antioxidant and antimicrobial properties, and gas chromatography–mass spectrometry analysis of various solvent extracts from grape seeds (Vitis vinifera L.). Food sci. biotechnol. 25(6): 1537-1544. https://doi.org/10.1007/s10068-016-0238-9.
- Fibach, E. (2014). Involvement of oxidative stress in hemolytic anemia. Systems biology of free radicals and antioxidants, 2499-2516.
- Fibach, E., and E. Rachmilewitz (2008). The role of oxidative stress in hemolytic anemia. Curr. Mol. Med. 8(7): 609-619. https://doi.org/10.2174/156652408786241384.
- Gan, J., Y. Feng, Z. He, X. Li and H. Zhang (2017). Correlations between antioxidant activity and alkaloids and phenols of maca (Lepidium meyenii). J. Food Qual. 2017. https://doi.org/10.1155/2017/3185945.
- Garratt, D.C. (2012). The quantitative analysis of drugs, Springer Science & Business Media.
- Gentili, A., and F. Caretti (2011). Evaluation of a method based on liquid chromatography–diode array detector–tandem mass spectrometry for a rapid and comprehensive characterization of the fat-soluble vitamin and carotenoid profile of selected plant foods. J. Chromatogr. A, 1218(5), 684-697.
- Ghate, N.B., D. Chaudhuri and N. Mandal (2013). In vitro antioxidant and free radical scavenging assessment of Tinospora cordifolia stem with DNA protective potential. Int. J. Pharm. Bio. Sci. 4(1): 373-88.
- Gill, N.S., J. Bajwa, P. Sharma, K. Dhiman, S. Sood, P. Sharma, B. Singh and M. Bali (2011). Evaluation of antioxidant and antiulcer activity of traditionally consumed Cucumis melo seeds. J. pharmacol. Toxicol. 6(1): 82-9.
- Hashemi, S.F., N. Tasharrofi and M.M. Saber (2020). Green synthesis of silver nanoparticles using Teucrium polium leaf extract and assessment of their antitumor effects against MNK45 human gastric cancer cell line. J. Mol. Struct. 1208, 127889. https://doi.org/10.1016/j.molstruc.2020.127889.
- Hedge, J., B. Hofreiter and R. Whistler (1962). Carbohydrate chemistry. Academic Press, New York, 17.
- Horwitz, W., and G.W. Latimer (2007). Official methods of analysis of AOAC International, 2000. Gaithersburg, Maryland.
- Imeh, U., and S. Khokhar (2002). Distribution of conjugated and free phenols in fruits: antioxidant activity and cultivar variations. J. Agric. Food Chem. 50(22): 6301-6306. https://doi.org/10.1021/jf020342j.
- Jamshidi-Kia, F., J.P. Wibowo, M. Elachouri, R. Masumi, A. Salehifard-Jouneghani, Z. Abolhasanzadeh and Z. Lorigooini (2020). Battle between plants as antioxidants with free radicals in human body. J. Herbmed Pharmacol. 9(3), 191-199. DOI: 10.34172/jhp.2020.25
- Jaradat, N.A. (2015). Review of the Taxonomy, Ethnobotany, Phytochemistry, Phytotherapy and Phytotoxicity of Germander Plant (Teucrium polium L.). Medicine, 3: 4.
- Julkunen-Tiitto, R. (1985). Phenolic constituents in the leaves of northern willows: methods for the analysis of certain phenolics. J. Agric. Food Chem. 33(2): 213-217. https://doi.org/10.1021/jf00062a013.
- Jurišić, R., S. Vladimir-Knežević, Z. Kalođera and J. Grgić (2003). Determination of selenium in Teucrium species by hydride generation atomic absorption spectrometry. Z. Naturforsch. C, 58(1-2): 143-145. https://doi.org/10.1515/znc-2003-1-224.
- Kalaivani, T., C. Rajasekaran, K. Suthindhiran and L. Mathew (2011). Free radical scavenging, cytotoxic and hemolytic activities from leaves of Acacia nilotica (l.) wild. ex. delile subsp. indica (benth.) brenan. Evid. Based Complementary Altern. Med. 2011. https://doi.org/10.1093/ecam/neq060.
- Karawya, M.S., N.M. Ammar and M.S. Hifnawy (2010). Phytochemical study and evaluation of the anti-inflammatory activity of some medicinal plants growing in Egypt. Med. j. islamic world acad. sci. 109(407): 1-12.
- Kathirvel, A., and V. Sujatha (2016). Phytochemical studies, antioxidant activities and identification of active compounds using GC–MS of Dryopteris cochleata leaves. Arab. J. Chem. 9: S1435-S1442. https://doi.org/10.1016/j.arabjc.2012.03.018.
- Khader, M., N. Bresgen and P. Eckl (2010). Antimutagenic effects of ethanolic extracts from selected Palestinian medicinal plants. J. Ethnopharmacol. 127: 319-324. https://doi.org/10.1016/j.jep.2009.11.001.
- KO, E.Y., D. Kim, S.W. Roh, W.J. Yoonc, Y.J. Jeon, G. Ahn and K.N. Kim (2015). Evaluation on antioxidant properties of sixteen plant species from Jeju Island in Korea. EXCLI journal, 14: 133. DOI: 10.17179/excli2014-589.
- Krishnaiah, D., R. Sarbatly and R. Nithyanandam (2011). A review of the antioxidant potential of medicinal plant species. Food bioprod. Process. 89(3): 217-233. https://doi.org/10.1016/j.fbp.2010.04.008.
- Kuda, T., M. Tsunekawa, H. Goto and Y. Araki (2005). Antioxidant properties of four edible algae harvested in the Noto Peninsula, Japan. Food Compos. Anal. 18(7): 625-633. https://doi.org/10.1016/j.jfca.2004.06.015.
- Kumaran, A., and R.J. Karunakaran (2007). In vitro antioxidant activities of methanol extracts of five Phyllanthus species from India. LWT-Food Sci. Technol. 40(2): 344-352. https://doi.org/10.1016/j.lwt.2005.09.011.
- Kurutas, E.B. (2015). The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: current state. Nutr. J. 15(1): 1-22. https://doi.org/10.1186/s12937-016-0186-5.
- Ljubuncic, P., H. Azaizeh, I. Portnaya, U. Cogan, O. Said, K.A. Saleh and A. Bomzon (2005). Antioxidant activity and cytotoxicity of eight plants used in traditional Arab medicine in Israel. J. Ethnopharmacol. 99(1): 43-47. https://doi.org/10.1016/j.jep.2005.01.060.
- Lourenço, S.C., M. Moldão-martins and V.D. Alves (2019). Antioxidants of natural plant origins: From sources to food industry applications. Molecules, 24(22): 4132. https://doi.org/10.3390/molecules24224132.
- Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265-275.
- Lowry, R.R., and I.J. Tinsley (1976). Rapid colorimetric determination of free fatty acids. J. Am. Oil Chem.' Soc. 53(7): 470-472. https://doi.org/10.1007/BF02636814.
- Maqsood, S., O. Adiamo, M. Ahmad and P. Mudgil (2020). Bioactive compounds from date fruit and seed as potential nutraceutical and functional food ingredients. Food chem. 308, p.125522. https://doi.org/10.1016/j.foodchem.2019.125522.
- Marzouk, R.I., S.M. El-Darier and A.M. Askar (2016). Nutlet micromorphological characters of Teucrium taxa (Lamiaceae) in Libya. Phytotaxa, 263(3): 245-254. http://dx.doi.org/10.11646/phytotaxa.263.3.5.
- Matos, M.J., S. Vazquez-rodriguez, L. Santana, E. Uriarte, C. Fuentes-edfuf, Y. Santos and A. Muñoz-Crego (2013). Synthesis and structure-activity relationships of novel amino/nitro substituted 3-arylcoumarins as antibacterial agents. Molecules, 18: 1394-1404. https://doi.org/10.3390/molecules18021394.
- Mehrabani, D., A. Rezaee, N. Azarpira, M.R. Fattahi, M. Amini, N. Tanideh, M.R. Panjehshahin and M. Saberi-Firouzi (2009). The healing effects of Teucrium polium in the repair of indomethacin-induced gastric ulcer in rats. Saudi Med. J. 30: 494-9.
- Naqinezhad, A., S.M. Nabavi, S.F. Nabavi and M.A. Ebrahimzadeh (2012). Antioxidant and antihemolytic activities of flavonoid rich fractions of Artemisia tschernieviana Besser. Eur. Rev. Med. Pharmacol. Sci. 16: 88-94.
- Nunes, X.P., S. Silva, J.R.G.D.S. Almeida, J.M. Barbosa Filho, J.T. De Lima, L.A. DE Araújo Ribeiro and L.J.Q. Júnior (2012). Biological oxidations and antioxidant activity of natural products. 1-20. INTECH Open Access Publisher, New York.
- Oguntibeju, O.O. (2018). Medicinal plants with anti-inflammatory activities from selected countries and regions of Africa. J. Inflamm. Res. 11: 307. DOI: 10.2147/JIR.S167789.
- Oktay, M., İ. Gülçin and Ö.İ. Küfrevioğlu (2003). Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. LWT-Food Sci. Technol. 36(2): 263-271. https://doi.org/10.1016/S0023-6438 (02)00226-8.
- Oyaizu, M. (1986). Studies on products of browning reaction: antioxidative activity of products of browning reaction. Jpn. J. Nutr. 44(6): 307-315.
- Panovska, T.K., and S. Kulevanova (2005). Effect of some Teucrium species (Lamiaceae) on lipid peroxidation in rat liver microsomes. Fresenius Environmental Bulletin, 14(10): 957-959.
- Pavithra, S., J. Manibala and J. Ramachandran (2016). Evaluation of in-vitro antioxidant and fibrinolytic activity of flavonoid-rich fraction from the whole plant of wedelia Chinensis. Asian J. Pharm. Clin. Res. 9: 234-9.
- Petropoulos, S.A., C. Pereira, N. Tzortzakis, L. Barros I.C. Ferreira (2018). Nutritional value and bioactive compounds characterization of plant parts from Cynara cardunculus L. (Asteraceae) cultivated in central Greece. Front. Plant Sci. 9: 459. DOI: 10.3389/fpls.2018.00459.
- Phongpaichit, S., J. Nikom, N. Rungjindamai, J. Sakayaroj, N. Hutadilok-Towatana, V. Rukachaisirikul and K. Kirtikara (2007). Biological activities of extracts from endophytic fungi isolated from Garcinia plants. FEMS Immunol. Med. Microbiol. 51: 517-525. https://doi.org/10.1111/j.1574-695X.2007.00331.x.
- Piozzi, F., M. Bruno and S. Rosella (2005). Advances on the chemistry of furano-diterpenoids from Teucrium genus. Heterocycles, 65(5), 1221-1234.
- Prieto, P., M. Pineda and M. Aguilar (1999). Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Biochem. 269(2), 337-341. https://doi.org/10.1006/abio.1999.4019.
- Purnavab, S., S. Ketabchi and V. Rowshan (2015). Chemical composition and antibacterial activity of methanolic extract and essential oil of Iranian Teucrium polium against some of phytobacteria. Nat. prod. res. 29(14): 1376-1379. https://doi.org/10.1080/14786419.2014.1000320.
- Rajabalian, S. (2008). Methanolic extract of Teucrium polium L potentiates the cytotoxic and apoptotic effects of anticancer drugs of vincristine, vinblastine and doxorubicin against a panel of cancerous cell lines. Exp. Oncol.
- Rajat Buragohain, R. (2015). Screening and Quantification of Phytochemicals and Evaluation of Antioxidant Activity of Albizia chinensis (Vang). One of the Tree Foliages Commonly Utilized for Feeding to Cattle and Buffaloes in Mizoram. Int. J. Curr. Microbiol. Appl. Sci. 49: 305-313.
- Reddy, V.J., P.G. Rao and G.R. Lakshmi (2014). A review on antiarthritic activity of some medicinal plants. J. Glob. Trends Pharm. Sci. 5(4): 2061-73.
- Rohman, A., S. Riyanto, N. Yuniarti, W. Saputra, R. Utami and W. Mulatsih (2010). Antioxidant activity, total phenolic, and total flavaonoid of extracts and fractions of red fruit (Pandanus conoideus Lam). Int. Food Res. J. 17: 97-106.
- Sagnia, B., D. Fedeli, R. Casetti, C. Montesano, G. Falcioni and V. Colizzi (2014). Antioxidant and anti-inflammatory activities of extracts from Cassia alata, Eleusine indica, Eremomastax speciosa, Carica papaya and Polyscias fulva medicinal plants collected in Cameroon. PloS one, 9(8), e103999.
- Saif-Elnasr, M., S.M. Abdel Fattah and T.A. Mohamed (2019). Ameliorative Effect of Teucrium polium Extract on γ-radiation Toxicity in Brain of Albino Rats. Egyptian Journal of Radiation Sciences and Applications, 32(1): 31-50. DOI: 10.21608/ejrsa.2019.9867.1065.
- Santos-Sánchez, N.F., R. Salas-coronado, C. Villanueva-Cañongo and B. Hernández-Carlos (2019). Antioxidant compounds and their antioxidant mechanism. In Antioxidants. IntechOpen. DOI: 10.5772/intechopen.85270.
- Sayyad, R., and R. Farahmandfar (2017). Influence of Teucrium polium L. essential oil on the oxidative stability of canola oil during storage. J. food sci. technol. 54(10): 3073-3081. https://doi.org/10.1007/s13197-017-2743-0.
- Shah, S.M.M., F. Ullah, S.M.H. Shah, M. Zahoor and A. Sadiq (2012). Analysis of chemical constituents and antinociceptive potential of essential oil of Teucrium Stocksianum bioss collected from the North West of Pakistan. BMC Complement. Altern. Med. 12(1): 244. https://doi.org/10.1186/1472-6882-12-244.
- Shamsa, F., H. Monsef, R. Ghamooshi and M. Verdian-rizi (2008). Spectrophotometric determination of total alkaloids in some Iranian medicinal plants. Thai. J. Pharm. Sci. 32: 17-20.
- Sharief, N., A. Srinivasulu and V. Uma Maheshwara Rao (2014). Estimation of alkaloids and total phenol in roots of Derris trifoliate and evaluation for antibacterial and antioxidant activity. Indian J. Appl. Res. 4(5): 1-3.
- Sharififar, F., G. Dehghn-Nudeh and M. Mirtajaldini (2009). Major flavonoids with antioxidant activity from Teucrium polium L. Food chem. 112: 885-888. https://doi.org/10.1016/j.foodchem.2008.06.064.
- Shtukmaster, S., P. Ljubuncic and A. Bomzon (2010). The effect of an aqueous extract of Teucrium polium on glutathione homeostasis in vitro: a possible mechanism of its hepatoprotectant action. Adv. Pharmacol. Sci. 2010. https://doi.org/10.1155/2010/938324.
- SIngleton, V.L., R. Orthofer and R.M. Lamuela-Raventós (1999). Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. Elsevier. https://doi.org/10.1016/S0076-6879 (99)99017-1.
- Sofowora, A., E. Ogunbodede and A. Onayade (2013). The role and place of medicinal plants in the strategies for disease prevention. Afr. J. Tradit. Complement. Altern. Med. 10(5): 210-229.
- Stankovic, M.S., M.G. Curcic, J.B. Zizic, M.D. Topuzovic, S.R. Solujic and S.D. Markovic (2011). Teucrium plant species as natural sources of novel anticancer compounds: antiproliferative, proapoptotic and antioxidant properties. Int. J. Mol. Sci. 12: 4190-4205. https://doi.org/10.3390/ijms12074190.
- Stankovic, M.S., N. Niciforovic, V. Mihailovic, M. Topuzovic and S. Solujic (2012). Antioxidant activity, total phenolic content and flavonoid concentrations of different plant parts of Teucrium polium L. subsp. polium. Acta Soc. Bot. Pol. 81(2).
- Suboh, S.M., Y.Y. Bilto and T.A. Aburjai (2004). Protective effects of selected medicinal plants against protein degradation, lipid peroxidation and deformability loss of oxidatively stressed human erythrocytes. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives, 18:(4) 280-284. https://doi.org/10.1002/ptr.1380.
- Sugimoto, M.A., L.P. Sousa, V. Pinho, M. Perretti and M.M. Teixeira (2016). Resolution of inflammation: what controls its onset? Front. immunol. 7: 160. https://doi.org/10.3389/fimmu.2016.00160.
- Takeshima, K., A. Chikushi, K.K. Lee, S. Yonehara and K. Matsuzaki (2003). Translocation of analogues of the antimicrobial peptides magainin and buforin across human cell membranes. J. Biol. Chem. 278(2): 1310-1315.
- Thawabteh, A., S. Juma, M. Bader, D. Karaman, L. Scrano, S.A. Bufo and R. Karaman (2019). The biological activity of natural alkaloids against herbivores, cancerous cells and pathogens. Toxins, 11(11): 656. https://doi.org/10.3390/toxins11110656.
- Ulubelen, A., G. Topu and U. Sönmez (2000). Chemical and biological evaluation of genus Teucrium. Stud. Nat. Prod. Chem. 23: 591-648. Elsevier. https://doi.org/10.1016/S1572-5995 (00)80139-8.
- Veiga, M., E.M. Costa, S. Silva and M. Pintado (2020). Impact of plant extracts upon human health: A review. Crit. Rev. Food Sci. Nutr. 60(5), 873-886. https://doi.org/10.1080/10408398.2018.1540969.
- Vladimir-Knežević, S., B. Blažeković, M. Kindl, J. Vladić, A.D. Lower-Nedza and A.H. Brantner (2014). Acetylcholinesterase inhibitory, antioxidant and phytochemical properties of selected medicinal plants of the Lamiaceae family. Molecules, 19: 767-782. https://doi.org/10.3390/molecules19010767.
- Wang, H., X. Lu, H. Yao, J. Feng and R. Liu (2009). Research progress on application of coumarin and its derivatives. Chemical Industry Times, 23(8): 40-3.
- Williams, L.A.D., A. O'connar, L. Latore, O. Dennis, S. Ringer, J. Whittaker, J. Conrad, B. Vogler, H. Rosner and W. Kraus (2008). The in vitro anti-denaturation effects induced by natural products and non-steroidal compounds in heat treated (immunogenic) bovine serum albumin is proposed as a screening assay for the detection of anti-inflammatory compounds, without the use of animals, in the early stages of the drug discovery process. West Indian Med. J. 57(4).
- Wong, C.C., H.B. Li, K.W. Cheng and F. Chen (2006). A systematic survey of antioxidant activity of 30 Chinese medicinal plants using the ferric reducing antioxidant power assay. Food chem. 97(4): 705-711. https://doi.org/10.1016/j.foodchem.2005.05.049.
- Wong, M.L., R.E. Timms and E. Goh (1988). Colorimetric determination of total tocopherols in palm oil, olein and stearin. J. Am. Oil Chem.' Soc. 65, https://doi.org/10.1007/BF02636412.
- Xia, L., Y.B. Wang, W.L. Huang and H. Qian (2013). Research advance of anticancer drugs with cou‐marin structures. Chin. J. New Drugs, 23: 2392-404.
- Zhishen, J., T. Mengcheng and W. Jianming (1999). The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food chem. 64: 555-559. https://doi.org/10.1016/S0308-8146 (98)00102-2.
|





