OPTIMAL PREPARATION OF ROSA CENTIFOLIA ‘UAF’ CUTTINGS
I. Ahmad*, K. Ziaf, M. Shahid, H. A. Iqbal, H. A. Saeed and H. K. Ali
Institute of Horticultural Sciences, University of Agriculture, Faisalabad-38040, Pakistan.
*Corresponding author’s email: iftikharahmadhashmi@gmail.com, iahmad@uaf.edu.pk
ABSTRACT
Rosa centifolia ‘UAF’ is being commercialized as a rose with high potential market value. A study was conducted comparing plantings from Nov. 24th to Feb. 10th in a subtropical climate. Traditional callusing method (cuttings buried in moist soil for 15 d) was compared to the use of synthetic auxins. Three experiments were set up in a split plot design having four replicates of 200 cuttings each. Cuttings planted on 24th Nov. and treated with indole 3-butyric acid (IBA) exhibited best survival rate (83%), highest number of leaves per branch (20) and number of branches per cutting (4). Among cutting types, hardwood cuttings treated with naphthalene acetic acid (NAA) had highest number of leaves per branch (6.5), plant diameter (13.3 cm) and fresh (3.0 g) and dry weight of a shoot (1.3 g). While cuttings prepared with straight cut on upper end and a slanting cut on lower end and treated with (NAA) application had better sprouting percentage (68.0%), number of branches per plant (2.9) and root length (17.7 cm). In summary, hardwood cuttings, with straight upper and slanting cut on lower end, prepared during early propagation season (Nov. 24th) and dipped in IBA solution for 5-10 sec. proved best for enhancing rooting percentage and survival rate of Rosa centifolia ‘UAF’ cuttings and may be used by nurserymen for commercial propagation.
Key words: Propagation, rose, synthetic auxins, callusing, nursery raising
https://doi.org/10.36899/JAPS.2021.6.0373
Published online March 31, 2021
INTRODUCTION
Rosa centifolia, a member of the family Rosaceae, is an economically important rose species, used as a garden plant, cultivated for medicinal purposes or used in the perfume industry (Semangun et al., 2007). Market value of Rosa centifolia is expanding, while cultivation is not keeping pace. Most imperative items prepared from Rosa centifolia are oil (attar), dried rose petals, perfumes, ascorbic acid (vitamin C) from hips, jam, honey, wine, marmalade (gulkand) and rose water (arq-e-gulab). These products have great economic significance in national and international trades (Farricielli, 2008).
Growth habit, flower form, fragrance, color and blooming period vary greatly among selections of Rosa centifolia. Vegetative propagation is the fundamental and sole method to reproduce plant species with true to type characters. Roses are propagated true to type from somatic cells through cuttings, grafting, layering and budding. Among these, stem cuttings are the simplest and common method for mass propagation of roses (Anderson and Woods, 1999). However, due to variable rooting capacity, survival percentage and growth rate of various rose genotypes propagated through cuttings, optimization of propagation protocol is mandatory for each genotype.
Cuttings of Chrysanthemum morifolium cv. Vasanthika planted every two weeks from 15th July to 30th Sep. revealed that those planted early in the season during July and August had highest plant height, number of branches, flowering duration, plant canopy, flower size, and weight of flowers (Dahiya et al., 2007). Semi hardwood cuttings of various shrubs, planted from July to October showed that during July-August, there was better survival percentage, which lowered in late August to September, while there was a dramatic increase in bud growth and survival rate when cuttings were planted in October (Hansen and Kristiansen, 2000)[AY1] [IA2] .
A study optimizing the cutting wood age for Duranta repens cuttings disclosed hardwood cuttings to have maximum rooting percentage (Okunlola and Ibironke, 2013). Studies have shown that auxins are directly related to adventitious root formation in cuttings, which are either naturally produced in the plant or applied exogenously (Wendling et al., 2015; Gudeva et al., 2017). The concentration of auxin is also important in regulating the rooting capacity of cuttings (Ahkami et al., 2013). The rooting percentage and all growth parameters of Rosa centifolia cuttings were highest when callused with IBA as compared to other treatments, viz. IAA, NAA, BAP (Akhtar et al., 2015). However, Abbas et al. (2006) reported that Rosa centifolia cuttings produced highest rooting with Seradix-A as compared to IBA.
Being a new genotype of Rosa centifolia with profuse flowering at higher temperatures compared to other genotypes, there is a need to enhance cultivation of Rosa centifolia ‘UAF’, as large numbers of cuttings are needed. Keeping in view its significance, a study was conducted to optimize planting time, optimum cutting wood age, cutting preparation methods and callus induction, to enhance rooting and survival percentage, and to prepare uniform quality cuttings of Rosa centifolia ‘UAF’.
MATERIALS AND METHODS
A study with three field experiments was conducted at Floriculture Research Area, Institute of Horticultural Sciences, University of Agriculture, Faisalabad, Pakistan, during 2017-18. Healthy canes of Rosa centifolia ‘UAF’ were harvested from rose plantations of the institute. Experiments were laid out according to split plot design with four replications, while each experimental unit consisted of 200 cuttings.
Expt. 1.Staggered planting time and callus induction technique: Hardwood cuttings of Rosa centifolia ‘UAF’ of about 15 cm length having 3-5 nodes were taken from healthy canes of previous season growth. Cuttings were bunched and kept vertically buried with upside down to enhance callus formation in moist soil for 15 days (Traditional practice). These cuttings were dug out after callus formation and at that time fresh cuttings from same age canes were prepared. These freshly prepared cuttings were dipped in 2000 mg L-1 IBA solution for 5-10 seconds as a rooting hormone before planting. All cuttings (callused by traditional method or treated with IBA) were planted at same time in 10 × 15 cm size polythene bags filled with silt. There were six planting times at 15 days interval from 24th Nov. to 10th Feb., the normal propagation season in the study area, along with three callus induction methods, viz. no callusing or IBA, natural callusing by dumping cuttings in moist soil or artificial callusing by quick dip in IBA, that makes a sum of eighteen treatments replicated four times with 200 cuttings in each replication.
Expt. 2. Cutting wood age along with callus induction technique: Softwood (<6-month age), semi-hardwood (6–9-month-old) and hardwood (>9-month-old) cuttings were prepared and callused either using traditional callusing by burying in moist soil for 15 days before planting or 2000 mg L-1 NAA dip for 5-10 sec., making a count of six treatments in this experiment, which were replicated four times each with 200 cuttings.
Expt. 3.Cutting preparation method with callus induction technique: Cuttings were prepared using four different methods, viz. straight cut on both ends, slanting cut on upper ends with straight cut on lower ends, straight cut on upper ends with slanting cut on lower ends and slanting cut on both ends. These different sized cuts were made to expose greater cambial surface to enhance rooting %age of the cuttings. Moreover, these cuttings were either treated with traditional callusing by burying in moist soil for 15 days or quick dip in 2000 mg L-1 NAA that cumulatively constituted eight treatments in this experiment, which were replicated four times with 200 cuttings per replication.
Data collection: Data were collected on sprouting percentage (%), calculated based on total spouted cuttings after 90 days of planting out of total planted cuttings, length of shoots (recorded after 90 days of planting), number of branches per plant, and number of leaves per branch. Number of roots per cutting and root length were determined from ten plants of each replicate after 90 days of planting. For this purpose, plant was taken out of the polythene bag carefully, root system was washed and length was measured with meter rod. Fresh weight of sprouted shoots per cuttings was recorded from ten cuttings in each replicate using digital balance. After recording fresh weight, shoots were packed in perforated brown paper bags with small holes to let the moisture exit. The bags were labeled accordingly and kept in oven at constant temperature of 70◦C for 72 h. After drying to a constant weight, bags were taken out and dry weight of shoots were recorded in gram.
Statistical analysis: All data were analyzed using Fisher’s analysis of variance technique and treatment means were compared using least significance difference test at P<0.05 (Steel et al., 1997).
RESULTS AND DISCUSSION
Expt. 1. Staggered planting time and callus induction technique: Rose cuttings planted on 24th Nov. (1st planting in the season) along with traditional callusing produced maximum root length (12.9 cm), highest number of roots (9.6), fresh weight of roots (0.39 g) and dry weight of roots (0.153 g). However, greatest shoot length (7.13 cm), shoot fresh weight (2.36 g) and shoot dry weight (0.69 g) were recorded in cuttings planted on 10th Dec. and treated with IBA 2000 mg L-1 (Table 1; Fig. 1). While, maximum survival percentage (83.9%) and number of leaves (20.6) were produced in cuttings planted on 24th Nov. and treated with IBA 2000 mg L-1.
Cuttings planted on 10th Feb. and treated with indole-3-butyric acid (IBA) at 2000 mg L-1 showed lowest shoot length (2.43 cm), number of leaves (5.33), number of roots (3.66), length of roots (2.43 cm), and survival percentage (37.8%). However, minimum shoot fresh weight (0.66 g) and shoot dry weight (0.11 g) was found in cuttings planted on 10th Feb. without callusing (Fig. 1). While, least root fresh and dry weight was recorded in cuttings planted on 10th Dec. and 10th Jan. without callusing treatment, respectively (Fig. 1). These results demonstrated that planting cuttings early in the season provided more time for development and may have higher carbohydrates reserves for enhanced growth as evident from Hansen and Kristiansen (2000)[AY3] [IA4] , who reported better growth of selected ornamental shrubs cuttings planted early in the season compared to the cuttings planted late in the season. Similar results were recorded by Dahiya et al. (2007) who reported better survival rate and growth of chrysanthemum cuttings planted early in the season during July and August.
Fig. 1. Shoot fresh (A) and dry (B) weight and root fresh (C) and dry (D) weight of Rosa centifolia UAF cuttings as influenced by various planting times and callusing induction technique. Bars represent means of 30 samples ± S.E.
Table 1. Effect of staggered planting time and callus induction techniques on survival rate, number of leaves, shoot length, number of roots and length of roots of Rosa centifolia-UAF cuttings. Means are averages of 30 samples.
|
Treatments
Planting time
|
Callus induction (CI)
method
|
Survival (%)
|
Number of leaves
|
Shoot length after 90 days (cm)
|
Number of roots
|
Length of roots (cm)
|
|
Nov. 24
|
No callusing or IBA
Traditional callusingy
IBA quick dip @ 2000 mg L-1
|
64.0 bcdz
78.3 ab
83.9 a
|
12.66 cde
18.33 ab
20.66 a
|
4.66 cdef
5.03 bcd
6.56 ab
|
4.00 fg
9.66 a
7.00 bcd
|
8.37 bcde
12.90 a
9.63 abcd
|
|
Dec. 10
|
No callusing or IBA
Traditional callusing
IBA quick dip @ 2000 mg L-1
|
77.1 abc
71.5 abc
71.3 abc
|
16.33 abc
18.00 ab
14.33 bcd
|
3.63 def
4.20 cde
7.13 a
|
4.33 fg
8.33 abc
7.66 abcd
|
5.93 def
10.46 abc
7.70 bcd
|
|
Dec. 25
|
No callusing or IBA
Traditional callusing
IBA quick dip @ 2000 mg L-1
|
57.3 cde
56.3 def
73.8 abc
|
10.66 def
19.00 ab
18.66 ab
|
5.70 abc
4.90 bcd
5.43 abc
|
4.66 efg
9.00 ab
5.00 efg
|
6.20 def
10.53 ab
7.76 bcd
|
|
Jan. 10
|
No callusing or IBA
Traditional callusing
IBA quick dip @ 2000 mg L-1
|
65.3 abc
69.8 bcd
78.0 ab
|
26.66 fg
9.66 defg
9.66 defg
|
4.73 cde
5.10 bcd
4.03 cde
|
6.33 cde
8.66 abc
8.66 abc
|
7.77 efg
5.87 def
6.70 cde
|
|
Jan. 25
|
No callusing or IBA
Traditional callusing
IBA quick dip @ 2000 mg L-1
|
67.0 bcd
59.6 cde
75.0 bcd
|
5.33 g
8.66 efg
8.66 efg
|
4.43 def
4.73 cde
4.76 cde
|
5.33 def
7.00 bcd
4.00 fg
|
3.96 fgh
5.50 efg
14.86 efg
|
|
Feb. 10
|
No callusing or IBA
Traditional callusing
IBA quick dip @ 2000 mg L-1
|
53.2 ef
52.0 ef
37.8 f
|
7.33 fg
5.66 g
5.33 g
|
3.06 efg
2.97 fg
2.43 g
|
3.33 g
4.00 fg
3.66 g
|
3.233 gh
3.30 gh
2.43 h
|
|
Significancex
|
|
|
|
|
|
|
|
Planting time (PT)
|
0.0001
|
0.0005
|
0.0008
|
0.0310
|
0.0138
|
|
Callus induction (CI)
|
0.0001
|
NS
|
0.0001
|
0.0392
|
0.0020
|
|
PT × CI
|
0.0240
|
0.0457
|
NS
|
0.0028
|
NS
|
zMeans indicated with the same letter within a column are not significantly different at P≤0.05.
yTraditional callusing involved dumping of cuttings in moist soil for two weeks before planting.
xP values were obtained using General Linear Models of Statistix (version 8.1).
NS Non-significant at P>0.05.
Expt. 2. Cutting wood age along with callus induction technique: Hardwood cuttings callused with 2000 mg L-1 exhibited highest number of leaves per branch and shoot length (5.7 cm). However, survival percentage semi hard wood cuttings callused with 2000 mg L-1 NAA (40.7%) and shoot length of semi hardwood cuttings with traditional callusing (5.1cm) was statistically similar to respective parameter values in hardwood cuttings callused with 2000 mg L-1. Moreover, hard wood cuttings with traditional callusing produced higher number of roots per cutting (6.23), root length (21.6 cm), and fresh (3.02 g) and dry (1.28 g) weight of branch (Table 2; Fig. 2). Furthermore, roots per cutting and fresh weight per branch in semi-hardwood cuttings callused with 2000 mg L-1NAA was statistically at par with hardwood cuttings callused naturally. Softwood cuttings treated with 2000 mg L-1NAA produced highest number of branches per plant (3.3). Otiende and Maimba (2020) have also reported that sprouting percentage was increased when different growth hormones were applied on Rosa hybrida cuttings.
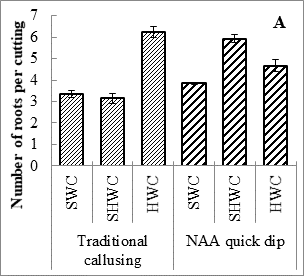 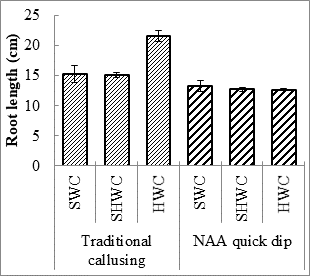
 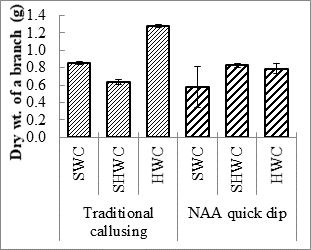
Fig. 2. Number of roots per cutting (A), root length (cm) (B), and fresh (C) and dry weight (D) weight of Rosa centifolia UAF cuttings as influenced by various cutting wood age/type and callusing induction technique. Bars represent means of 30 samples ± S.E.
Table 2. Effect of cutting wood age/type with or without callusing on sprouting percentage, number of leaves per branch, number of branches per plant and length of shoots after 90 days of Rosa centifolia cuttings. Means are averages of 30 samples.
|
Treatments
Cutting wood age
|
Callusing method
|
Sprouting percentage (%)
|
Number of leaves per branch
|
Number of branches per plant
|
Length of shoots (cm)
|
|
Softwood
|
Traditionalz
|
27.7 bcy
|
3.9 d
|
1.5 d
|
2.5 d
|
|
Semi hardwood
|
Traditional
|
29.7 b
|
5.9 b
|
1.9 c
|
5.1 a
|
|
Hardwood
|
Traditional
|
21.0 c
|
3.2 d
|
1.6 d
|
3.0 cd
|
|
Softwood
|
NAA @ 2000 mg L-1
|
29.2 b
|
4.5 c
|
3.3 a
|
3.3 ab
|
|
Semi hardwood
|
NAA @ 2000 mg L-1
|
40.7 a
|
5.1 bc
|
2.3 b
|
4.2 bc
|
|
Hardwood
|
NAA @ 2000 mg L-1
|
40.5 a
|
6.5 a
|
2.0 bc
|
5.7 a
|
|
Significancex
|
|
0.0002
|
0.0001
|
0.0001
|
0.0001
|
zTraditional callusing involved dumping of cuttings in moist soil for two weeks before planting.
yMeans indicated with the same letter within a column are not significantly different at P≤0.05.
xP values were obtained using General Linear Models of Statistix (version 8.1).
NS Non-significant at P>0.05.
Semi-hardwood or hardwood cuttings callused naturally or with NAA @ 2000 mg L-1 for 5-10 sec. may have increased the surface area of callus formation and early root formation, which provided additional time for growth and cell division that ultimately increased metabolites and food reserves in the shoots with increased shoot length (Otiende and Maimba, 2020) and fresh weight per branch as reported by Dick et al. (2004) and Leakey (2004) for hardwood cuttings of Triplochiton sceroxylon. Khan et al. (2004) and Okunlola and Ibironke (2013) also reported longest shoots when auxins were applied along with cutting wood age while propagating Rosa spp. and Duranta repens, respectively, through cuttings. Moreover, this increased fresh weight of branches was associated with higher number of leaves, indicating improved photosynthetic machinery, as reported earlier in Dalbergia sisso by Khudhur and Omar (2015). Hardwood cuttings with callusing may have increased rooting percentage, rooting capacity and root length as reported by Moalemi and Chehrazi (2005), that root length was increased when different auxins were applied for rooting of bougainvillea.
Expt. 3. Cutting preparation method with callus induction technique: Straight cut on upper ends with slanting cut on lower ends (USC) with quick dip in 2000 mg L-1NAA produced longest shoots after 90 days (6.5cm), number of roots per cutting (6.95), highest sprouting percentage (68.0%), number of branches (2.9) and number of leaves per branch (6.9), the latter two were statistically similar to slanting cut on both ends with dipping in 2000 mgL-1NAA (Table 3; Fig. 3). had highest fresh weight of branch (4.5g). Lower straight cut with upper slanting cut (LSC) with traditional callusing resulted in highest root length (17.7 cm) and fresh weight of branch (3.67 g) while, highest dry weight of branch (1.92g) was recorded in LSC with 2000 mgL-1NAA dipping.
Cuttings with straight cut on upper ends and slanting cut on lower ends (USC) treated with 2000 mg L‑1 NAA for 5-10 sec. had greatest effect on shoot length after 90 days, number of branches, number of leaves and root length. NAA stimulated cell division leading to formation of tissues in roots that ultimately increased uptake and supply of water and nutrients to leaves that may have increased photosynthesis and in turn food reserves in the shoot and increased shoot length by increase in shoots cell division and cell expansion. Root length was increased when rose and dwarf peach cuttings were treated with root promoting hormones (Khan et al., 2004; Nasri et al., 2015). Khan et al. (2004) also reported that sprouting percentage and length of shoots of Damask rose was increased when rooting hormones were applied.
Fig. 3. Number of roots per cutting (A), root length (cm) (B), and fresh (C) and dry weight (D) weight of Rosa centifolia ‘UAF’ cuttings as influenced by various types of cutting preparation method and callusing induction technique. Bars represent means of 30 samples ± S.E. SCB (straight cut on both ends), LSC (lower straight cut with upper slanting cut), USC (Upper straight cut with lower slanting cut) and SlCB (Slanting cut on both ends).
Table 3. Sprouting percentage, number of leaves per branches, number of branches per plant, shoot length after 90 days influenced by cutting wood preparation method with or without callusing. Means are averages of 30 samples.
|
Treatments
Cutting method
|
Callusing method
|
Sprouting percentage (%)
|
Number of leaves per branches
|
Number of branches per plant
|
Shoot length after 90 days (cm)
|
|
SCBz
|
Traditionaly
|
38.7 dx
|
5.1 c
|
1.8 c
|
3.7 d
|
|
LSC
|
Traditional
|
64.7 ab
|
6.1 b
|
2.5 ab
|
4.8 c
|
|
USC
|
Traditional
|
49.0 bcd
|
5.6 bc
|
2.3 bc
|
5.9 ab
|
|
SlCB
|
Traditional
|
50.2 bcd
|
5.5 bc
|
2.8 a
|
5.3 c
|
|
SCB
|
NAA @ 2000 mg L-1
|
45.0 cd
|
5.6 bc
|
2.6 ab
|
4.9 c
|
|
LSC
|
NAA @ 2000 mg L-1
|
58.2 abc
|
5.8 bc
|
2.6 ab
|
5.5 bc
|
|
USC
|
NAA @ 2000 mg L-1
|
68.0 a
|
6.9 a
|
2.9 a
|
6.5 a
|
|
SlCB
|
NAA @ 2000 mg L-1
|
41.7 cd
|
6.9 a
|
2.7 a
|
5.0 c
|
|
Significancew
|
|
**
|
**
|
***
|
**
|
zSCB (straight cut on both ends), LSC (lower straight cut with slanting upper end), USC (Upper straight cut with slanting cut on lower ends) and SlCB (Slanting cut on both ends).
yTraditional callusing involved dumping of cuttings in moist soil for two weeks before planting.
xMeans indicated with the same letter within a column are not significantly different at P≤0.05.
wP values were obtained using General Linear Models of Statistix (version 8.1).
Conclusion: Studies were conducted to optimize cutting preparation protocols for Rosa centifolia ‘UAF’, a leading rose in the country for value addition, which has high market demand. Results demonstrated that cuttings planted during late November to mid-December performed best as the optimal planting time, with semi-hard or hard wood cuttings being more successful than soft wood cuttings. Cuttings with straight upper and slanted lower cuts had the highest survival rate, root initiation and growth due to greater cambial surface exposed for root initiation and less area exposed on upper end to reduce desiccation. Callusing using synthetic auxin applications (IBA or NAA) proved better than traditional callusing to simplify the process, enhance rooting percentage and survival of Rosa centifolia ‘UAF’ cuttings and may be recommended for commercial propagation.
Acknowledgements: Authors are grateful to M. Abdul Salam Khan, graduate student, for technical help during the studies.
REFERENCES
- Abbas, H., M. J. Jaskani, Z. Hussain and M. Asif (2006). Response of rose cuttings against root promoting hormones during spring and autumn. Int. J. Biol. Biotechnol. 3(1): 201-204.
- Ahkami, A. H., M. Melzer, M. R. Ghaffari, S. Pollmann, M. G. Javid, F.M. R Shahinnia, M. J. Hajirezaei and U. Druege (2013). Distribution of indole-3-acetic acid in Petunia hybrida shoot tip cuttings and relationship between auxin transport, carbohydrate metabolism and adventitious root formation. Planta. 238: 499–517.
- Akhtar, G.A., A. Akram, Y. Sajid, R. M. Bilal, M. A. Shahid, H. Sardar, K. Naseem and S. M. Shah (2015). Potential of plant growth regulators on modulating rooting of Rosa centifolia. Am. J. Plant. Sci. 6: 659-665.
- Anderson, R. G. and T. A. Woods (1999). An economic evaluation of single stem cut rose production. Acta Hort. 481:629-34.
- Dahiya, D. S., S. K. Sehrawat., A. K. Godara and B. S. Yadav (2007). Effect of staggered planting on growth, flowering and yield of chrysanthemum cv. Vasantika. Haryana J. Hortic. Sci. 36(1): 78-79.
- Dick, J., R. R. B. Leakey, C. McBeath, F. Harvey, R. I. Smith and C. Woods (2004). Influence of nutrient application rate on the growth and rooting potential of the West African hardwood Triplochiton scleroxylon. Tree Physiol. 24: 35-44.
- Farricielli, C. (2008). The ancient rose lore the romance of the rose. (online available) http://www.rosefarm.com\article_info.php\articles_id\11.
- Gudeva, L. K., F. Trajkova, L. Mihajlov and J. Troick (2017). Influence of different auxins on rooting of rosemary, sage and elderberry. ARRB 12(5): 1-8.
- Hansen, J. and K. Kristiansen (2000) Root formation, bud growth and survival of ornamental shrubs propagated by cuttings on different planting dates. J. Hortic. Sci. Biotechnol. 75(5): 568-574.
- Khan, M. A., K. Ziaf and I. Ahmad (2004). Effect of various hormones and different rootstocks on rose propagation. Pak. J. Biol. Sci. 7(10): 1643-1646.
- Khudhur, S. A. and T. J. Omer (2015). Effect of NAA and IAA on stem cuttings of Dalbergia Sissoo (Roxb). J. Biol. Life Sci. 6(2): 208-220.
- Leakey, R. R. B. (2004). Physiology of vegetative reproduction. In: J. Burley, E. Evans, J. A. Younquist (eds.), Encyclopaedia of Forest Sciences. P. 1655-1668. Academic Press, London, UK.
- Moalemi, N. and M. Chehrazi (2005). The effects of different auxins on rooting of leafy and leafless cutting of Bougainvillea spectabilis in plastic tunnel. The Sci. J. Agric. 27(2): 127-138.
- Nasri, F., A. Fadakar, M. K. Saba and B. Yousefi (2015). Study of indole butyric acid (IBA) effects on cutting rooting improving some of wild genotypes of damask roses (Rosa damascena Mill.). J. Agric. Sci. Belgrade. 60(3): 263-275.
- Okunlola, and A. Ibironke (2013). The effects of cutting types and length on rooting of Duranta repens in the nursery. Global J. Human Soc. Sci. 13(3): 1-4.
- Otiende, M. A. and F. M. Maimba (2020). Endogenous carbohydrate content of the cutting positions at time of severance and IBA concentration influence rooting of Rosa hybrida rootstocks. J. Environ. Agric. Sci. 22(1): 1-9.
- Semangun, H., P. Penyakit and T. Hortikultura (2007). di Indonesia. Edisi ke-2. UGM Press. Yogyakarta. pp. 679.
- Steel, R. G. D., J. H. Torrie and D. A. Dicky (1997). Principles and Procedures of Statistics: A Biometrical Approach, 3rd Ed. McGraw Hill Book Co., New York.
- Wendling, I., P. R. Brooks and S. J. Trueman (2015). Topophysis in Corymbia torelliana × Citriodor seedlings: Adventitious rooting capacity, stem anatomy, and auxin and abscisic acid concentrations. New Forest.46: 107-120.
|





