OVERPRODUCTION OF STREPTOKINASE FROM STREPTOCOCCUS MUTANS EBL-37-UV90 BY PERFORMING OPTIMIZATIONAL STUDIES
G. Akbar1*, M. A. Zia1, A. Jamil1 and F. A. Joyia2
1Department of Biochemistry, University of Agriculture, Faisalabad, Pakistan.
2Centre of Agricultural Biochemistry and Biotechnology (CABB), University of Agriculture, Faisalabad, Pakistan
Corresponding author’s email: ghulamakbardgk@gmail.com
ABSTRACT
Beta-hemolytic Streptococci are being used to produce streptokinase (SK) for decades however, increase in production rate is lucrative. The focus of this study was to enhance the production of SK from mutant derived strain Streptococcus mutants EBL-37-UV90. Fermentation parameters were studied by response surface methodology (RSM) like pH (5-8), fermentation time (12-60 hours), temperature (22-66°C) and inoculum size (1-5) in the presence of carbon source (glucose 1%), yeast extract (3.5%) and corn steep liquor (4%). Model was statistically analyzed by ANOVA using RSM (design expert version 7.0) and coefficient with P≤0.05 was taken as significant. Statistical examination on response surface and contour plot was carried out the effects of single factor and interaction between two factors. Streptokinase was purified by standard methodologies such as ammonium sulphate, salting out process, dialysis, ion exchange and size exclusion chromatography. The maximum streptokinase production was obtained with the mutual interaction of different optimization parameters at 37°C temperature, 36 hours fermentation time, 6.5 pH and 3.0 ml inoculum size. Results indicated that mutant Streptococcus mutans EBL-37-UV90 exhibited the highest production of SK with enzyme activity 275.6 U/mL which is 4.6-fold higher yield than control. Moreover, after purification processes 2.6 folds enzyme activity has been decreased and 75.66 folds specific activity has been increased as compared to crude enzyme. An amount of 38.09% streptokinase was recovered in purified form as compared to crude extract. The present study is a step forward towards cost effective production of streptokinase by virtue of enhanced activity per unit of the purified enzyme which may have important application in treatment of cardiovascular diseases.
Keywords: Mutagenesis, Purification, RSM, Streptokinase, Streptococcus mutans EBL-37-UV90
https://doi.org/10.36899/JAPS.2021.6.0383
Published online March 31, 2021
INTRODUCTION
The process of thrombogenesis leads to obstruction of blood flow that brings about lethal presentations like pulmonary embolism, coronary heart disease and acute cardiac failure. The important remedies with thrombolytic enzymes include streptokinase, urokinase or other agents like tissue plasminogen activator (tPA) (Zia, 2020). The streptokinase as fibrinolytic agent was firstly identified by Tillett and Graner in 1933, while examining fibrinolytic activity in fresh cultures of beta hemolytic Streptococci. There is a range of Streptococcal strains which have ability to produce fibrinolytic enzyme streptokinase. It is a protein consisting of 414 residues of amino acids with 47kDa molecular weight, and has three structural domains: α (alpha) domain comprising of 1 to 150 amino acids residues, β(beta) domain ranging from 151 to 287amino acids residues and γ (gamma)domain consisting of 288 to 414 amino acid residues. The site of SK interaction with plasmin is highly conserved involving α and γ domains which display synergistic influence on plasminogen activation. However, β- domain plays a key role in the synthesis of streptokinase-plasminogen complex which transforms plasminogen into plasmin (Cook et al., 2012). All these three domains also carry antigenic sites in their origin (Akbar et al., 2020). The fibrinolytic activity of this enzyme depends upon its capability to convert plasminogen into its active form which is referred to as plasmin. There are many substrates like casein, fibrin clots, and different types of synthetic esters, upon which plasmin acts to degrade them hydrolytically (Aneja et al., 2013).
The morbidity and mortality rates from myocardial infarction are extremely high around the globe and streptokinase is the major embolus dissolving enzyme being commonly employed as myocardial infarction (MI) therapy in many countries. Almost half million cardiac patients are being fruitfully healed with this fibrinolytic enzyme every year (Aslanabadi et al., 2018). Microbial sources produce small number of enzymes, hence for vigorous enzyme production strain development with medium optimization is needed. For strain improvement many physical and chemical mutation techniques are employed for the over-production of SK exhibiting high quality, better stability and less immunogenic response (Anjum et al., 2013). These ionizing rays penetrate in tissues induce break down of several compounds cause microbial inactivation depending upon the quantity of dose, kind of energy source and quantity of dose absorbed (Mucka et al., 2018). For strain improvement 99.9% killing is the most suitable because few survivor strains in the medium have undertaken many fold mutations resulting in increased metabolite production (Zhou et al., 2005). Response surface technique is an arithmetical tool which makes possible to find the mutual relationship between several response variables and explanatory variables. The key point of RSM application is the use of designed experiments in an order for the optimum production of required metabolite (Jasni et al., 2020). By applying second order polynomial the predicted response was measured (equation 1) that showed interaction with the optimization parameters of experiments.
In the current research work, an attempt was made to produce high yield of fibrinolytic enzyme from Streptococcus mutans EBL-37-UV90. This goal was achieved through random mutagenesis of the Streptococcus equisimilis cells in UV-irradiation treatments and by optimizing medium components. This enhanced production methodology will be helpful as an alternative for commercial production of streptokinase with clinical applications.
MATERIALS AND METHODS
Chemicals: Triton X-100 USA, TCA MERCK Germany, DEAE-cellulose Sigma Aldrich, all these imported chemicals have been used. Carbon sources, nitrogen sources and substrate sources used of GPRTM, Anala® , and Bioworld chemical companies.
Microorganism: Streptococcus mutansEBL-37 was collected from Enzyme Biotechnology Laboratory University of Agriculture Faisalabad, Pakistan, previously identified by API kit and was re-cultured on nutrient agar plates kept at 37⁰C incubator for 24 hours. These cultured colonies expressed hemolysis on blood agar medium having 10% (v/v) defibrinated sheep blood.
Mutagenesis: The process of mutation was performed with ultraviolet rays to wild type Streptococcus mutans EBL37 by adjusting various time interval on petri plate at a fixed distance from light source. UV light exposed mutant colonies were re-cultured to check streptokinase production efficiency. This method was also applied by Devi et al., (2013).
Plate screening method: The selection of the best colonies was carried out by culturing on nutrient agar medium containing 1% triton X-100. The measurement of colonies length was done for 1-2 days at incubation temperature 37°C and the largest actively growing mutant colonies were selected for further studies. The kill curve was formulated between colony forming units (CFU) against various time interval’s UV-mutants. Mutant colonies with survival rate of less than 1%were taken and screened on the basis of fibrinolytic assays. This method was also applied by (El-Mongy et al., 2012; Naeem et al., 2018).
Streptokinase production: Mutant derived Streptococcus strain was added in 25 ml phosphate buffer pH 7.5. This suspension was placed on the orbital shaker (120rpm and at 37°C) for 24hours to prepare inoculum. Fermentation medium comprises glucose, corn steep liquor (CSL), yeast extract, pH (6-8), inoculum (5ml) added in this media. All these parameters were studied by applying RSM to observe maximum production of SK. This procedure was also applied by (Taiwo et al., 2018).
Blood clot lysis assay: In this assay 5 ml fresh blood was added in pre-weighed tubes separately. After clot formation tubes were again weighted and different concentration of streptokinase extract were added in all tubes except control where distilled water was added instead of streptokinase. Tubes were placed at 37 °C for 18 hours. After that liquid portion in each tube was removed carefully and again weighted. So, the difference in weight of blood clot before and after lysis was noted and enzymatic activity was estimated by calculating percentage clot lysis.
Casein digestion assay: In this assay test solution including 2ml of 4% casein was added which followed by the addition of 0.1ml of cold streptokinase. This solution was kept at 37°C upto 30 min and 2ml of TCA wad added to stop the reaction. Absorbance in spectrophotometer was measured at 280nm (Keramati et al., 2013). One unit of streptokinase is equivalent to micromole of tyrosine released per milliliter per second. Biuret method was used to determine protein content before extraction (Liu et al., 2017). This current research work followed this procedure.

   Units/ml enzyme = Units/ml enzyme =
Where
ΔA280nm/min Test = Change in absorbance of test solution
ΔA280nm/min Blank= Change in absorbance of test solution
B = Total volume of reaction mixture
Extinction coefficient = 8.25 (mg/ml)-1cm-1
Dilution factor = d. f
C = Volume of enzyme
Enzyme purification and characterization: Crude fermented broth of SK was subjected to a process called salt precipitation. The saturation of crude SK extract was met by mixing 40% ammonium sulfate salt in the process of salting in and 60% in salting out and sample was desalted by dialysis procedure. Chromatographic column was prepared using diethyl amino ethyl (DEAE) cellulose for purification in ion exchange chromatography. Fifty fractions for both wild and mutant produced SK were collected and their specific activities with protein contents were measured. Active fractions were pooled and subjected to size exclusion chromatography for further refinement. The collected enzyme fractions were taken from mutant and wild type strains. S. mutans were analyzed with their protein estimation and enzyme assays (Erickson et al., 2009; Karimi et al., 2011). This current research work followed this method. The Michaelis constants Km and Vmax for streptokinase were computed by following Khudir et al., (2019).
Statistical analysis and model validation
The computation of predicted response was carried out using second order polynomial equation (Eq. 1) which involved all the interacting terms of the experiment as given below.
Y = β0 + β1X1 + β1X2 + β3X3 + β11X21 + β22X22 + β33X23 + β44X24 + β12X1X 2 + β13X1X3 + β14X1X4 + β23X2X3 + β24X 2X4 + β34X34 + ε ------------------------- (Eq. 1)
Where
X1, X2 and, X3 = Independent variables
β1, β2, β3 = Linear terms coefficients
β11, β22, β33, β44 = Coefficient of polynomials for quadratic terms
β12, β13, β14, β23, β24, β34 = Coefficients of polynomials for combine effect of variables
In this research work four parameters (pH, temperature, incubation period and inoculum size) were optimized through RSM following screening of run orders. The statistical significance of the data obtained from experiments was analyzed using analysis of variance (ANOVA). The interactive effect of the four process variables was analyzed by three-dimensional surface plots to eventually determine the optimum levels of these parameters for enhanced SK production (Vellanki etal., 2013). The fitness of the regression model obtained was given by the coefficient of determination, i.e., R2 following Bhardwaj et al., (2017). The probability level P≤0.05 was assumed as significant for model terms and probability values >0.1000 indicates non-significant model terms.
RESULTS
Streptococcus mutans EBL-37 cultured broth medium was placed in ultraviolet rays chamber for random mutagenesis to obtain increased production of streptokinase (SK). Inoculum (1ml) was poured into 10 tubes and exposed to UV-irradiations (254nm) at various time interval of 0 to 90min (UV0, UV10, UV20, UV30, UV40, UV50, UV60, UV70, UV80 and UV90) with a distance of 8.0 cm from fixed UV source. A kill curve was derived for all these doses of UV-exposure representing survival percentage of bacterial colonies along with exposure time shown in Fig.1. Mutant colonies of Streptococcus mutans EBL-37-UV90 which expressed survival rate of 0.1% exhibited the largest clear zones than other mutants and wild type colonies.

Fig. 1. Kill curve for Mutant colonies Streptococcus mutansEBL-37
Optimization parameters: By optimizing nutritional parameters such as carbon, nitrogen and substrate sources were evaluated to get high production of streptokinase. In this study these parameters were studied for SK production from mutant S.mutansEBL-37-UV90. Among carbon sources the maximum production of 270.0UmL⁻¹ SK was obtained using 0.6% glucose with 7.0 pH at 37°C, 1ml inoculum size after 36hrs of incubation. Enzyme production was not significantly supported using starch and lactose while fructose exhibited SK production was 217.0UmL⁻¹ with 0.6% concentration (Fig. 2.A).The degradation of a suitable substrate by microorganisms support the production of a specific metabolic product inside or outside the microbial cell. In present study different substrates were used for SK production in which 0.4% corn steep liquor expressed maximum production with 280.0UmL⁻¹ by using 1.5ml inoculum with 7.0 pH at 37°C after 36hrs incubation time. Among other substrates molasses expressed higher amount of streptokinase as compared to sugarcane bagasse and rice polishing as shown in (Fig. 2.B). However, in case of nitrogen source optimum production was obtained with 0.6% peptone with 255.0UmL⁻¹ enzyme activity by using 1.0 ml inoculum with 7.5 pH at 37°C after 36hrs as compared to other nitrogen sources (Fig. 2.C).
Fig.2. (A) Evaluation of carbon sources for the optimum production of SK from mutagenized S. mutansEBL-37-UV90, (B) Nitrogen sources estimation for the optimum production of SK (C) substrates evaluation for the production of SK from mutagenized S. mutans EBL-37-UV90.
The ranges of different fermentation parameters which were studied via RSM are given in Table1. RSM-based statistical analysis was applied on four parameters which provided combinations of 30 run orders with predicted values. These statistically predicted and experimentally observed values were found very close to each other for streptokinase (SK) production. Significance of each coefficient was determined by comparing with P-values given in Table 2.
Table 1. Experimental ranges of different factors required for optimization
|
Ser. No.
|
Factors
|
Low range
|
Middle range
|
High range
|
|
1.
|
A-pH
|
5
|
6.5
|
8
|
|
2.
|
B-Incubation period
|
12
|
36
|
60
|
|
3
|
C-Temperature
|
22
|
44
|
66
|
|
4
|
D-Inoculum size
|
1
|
3
|
5
|
Polynomial model
Second order polynomial model equation 1 is fitted in the central composite design’s (CCD) optimization parameters obtaining equation 2.
R1-Enzyme activity (U/ml) = - 30.42134 +78.05812A - 9.28398B +7.40613C +16.81296D + 0.75660AB - 0.57871AC - 4.33868AD + 0.044200BC + 0.73729BD - 0.4034CD - 4.16473A2 + 2.80562B2 - 0.053113C2 + 0.43056D2_______________-(2)
Where
A = pH
B = Incubation period
C = Temperature
D =Inoculum size
Analysis of variance: The interactions of these independent variables of 2nd order polynomial were observed by CCD to check the model significance and lack of fit. Using many indicators, the adequacy of fitted model was assessed and their inferences were given in Table 2.
Table 2. Analysis of variance for response surface reduced quadratic model [partial sum of square-type iii]
|
Source
|
Sum of Squares
|
df
|
Mean Square
|
F Value
|
p-value
Prob > F
|
|
Model
A-pH
B-incubation period
C-Temperature
D-Inoculum size
AB
AC
AD
BC
BD
CD
A2
B2
C2
D2
Residual
Lack of Fit
Pure Error
Cor Total
|
41968.63
2044.80
0.18
817.78
0.18
3077.85
7505.58
106.10
9881.37
2904.80
811.24
1257.69
49.11
10938.52
61.28
363.31
115.04
248.27
42331.94
|
14
1
1
1
1
1
1
1
1
1
1
1
1
1
1
6
2
4
20
|
2997.76
2044.80
0.18
817.78
0.18
3077.85
7505.58
106.10
9881.37
2904.80
811.24
1257.69
49.11
10938.52
61.28
60.55
57.52
62.07
|
49.51
33.77
2.973
13.51
2.973
50.83
123.95
1.75
163.19
47.97
13.40
20.77
0.81
180.65
1.01
0.93
|
0.0001*
0.0011*
0.9583ns
0.0104*
0.9583 ns
0.0004*
0.0001*
0.2338 ns
0.0001*
0.0004*
0.0106*
0.0039*
0.4025 ns
0.0001*
0.3533 ns
0.4670 ns
|
Note: ‘*’ for significant and ‘ns’ for non-significant. Std. Dev. = 7.78, C.V. %= 3.12, R-Squared=0.9914, Adj. R-Squared=0.9714, Pred. R-Squared =0.7888
Analysis of variance represented for different treatments relative variations and random error variations. The variation in predicted R2 (0.7888) and adjusted R2 (0.9714) are less than 0.2 which indicate that the model is suitable. However, the low value of coefficient of variation (C.V) means conducted experiments are precise and reliable. If p-values are below 0.05 then model terms are significant and the model terms are non-significant for the values which are greater than 0.1. In this case A, C, AB, AC, BC, BD, CD, A2, C2 are significant model terms. All the interaction terms are significant except AD (pH vs Inoculum size). Therefore, present model is highly significant which representing < 0.0001 p-value for model reproducibility. The lack of fit must be non-significant so that model should be best fit upon undergone research parameters level. In this model lack of fit (0.4670) is non-significant which is indicating a good fit of the present model upon the experimental factor’s main effect, their cross interactions and self-interactions between different factor levels (pH, temperature, incubation period, inoculum size).
Predicted versus actual plot:
The graphical representation of predicted values versus actual values showing close resemblance to each other for the production of SK from Streptococcus mutans EBL-37-UV90 while normal plot of residuals is showing close proximity between normal % probability and internal studentized residuals. So, the statistical model applied on present research parameters was good fit for the production of streptokinase. The maximum SK activity for crude extract was 275.6 U/ml and at final stage after gel filtration enzyme activity was 105.0 U/ml. Protein contents obtained for these steps were 13.12 and 0.06 mg/ml correspondingly (Fig. 3.A). Plot of residual representing internally studentized residual and normal % probability (Fig. 3B) Comparison between predicted versus actual values of SK production from Streptococcus mutans EBL-37-UV.
Fig.3. (A). Plot of residual representing internally studentized residual and normal % probability (B) Comparison between predicted versus actual values of SK production from Streptococcus mutans EBL-37-UV90
Interaction between factors: The 2D contours and 3D response surface graphs representing that model is significant on designed optimization parameters. The (2D) contour plots are used to express the main effect of variables and 3D response surface graphs are used for the mutual interaction of experimental parameters required for optimization. The 2D contour plots may be elliptical, circular, rising or saddle nature to represent results interpretation. Among them elliptical and saddle ridges of contour plots represent significant results and circular lines in contour plots indicate non-significant results (Fig.4). A significant relationship was found in experiment between pH and incubation period, temperature and incubation period, incubation period and inoculum size, inoculum size and temperature for the production of SK enzyme, which are similar to the results predicted by ANOVA and 3D response surface graph. Combine pH and temperature influence on SK yield found significant in the presence of glucose (1%), corn steep liquor (4%) and yeast extract (3.5%). There was also significant interaction between pH and inoculum size for the optimum production of streptokinase that was also shown in response surface (Fig.5).

Fig.4. (A) 2-D contour plot representing pH and incubation period impact on SK production from S. mutans EBL-37-UV90 with (B) Contour plot is representing interaction of pH along with temperature on SK secretion(C) Plot is representing inoculum size with pH combination influence on enzyme manufacturing (D) Contour plot is representing main effect of temperature vs incubation period on enzyme production (E) Contour plot is representing main effect of inoculum size vs incubation period on enzyme production(F) Contour plot is representing main impact of temperature along with inoculum size on SK synthesis.
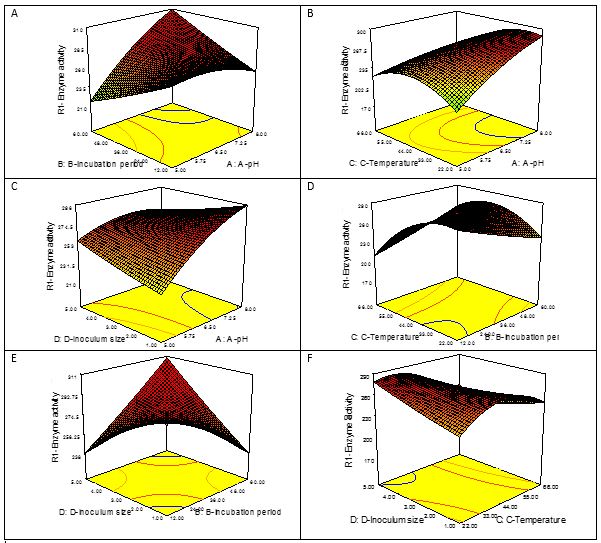
Fig. 5.(A) 3D Response surface graph is exhibiting mutual interaction of pH and incubation period on SK production from StreptococcusmutansEBL-37-UV90 (B) Response surface graph is displaying mutual interaction of pH and temperature on enzyme production (C) Response surface graph is presenting mutual interaction of pH and inoculum size on SK production (D) 3D graph is unveiling mutual interaction of temperature and incubation period on SK production (E) 3D graph is representing mutual interaction of inoculum size and incubation period on SK production (F) Response surface graph is representing mutual interaction of inoculum size and temperature on SK production.
Enzyme purification and characterization: After ion exchange chromatography absorbance (at OD 280nm) and enzyme activity were measured for the collected 100 fractions and its summary is graphically represented in Fig. 6. The same procedure was performed in gel filtration chromatography by taking absorbance (at OD 280nm) and analyzing enzyme activity which are graphically shown in Fig. 7.
In enzyme purification enzyme activity, protein contents and percentage recovery have been decreased after each successive step of purification from 275.6 to 105.5 U/ml, 13.12 to 0.06 mg/ml and 100 to 38.09% respectively. However, specific activity and purification have been increased after each respective step of purification from 21.0 to 1589.0 U/mg and 0.0 to 75.66 % respectively and its complete summary is also given in the following Table 3.

Fig. 6.Partial purification profile of SK via ion exchange column (DEAE-celluose) fractions elution

Fig. 7.Partial purification summary of SK via gel filtration chromatography using sephadex G-100
Table 3: Summary of streptokinase purification produced from mutant derived Streptococcus mutans EBL-37-UV90
|
Purification Stages
|
Total Volume
(ml)
|
Enzyme Activity
(U/ml)
|
Enzyme
(Units)
|
Specific Activity
(U/mg)
|
Protein contents
(mg/ml)
|
Purification
(fold)
|
Recovery
(%)
|
|
1. Crude Extract
|
200
|
275.6
|
55,120
|
21.0
|
13.12
|
0.0
|
100
|
|
2. Dialysis
|
100
|
135.0
|
13,500
|
73.0
|
1.85
|
3.47
|
48.98
|
|
3. Ion exchange
|
60
|
119.0
|
7,140
|
298.0
|
0.40
|
14.19
|
43.17
|
|
4. Gel filtration
|
40
|
105.0
|
4,200
|
1589.0
|
0.06
|
75.66
|
38.09
|
Estimation of kinetic parameters Km and Vmax: Michaelis-Meten constant ‘Km’ and maximum velocity ‘Vmax’ of partially refined streptokinase were calculated by using various concentration of casein (substrate). The graph was drawn between initial velocities versus substrate concentrations, and Vmax and Km were determined having values 3.02 µM/min and 0.3 µM respectively (Fig.8). The total enzyme concentration [Et ] used was 4.0 µM attaining Kcat value(1.26 ×10-2S-1) along with the catalytic efficiency (Ec) 4.2 ×10-2 M-1.S-1.
Effect of pH on enzyme activity: In current research work enzyme activity was measured for 3-10 ranges of pH values at the same substrate concentration where 7.0 pH expressed the higher activity over others graphically shown in Fig. 9. Beyond this limit enzymatic activity was decreased largely.

Fig. 9. Effect of pH on SK activity at various pH levels
Effect of temperature on enzyme activity: The incompletely purified streptokinase remained active over a wide range of temperature 20-90°C by using casein at 7.0 pH. The maximum activity was observed at 40°C and above this range enzyme activity was began to decrease as shown in Fig.10.

Fig. 10. Effect of temperature on SK activity over a range of temperature (20-90)
DISCUSSION
There are many techniques through which random mutations are made in microorganisms. Such techniques include gamma-rays, X-rays, and ultraviolet radiations, and chemicals such as ethyl methane sulfonate, NTG, and ethidium bromide (Et-Br) (Iftikhar et al., 2010). It has been described that gamma and ultra violet rays have more mutational effect than chemicals such as ethyl methane sulfonate (EMS) due to high piercing efficiency (Dhulgande et al., 2011). Similarly in a research investigation Lactobacillus amylovorus was subjected to 0, 0.2, 0.4, 0.6, 0.8, and 1.0 kGy gamma doses and was found that higher doses decreased the viable number of L. amylovorus per milliliter (Neysens et al., 2003).
The evaluation of strains for increased streptokinase (SK) production was performed by screening with enzyme assays in which UVM6 mutant derived strain expressed the maximum activity. UV-irradiations have been found to decrease viable growth of Streptococci strains and its correct dosage is necessary to get a useful mutant. After applying UV-irradiation for strain improvement in enhanced streptokinase production was observed and in this regards mutant derived Streptococcus equisimilis expressed maximum activity (Doss et al., 2011).
Nutritional sources are necessary for the enhanced production of streptokinase. Naeem et al. (2018) and Arshad et al. (2019) evaluated work on enhanced production of streptokinase from Streptococcus equisimilis by optimizing carbon sources, nitrogen sources and substrate sources to study enzyme production. They observed glucose, peptone and corn steep liquor as optimum producers among estimated carbon sources, nitrogen sources and substrate sources respectively. Glucose recognized as the best carbon source evaluated from earlier studies for the production of SK from Streptococcus species. In another investigation Zia et al., (2013) obtained highest growth and SK yield by using 0.3% corn steep liquor as substrate in the fermentation medium. In a study 0.6% glucose, 0.2% substrate (corn steep liquor) and 0.6 % peptone expressed maximum growth and SK production from Streptococcus agalactiaeEBL-32 (Arshad et al., 2019).
In several investigations microorganisms underwent strain development process to get better yield of thrombolytic and other industrial and pharmaceutical enzymes (Khudir et al., 2019; Alinodehi et al., 2019). Faran et al. (2015) obtained hyper yield of thrombolytic enzyme in mutant Streptococcal species with 335U/ml enzyme activity and 1116.66 unit/mg specific activity (Faran et al., 2015). Felsia et al. (2011) in a research work found 6.7 IU/mg enzyme activity and 1.21mg protein content in crude enzyme, and after dialysis they observed 76 IU/mg enzyme activity and 0.77mg protein contents with 44.2% recovery. Separation through ion exchange chromatography was performed on this dialyzed sample using DEAE-Sepharose column and obtained 31.10 units enzyme activity with protein components 18.70 mg (Hua et al., 2008).In an investigation, seven doses of gamma radiations were reported for mutagenesis of S. equisimilis cell suspension. The doses were 20, 40, 60, 80, 100, 120, 140 K Rad (unit of absorbed radiation dose) and optimal mutant gamma dose was 120 K Rad formulated by 3 log kill curve (Weigel et al., 2012). Moreover, in another study, Streptococcus equisimilis was isolated and subjected to random mutagenesis by UV-rays for strain improvement.
In a research finding S. equisimilis T3 mutant strain expressed 120% higher enzyme yield with 821 U/ml than wild S. equisimilis while other UV mutant S. equisimilis UV10 also exhibited higher SK production of 0.3U/ml (Abdelghani et al., 2005). In a recent attempt EMS (ethyl methanesulfonate) mutant S. equisimilis SK-6 produced streptokinase with enzyme activity 168.46 UmL-1 which was 3.2 fold increase in production rather than unoptimized medium (Arshad et al., 2019). In other research work enhanced production of Streptokinase was achieved by using UV-mutant Streptococcus equisimilis and EtBr mutant S. equisimilis where UV- mutant was detected for the higher production of Streptokinase 400% as compared to EtBr-mutant 146% more production respectively (Naeem et al., 2018).
The key point of RSM application is the use of designed experiments in an order for the optimum production of required metabolite. The response surface graphs help to predict the enzyme production for various levels of testing factors and its contour plots help to identify the kind of interaction between variables. Contour plot curves indicate unlimited combinations of two factors while keeping other variable constant at zero level. Circular lines of contour plot represent a non-significant interaction among different variables. On the other hand, elliptical lines of contour plots indicated significant interaction between variables. 3D graphs and 2D contour plot are the graphical representation to manipulate the interaction between variables and to minimize the optimal conditions (Oberoi et al., 2014; Jasni et al., 2020).
Faran et al., (2015) used the same methods for the purification of streptokinase and obtained 335.0 U/mL enzymatic activity, 1116.66 U/mg specific activity and 0.30 mg/mL protein contents 41.92 fold purification and 69.79% recovery. While gamma mutant strain showed 300 U/mL activity, 1428.57 Umg-1 specific activity, 0.21 mgmL-1 protein, 59.52 fold purification and 75.94% recovery which are resembling to current research findings. Khudir et al., (2019) purified the protease enzyme from Staphylococci sciuri and obtained 25.49 fold purification, 235.1 UmL-1 enzyme activity, 982.22 Umg-1 specific activity and 3.76% enzyme was recovered. Alinodehi et al., (2019) also applied chromatographic technique for streptokinase purification and obtained 98% purified enzyme after cloning, expression and purification processes. These findings are also close relevant to my findings.
By following Michaelis-Menten equation rate of reaction along with Km and Vmax were measured in several research investigations (Khudir et al., 2019: Arshad et al., 2019). El-mongy and Taha (2012) observed optimum SK activity (91.6 U/mL) at pH 7.5 which are closely related to the current findings. Dubey et al. (2011) and Abdulghani et al. (2005) examined the highest activity of bacterial origin streptokinase at temperature 37°C that is closely associated with current findings.
Conclusion: It was concluded that UV rays have good efficacy for the mutagenesis of industrially important microbes to develop specific desired products. In this research work the most incredible achievement is the boost production of streptokinase from highly potent perfect mutant Streptococcus mutansEBL-37-UV90. The evaluated and optimized culture conditions with the help of response surface technique are also strongly supported for this higher level production of streptokinase. This strain can be used as alternative source for the large scale cost effective streptokinase production. Hence, in future mutant derived or genome edited expression of Streptococcus genes by virtue of increased SK production and reduced immunogenicity need to be used in laboratory scale for the evaluation at industrial scale. That’s the way to save the lives of millions of cardiac patients throughout the world.
Acknowledgement: The authors gratefully acknowledge HEC for providing financial support to carry out this research work under HEC project [NRPU project# 4921].
Disclosure of conflict of interest: The authors declared that there is no conflict of interest REFERENCES
- Abdelghani, T.T.A., A. Kunamneni and P. Ellaiah (2005). Isolation and mutagenesis of streptokinase producing bacteria. Am. J. Immun. 1(4), 125-129
- Akbar, G., M.A. Zia, A. Ahmad, N. Arooj, and S. Nusrat. (2020). Review on streptokinase with its antigenic determinants and perspectives to develop its recombinant enzyme with minimum immunogenicity. J. Inn.Sci. 6(1): 17-23.
- Alinodehi, N.N, S. Sadeh. F.K. Nezamiha, M. Keramti, M. Hasaqnzadeh and R.A. Mianroodi (2019). Evaluation of activity kinetic parameters of Sk319cys, as a new cysteine variant of streptokinase: A comparative study. Cur. Pharm. Biotech. 20: 1-3.
- Aneja, R., M. Datt, S.Yadav and G. Sahni (2013). Multiple exosites distributed across the three domains of streptokinase co-operate to generate high catalytic rates in the streptokinase-plasmin activator complex. Biochem. 52(49): 8957-8968.
- Arshad, A., M.A. Zia, M. Asgher and F.A. Joyia (2019). Enhanced production of streptokinase by chemical mutagenesis of Streptococcus agalactiae EBL-20 by response surface methodology. Braz. Arch. Bio. Tech. 62.
- Aslanabadi, N., N. Safaie, F. Talebi, S. Dousti and T.E. Maleki (2018). The streptokinase therapy complications and its associated risk factors in patients with acute ST elevation myocardial infarction. Iran. J. Pharm. Reas. 17: 53-63.
- Bhardwaj S, S. Bhattacharya, A. Das, M. Palaniswamy and J. Angayarkanni (2017). Application of multifactorial experimental design for optimization of streptokinase production using Streptococcus equisimilis SK-6. Arab. J. Sci. Eng. 42: 2273-2277.
- Cook, S.M., A. Skora, C.M. Gillen, M.J. Walker and J.D. McArthur (2012). Streptokinase variants from Streptococcus pyogenes isolates display altered plasminogen activation characteristics–implications for pathogenesis. Mol. Micro. 86(5): 1052-1062.
- Devi, C.S., V. Mohanasrinivasan, B. Vaishnavi, E. Selvarajan and S.J. Naine. (2013). Optimization studies for enhanced production of streptokinase by Streptococcus equisimilisUVM6. J. P. & Appl. Micro. 7: 1-5.
- Dhulgande, G.S., D.A. Dhale, G.L. Pachkore and R.A. Satpute (2011). Mutagenic effectiveness and efficiency of gamma rays and ethyl methane sulphonate in pea Pisumsativum L.). J. Exp. Sci. 2(3): 7-8.
- Doss, M., M. Manohar and N.A. Singh (2011). Studies on isolation, screening and strain improvement of streptokinase producing β-hemolytic Streptococci. World. 1(3): 07-11.
- Dubey, R., J. Kumar, D. Agrawala, T. Char and P. Pusp (2011). Isolation, production, purification, assay and characterization of fibrinolytic enzymes (Nattokinase, Streptokinase and Urokinase) from bacterial sources. Afr. J. Biotech. 10: 1408-1420.
- El-Mongy, M.A. and T.M. Taha (2012). In vitro detection and optimization of streptokinase production by two Streptococcal strains in a relatively low cost growth medium. Intern. Res. J. Microb. 3(4): 153-163.
- Erickson, H.P. (2009). Size and shape of protein molecules at the nanometer level determined by sedimentation, gel filtration, and electron microscopy. Biological procedures online. 11(1): 32.
- Faran, G., M.A. Zia, M. Shahid and S. Abdullah (2015). Improved streptokinase
production; U.V-irradiation of Streptococcus equisimilis. Prof. Med J. 22: 656-663.
- Felsia, F.X., R.V. Kummar and S. Kalpana (2011). Production and partial purification of streptokinase from Streptocccus pyogenes. J. Biochem. Tech. 3: 289-291.
- Hua, Y., B. Jiang and W. Mu (2008). Purification and Characterization of a Novel Fibrinolytic Enzyme from Bacillus sp. nov. SK006 Isolated from an Asian Traditional Fermented Shrimp Paste. J. Agric. Food. Chem. 56: 1451-1457.
- Iftikhar, T., M. Niaz, S.Q. Abbas, M.A. Zia, I. Ashraf and K.J. Lee (2010). Mutation induced enhanced biosynthesis of lipases by Rhizopus oligosporus var. microsporus. Pak. J. Bot. 42(2): 1235-49.
- Jasni, A.B., H. Kamyab, S. Chelliapan, N. Arumugam, S. Krishnan and M.F.M. Din (2020). Treatment of Wastewater Using Response Surface Methodology: A Brief Review. Chem. Eng. Trans. 78: 535-540.
- Karimi, Z., M. Babashamsi, E. Asgarani, M. Niakan and A. Salimi (2011). Fermentation, fractionation and purification of streptokinase by chemical reduction method. Iran. J. Micro. 3(1): 42.
- Keramati, M., F. Roohvand, M.M. Aslani, S. Khatami, M. Aghasadeghi, M. Sadat and F. Motevalli (2013). Screening, Cloning and Expression of Active Streptokinase from an Iranian Isolate of S. equisimilis Group C in E. coli. Iran. J. B. Med. Sci. 16(4): 620.
- Khudir, R.A, M.M. Salem, N.G. Allam and E.M.M. Ali (2019).Production, partial purification and biochemical characterization of a thermos-tolerant alkaline metallo-protease from Staphylococci sciuri. Appl. Biochem. Biotech.189: 87-102.
- Liu, F., Z. Chen, J. Shao, C. Wang and C. Zhan (2017). Effect of fermentation on the peptide content, phenolics and antioxidant activity of defatted wheat germ. F. Bios. 20: 141-148.
- Mucka, V., J. Cervenak, D. Reimitz, V. Cuba, P. Blaha and B. Neuzilova (2018). Effect of irradiation conditions on the radiation sensitivity of microorganisms in the presence of OH-radical scavengers. Int. J. Rad. Bio. 94(12): 1142-55
- Naeem, M., B. Sadia, F.S. Awan and M.A. Zia (2018). Enhanced Production of Streptokinase by UV-and Ethidium Bromide-Treated Streptococus equisimilis Mutant. Pak. J. Zool. 50(2).
- Neysens, P., W. Messens, D. Gevers, J. Swings and L. De Vuyst (2003). Biphasic kinetics of growth and bacteriocin production with Lactobacillus amylovorus DCE 471 occur under stress conditions. Micro. 149(4): 1073-82.
- Oberoi, H.S., R. Rawat and B.S. Chadha (2014). Response surface optimization for enhanced production of cellulases with improved functional characteristics by newly isolated Aspergillus niger HN-2. Ant. Van. L. 105(1): 119-34.
- Taiwo, A.E., T.N. Madzimbamuto and T.V. Ojumu (2018). Optimization of corn steep liquor dosage and other fermentation parameters for ethanol production by Saccharomyces cerevisiae type 1 and Anchor Instant yeast. E. 11(7): 1740.
- Tillet, W.S. and R.L. Garner (1933). The fibrinolinolytic activity of hemolytic Streptococci fibrinolysis J. Immun. 42: 109-13.
- Vellanki RN, R. Potumarthi, K.K. Doddapaneni, N. Anubrolu and L.N. Mangamoori (2013). Constitutive optimized production of Streptokinase in Saccharomyces cerivisiae utilizing glyceraldehydes-3-phosphate dehydrogenase promoter of Pichia pastoris. Biomed. Res. Int. 158: 25-40.
- Weigel, P.H. and B.A. Baggenstoss (2012). Hyaluronan synthase polymerizing activity and control of product size are discrete enzyme functions that can be uncoupled by mutagenesis of conserved cysteines. Gl. 22(10):1302-10.
- Zhou, X., X. He, J. Liang, A. Li, T. Xu and T. Kieser (2005). A novel DNA modification by sulphur. Mol. Micro. 57(5): 1428-38.
- Zia, M.A., R. Faisal, R.Z. Abbas, G. Faran, M.K. Saleemi and J.A. Khan (2013). Comparison of Streptokinase activity from Streptococcus mutans using different substrates. Pak. Vet. J. 33(1): 77-79.
- Zia M.A. (2020). Streptokinase: An efficient enzyme in Cardiac Medicine. Protein Peptide Lett. 27(2): 111-119
|





