EFFECTS OF ALMOND MEAL (TERMINALIA CATAPPA) BASED DIETS ON NUTRIENT UTILIZATION, GROWTH AND HEMATOLOGY OF LABEO ROHITA FINGERLINGS
S. M. Hussain*, S. Nisar, M. Jamil, F. Bashir, M. Z. ul H. Arslan, S. Tabassum, B. Ahmad, S. Ahsan and A. Sharif
1 Fish Nutrition Lab, Department of Zoology, Government College University, Faisalabad, Pakistan
Corresponding author’s email:drmakhdoomhussain@gcuf.edu.pk
ABSTRACT
Nutrient utilization, hematological indices and growth performance of Labeo rohita fingerlings were examined while substituting fish meal with almond meal-based diet. Five diets were prepared by substituting different levels such as 0%, 20%, 40%, 60%, 80% and 100% of fish meal with almond meal in diets. Test diets were allocated to each fish group by using completely randomized design (CRD). In triplicate tanks, fifteen L. rohita fingerlings were kept and fed at 5% of their body weight. Growth indices such as weight gain (%), feed conversion ratio (FCR) and standard growth rate (SGR) were evaluated using standard formulae. Nutrient utilization was determined by proximate analysis of feces. There was significant difference (p≤0.05) among hematological indices and growth performance of fingerlings fed 40% almond meal-based diet and control diet. By increasing almond meal in diets more than 40%, the hemoglobin and RBCs of fish indicated a significant (p≤0.05) inverse correlation. The outcomes of present study revealed that replacement of fish meal by almond meal in fish feed is cost effective with significant improvement in growth response, nutrient digestibility and hematology.
Key words: Replacement, growth rate, hemoglobin, nutrient utilization, plant proteins.
https://doi.org/10.36899/JAPS.2021.6.0387
Published online March 31, 2021
INTRODUCTION
Farming of fish and other aquatic organisms is the only feasible choice to increase the protein production. Aquaculture production has increased about 12 folds over the last three decades and is now a good source of high-quality protein. To prepare fish feed; fishmeal is an outstanding source of protein due to its excellent composition of essential amino acids, fatty acids and other nutrients. It is highly digestible and palatable (Olsen and Hasan, 2012). On the other hand, due to high prices, unstable supply and increase in demand of the fish meal, it is the need of time to search alternate plant protein sources (Lech and Reigh, 2012; Lazzarotto et al., 2018). Plant feed stuffs are best source of proteins to make environment friendly and economical fish feed (Hussain etal., 2015). The use of almond meal in fish diets has been studied by few fish nutritionists. Almond kernel is covered by brown skin and consists of an intermediate shell with outer hull. Almond meal is prepared from whole almond including skin. Almond meal is a rich source of vitamin E and contains unique antioxidant properties that improve the immune system. The almond kernel contains crude protein (24.5%), ether extract (36%) and ash (6%). This makes the almond kernel meal an effective substituent with fish meal (Elezuo, 2016). By products of almond industry are commonly used for livestock feed. Almond comprises 33% crude protein (Akpabio, 2011) while 40% crude protein is reported by (Falaye et al., 2016).
Almond meal is gaining popularity among fish culturists as a favorable feed ingredient due to its rich phenolic contents (Annegowda et al., 2010). To monitor the health condition of fish, hematology is attaining importance in fish culture. Sometimes, there is a problem of feed toxicity so in order to monitor the toxicity, hematological components of blood are valuable. Hematological parameters in a number of fish species have been studied for establishing normal value range (Arsalan et al., 2016). Digestibility study is very important to determine the nutrient digestibility, evaluating the efficiency of animal feedstuffs and nutrients used in feed formulation. Digestibility studies provide us information about diet preparation and it also provides the right estimate for fish growth (Falaye et al., 2016).
Plant based proteins are ideal and probably will continue to be the main substitute of fishmeal in aquaculture diets (Olsen and Hasan, 2012). Available literature has revealed that T. catappa (TC) is under investigation as a possible substitution for fish meal in diet of various fish species, however a lesser amount of literature is available on the almond meal-based diets for Indian major carp i.e., Catla catla, L. rohita and Cirrhinus mrigala. In Pakistan, L. rohita is very important major carp and is cultured on large scale. It is generally grown in polyculture. Unfortunately, no economical feed is available at local level to the fish farmers. However, to further increase the fish production, various crude preparations are being used in the form of mesh (Hussain et al., 2018). To get maximum production in short period of time artificial feed must be provided in order to increase the production rate and gain maximum weight. Therefore, present research study is aimed to find out the efficacy of almond meal as an alternative protein source for commercially important species i.e., L. rohita to improve the growth performance, nutrient utilization and hematological parameters.
MATERIALS AND METHODS
The present research study was conducted in June 2017 to May 2018 at Fish Nutrition Laboratory, Department of Zoology, Government College University, Faisalabad.
Experimental conditions: For the experimental purpose, L. rohita fingerlings were obtained from Government Fish Seed Hatchery, Faisalabad. The fingerlings were acclimatized to experimental conditions in V-shaped tanks. These tanks having 70 L water capacity are particularly designed to collect fecal material. The fingerlings were given NaCl (5g L-1) bath to prevent fungal infection and ectoparasites attack. Two weeks’ fingerlings were fed basal diet once every day (Allan and Rowland, 1992). Measurement of water quality indices such as dissolved oxygen, pH and temperature was done on daily basis. pH meter (Jenway 3510) and DO meter (Jenway 970) were subsequently used to monitor pH and dissolved oxygen. Throughout experimental period, air pumps were used to provide aeration to tanks via the capillary system (Rowland and Ingram, 1991).
Preparation of Experimental Diets and Experimental Design: The feed ingredients were purchased from market and analyzed to check the chemical composition by following AOAC (1995), (Table 1). Chromic oxide (1%) was also added as an inert marker. Experimental diets were divided into one control and five test diets. Almond meal was used for the experimental diet formulation by substituting fishmeal at levels of 0%, 20%, 40%, 60%, 80% and 100%, respectively (Table 1).
Table 1. Ingredients composition (%) of test diets
|
Ingredients
|
Test Diet I (Control)
|
Test Diet II
|
Test Diet III
|
Test Diet IV
|
Test Diet V
|
Test Diet VI
|
|
Fishmeal
|
100
|
80
|
60
|
40
|
20
|
0
|
|
Almond meal
|
0
|
20
|
40
|
60
|
80
|
100
|
|
Wheat flour
|
22
|
22
|
22
|
22
|
22
|
22
|
|
Corn gluten (60%)
|
16
|
16
|
16
|
16
|
16
|
16
|
|
Rice polish
|
12
|
12
|
12
|
12
|
12
|
12
|
|
Fish oil
|
6
|
6
|
6
|
6
|
6
|
6
|
|
Vitamin Premix
|
1
|
1
|
1
|
1
|
1
|
1
|
|
Mineral Premix
|
1
|
1
|
1
|
1
|
1
|
1
|
|
Ascorbic acid
|
1
|
1
|
1
|
1
|
1
|
1
|
|
Chromic oxide
|
1
|
1
|
1
|
1
|
1
|
1
|
|
Total
|
100
|
100
|
100
|
100
|
100
|
100
|
Almond meal was added at the expense of wheat flour
Triplicate tanks (having capacity of 70 L water) were used for each treatment and each replicate had 15 fingerlings each. The experimental trial was conducted following a completely randomized design (CRD) for a period of 70 days. Fingerlings fed with test diets were compared with control as well as with test groups to assess nutrient digestibility, growth performance and hematological parameters. Electric mixer was used for thorough mixing of all ingredients for at least 10 minutes. During mixing, 10-15% water was also added. Pelleting machine was used to form suitable sized pellets. After that pellets were oven dried and stored for further use. For each treatment three replicates were used. There were fifteen fingerlings in each tank. This experimental work was conducted for 70 days. For proximate analysis, feces were collected from each triplicate tank.
Table 2. Chemical composition (%) of feed ingredients
|
Ingredients
|
Dry matter
(%)
|
Crude Protein
(%)
|
Crude Fat
(%)
|
Crude Fiber
(%)
|
Ash
(%)
|
Gross Energy
(kcal/g)
|
Carbohydrates
|
|
Fish meal
|
91.63
|
48.14
|
7.06
|
1.07
|
24.74
|
2.30
|
16.69
|
|
Wheat flour
|
92.45
|
10.01
|
2.25
|
2.30
|
2.06
|
2.76
|
80.62
|
|
Corn gluten 60%
|
92.33
|
59.48
|
4.56
|
1.19
|
1.39
|
4.32
|
29.06
|
|
Rice polish
|
94.09
|
11.02
|
13.54
|
12.7
|
10.18
|
3.33
|
49.23
|
|
Almond meal
|
91.76
|
38.68
|
5.19
|
3.37
|
13.43
|
3.34
|
35.99
|
Chemical Analysis of Feed and Feces: The standard method of (AOAC, 1995) was used to analyse chemical composition of homogenized samples of feed. According to this method, moisture was examined at 105°C temperature by drying in ovenfor 12 hours. Analysis of Crude fat was carried out through Soxtec HT2 1045 system by petroleum ether extraction method. The analysis of crude protein (N × 6.25) was made by micro Kjeldahl apparatus. To analyse crude fibre (CF), digestion of dried lipid free residues with 1.25% H2SO4 and 1.25% NaOH was carried out. Ash was analysed in electric furnace at constant heat by ignition at 650°C for 12 hours (Eyela-TMF 3100). Following difference was used to calculate the total carbohydrate, i.e.
Total carbohydrates % =100- (CP%+ EE%+ CF%+ Ash% +Moisture %)
Gross energy of ingredients was determined byusing oxygen bomb calorimeter.
Growth Study: L. rohita fingerlings were fed twice a day (morning and afternoon). At the start of the experiment the fish fingerlings were fed at the rate of 5% of live wet weight on their prescribed diet and later on adjusted to a daily basis intake of feed by fish. For each test diet, three replicates were used, and in each replicate, 15 fingerlings (average weight: 6.88g fish-1) were stocked. To observe the growth performance, L. rohita fingerlings were bulk weighed at the start and end of feeding trial. From each tank, the uneaten diet was drained out after the feeding period of two hours. Before refilling the water, the tanks were washed completely to remove the particles of uneaten diets. Following standard formulae were used to estimate weight gain percentage and feed conversion ratio (FCR) of fingerlings:


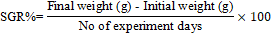
Nutrient Digestibility: Two hours after feeding process, tanks were washed completely to remove the uneaten particles of diet. For estimation of mineral absorption and nutrient utilization, fecal collection tube was used to collect feces from each tank. Oven was used to dry the fecal material at 60oC, as it will be used for further chemical analysis. To examine the chromic oxide content in diet and feces, its oxidation with molybdate reagent was done at 370 nm absorbance using UV-VIS 2001 spectrophotometer (Divakaran et al., 2002). At the end of experimental work, chromic oxide was used as an inert marker, for determination of apparent gross energy, apparent nutrient digestibility coefficients (ADC %) of crude protein and crude fat.
ADC (%) of diets was calculated by the following standard formula (NRC 1993):

Blood Collection and Hematological Analysis: To study hematological parameters, collection of blood samples was done at the end of experimental period. After 70 days of feeding trial, 3 fish from each group were collected, and anesthetized by injecting 150 mg-1solution of tricane methane sulfonate (MS222) (Wagner et al., 1997). Blood samples were taken to the Molcare Lab, Department of Biochemistry, University of Agriculture, Faisalabad, Pakistan. Blaxhall and Daisley (1973) described method of Wintrobe and Westergreen was used to determine hematocrit with micro hematocrit technique, using heparinized capillary tube. Red blood cells (RBCs) and white blood cells (WBCs) were calculated using a haemocytometer with improved Neubauer counting chamber. In order to determine hemoglobin (Hb) concentration, Wedemeyer and Yastuke (1977) method was used. For hematological studies, mean cell volume (MCV), mean corpuscular hemoglobin concentration (MCHC) and mean corpuscular hemoglobin (MCH) were analyzed.
MCHC = (Hb/PCV) × 100
MCV = (PCV/RBC) × 10
MCH = (Hb/RBC) × 10
Statistical analysis: Data of growth, hematology and nutrient digestibility of fish fed with almond meal diet and control diet was subjected to one-way Analysis of Variance (Steel et al.,1996). Tukey’s Honestly Significant Difference Test was used to relate difference between means, and considered significant at p≤0.05 (Snedecor and Cochran, 1991). The statistical analysis was performed using CoStat computer software (Version 6.303, PMB 320, Monterey, CA, 93940 USA).
RESULTS AND DISSCUSION
Growth performance: The growth performance of L. rohita fingerlings fed on diets containing almond meal at 0%, 20%, 40%, 60%, 80% and 100% level is shown in Table 3.
Table 3. Growth performance of L. rohita fingerlings fed on almond meal based diets
|
Growth parameters
|
Test Diet I
(Control diet)
|
Test Diet
II
|
Test Diet
III
|
Test Diet
IV
|
Test Diet
V
|
Test Diet
VI
|
|
Almond meal levels
|
|
0%
|
20%
|
40%
|
60%
|
80%
|
100%
|
|
IW (g)
|
6.87±0.05 a
|
6.89±0.06 a
|
6.87±0.06 a
|
6.87±0.03 a
|
6.88±0.05 a
|
6.87±0.07 a
|
|
FW (g)
|
19.77±0.29 c
|
21.42±0.23b
|
22.96±0.14 a
|
20.43±0.44 c
|
17.86±0.24 d
|
14.85±0.36 e
|
|
WG (g)
|
12.90±0.25c
|
14.53±0.21b
|
16.09±0.17 a
|
13.55±0.46 c
|
10.98±0.19 d
|
7.98±0.32 e
|
|
WG (%)
|
187.77±2.86c
|
211.04±3.14b
|
234.06±3.90a
|
197.20±7.16c
|
159.51±1.84d
|
116.05±4.22e
|
|
WG (fish-1 day-1)g
|
0.18±0.004 c
|
0.21±0.003 b
|
0.23±0.002 a
|
0.19±0.007 c
|
0.16±0.003 d
|
0.11±0.005 e
|
|
FI (g)
|
0.28±0.009 a
|
0.30±0.008 a
|
0.30 ±0.011 a
|
0.28±0.014 a
|
0.25±0.012 b
|
0.21±0.008 c
|
|
FCR
|
1.54±0.019bc
|
1.44±0.032 b
|
1.29±0.039 d
|
1.46±0.028 c
|
1.62±0.056 b
|
1.86±0.046a
|
|
SGR
|
1.17±0.01 c
|
1.26±0.01 b
|
1.34±0.01 a
|
1.21±0.03 c
|
1.06±0.01 d
|
0.86±0.02 e
|
Means within rows having different superscripts are significantly different at p≤0.05
Data are means of three replicates
(IW= Initial Weight, FW= Final Weight, WG= Weight gain, FI= Feed Intake, SGR= Specific Growth Rate,
FCR= Feed Conversion Ratio)
The results showed that replacement of almond meal with fish meal at 40% level improved the WG (g), WG (%), FCR and SGR of fish (p≤0.05). However, the further increase of almond meal level in diet decreased the weight gain of fish. A significant difference (p≤0.05) was observed in fingerlings fed with control diet and diet containing 40% almond meal. But the higher levels (80%, 100%) of almond meal did not affect the fish performance when compared with control diet. Results of the experiment showed that 40% almond meal diet is best for the optimum growth performance of L. rohita. Many researchers investigated the health effects of almond meal by determining the feed conversion ratio and weight gain values. But in present study, health status of fish was determined on the basis of FCR, SGR% and weight gain %. Although literature is available on fish meal replacement with plant by-products, yet limited data is available on growth parameters of L. rohita fingerlings fed with almond meal. These results are quite similar with the findings of Falaye et al. (2016). According to them, almond kernel meal improved the growth performance and weight gain of Clarias gariepinus juveniles when fed for 28 days. The increase in weight gain supported the fact that fish was able to digest protein present in differentially processed almond meal-based diet. Similarly, Barrows and Frost, (2014) suggested that when trout fry were fed with 58% almond meal diet, gained weight and showed equivalent survival than the control diet having 45% fishmeal. In contrast to our results, Christian and Ukhun, (2006) reported that the extracts of defatted T. catappa seed meal depressed the growth rate in rats.
Nutrient Digestibility: Analyzed values of apparent crude protein, ether extract and gross energy of almond meal-based diets and feces of L. rohita fingerlings are mentioned in Table 4, 5 and 6. The results revealed that fish fed on 40% almond meal containing diet released minimum values of the crude protein, gross energy and ether extract through feces. These lower values of crude protein and gross energy were found to be in feces at 40% level of almond meal diet having significant difference from remaining test diets and control diet. However, the maximum release of nutrients in the feces was observed when fed with 100% almond meal containing diet. The highest apparent digestibility coefficient (ADC %) of crude fat, crude protein and gross energy were observed at the 40% almond meal level which varies significantly from other treatments (Table 6). The level of ADC% starts to increase at 20% and followed by 40% almond meal while further increase of almond meal resulted in decreased ADC% of nutrients.
Table 4. Analyzed compositions (%) of apparent crude protein (CP), ether extract (EE) and gross energy (GE) and chromic oxide in feed of L. rohita fingerlings fed on almond meal based diets
|
Experimental diets
|
Almond meal levels
|
CP (%)
|
EE (%)
|
GE (kcalg-1)
|
Chromic oxide (%)
|
|
Test diet-I (control)
|
0%
|
31.79±0.33a
|
7.13±0.08a
|
3.50±0.10a
|
0.97±0.07a
|
|
Test diet-II
|
20%
|
31.80±0.55a
|
7.11±0.08a
|
3.49±0.16a
|
0.94±0.05a
|
|
Test diet-III
|
40%
|
31.80±s0.34a
|
7.12±0.10a
|
3.49±0.13a
|
0.96±0.05a
|
|
Test diet-IV
|
60%
|
31.80±0.10a
|
7.13±0.06a
|
3.50±0.05a
|
0.95±0.03a
|
|
Test diet-V
|
80%
|
31.79±0.18a
|
7.14±0.06a
|
3.48±0.11a
|
0.96±0.05a
|
|
Test diet-VI
|
100%
|
31.79±0.26a
|
7.15±0.08a
|
3.47±0.12a
|
0.97±0.07a
|
Means within columns having different superscripts are significantly different at p≤0.05
Data are means of three replicates
Digestibility or nutrient utilization values are an important parameter to consider in the diet preparation. Poorly digested feedstuffs would be less valuable to an animal. To fulfill energy demands for vital body function, fish body utilizes lipid reserves of body. According to the findings of present study, it was concluded that highest values of ADC% of crude protein was in fish fed with 40% almond meal-based diet. Whereas minimum digestibility values of crude protein were found in fish fed control diet. However, the ADC% of nutrients could not improve significantly at higher levels such as 60%, 80% and 100% almond meal-based diets. In line with our study, Soltan et al. (2008) also found that 45% plant protein meal showed best ADC% for tilapia and ADC% decreases by the further increase of plant protein meal.
Table 5. Analyzed compositions (%) of apparent CP, EE, GE and chromic oxide in feces of L. rohita fingerlings fed on almond meal based diets
|
Experimental diets
|
Almond meal levels
|
CP (%)
|
EE (%)
|
GE (kcalg-1)
|
Chromic oxide (%)
|
|
Test diet-I (control)
|
0%
|
12.80±0.24c
|
2.79±0.09bc
|
1.40±0.04c
|
1.01±0.05a
|
|
Test diet-II
|
20%
|
11.87±0.44d
|
2.50±0.11c
|
1.30±0.02c
|
1.02±0.06a
|
|
Test diet-III
|
40%
|
10.18±0.95e
|
2.01±0.16d
|
1.11±0.11d
|
1.12±0.16a
|
|
Test diet-IV
|
60%
|
12.40±0.81d
|
3.01±0.20b
|
1.37±0.06c
|
1.04±0.02a
|
|
Test diet-V
|
80%
|
14.65±0.32b
|
3.61±0.14a
|
1.62±0.03b
|
1.05±0.04a
|
|
Test diet-VI
|
100%
|
16.83±0.59a
|
3.47±0.15a
|
1.85±0.09a
|
1.03±0.05a
|
Means within columns having different superscripts are significantly different at p≤0.05
Data are means of three replicates
Table 6. Apparent nutrient digestibility of L. rohita fingerlings fed on almond meal based diets
|
Experimental diets
|
Almond meal levels
|
CP (%)
|
EE (%)
|
GE (kcalg-1)
|
|
Test diet-I (control)
|
0%
|
61.49±0.72c
|
62.58±0.81c
|
61.80±0.97c
|
|
Test diet-II
|
20%
|
65.49±0.88b
|
67.46±0.77b
|
65.55±0.78b
|
|
Test diet-III
|
40%
|
72.40±0.78a
|
75.63±0.94a
|
72.62±0.93a
|
|
Test diet-IV
|
60%
|
64.43±0.94b
|
61.46±0.88c
|
64.28±0.96bc
|
|
Test diet-V
|
80%
|
57.61±0.95d
|
53.50±1.72d
|
57.24±0.85d
|
|
Test diet-VI
|
100%
|
50.38±0.60e
|
54.46±0.90d
|
50.20±0.99e
|
Means within columns having different superscripts are significantly different at p≤0.05
Data are means of three replicates
Hematology: Hematological parameters of L. rohita fingerlings fed almond meal diets are presented in Table 7 and 8. The highest values of RBCs (2.98×106 mm-3), WBCs (7.77 ×103 mm-3) and Hb (8.31g/100ml) were observed in fish fed at 40% almond meal diet and was significantly different (p≤0.05) from all other diets. The next higher levels of almond meal inclusion in diet decreased the RBCs, WBCs, and Hb concentration. The values of PCV (25.01%) and MCHC (34.81%) were increased in fish fed on 60% almond meal diet. These values were significantly different (p≤0.05) from other experimental diets and the lowest values of PCV and MCHC was recorded at 20%, 40% and 80% almond meal based diet. From the results of MCH and MCV data, it was concluded that fish fed 100% and 80% almond meal based diet had the highest values whereas, the least values were recorded in fish fed 20% almond meal based diet.
Table 7. Hematological parameters (WBCs, RBCs and PLT) of L. rohita fingerlings fed on almond meal based diets
|
Experimental diets
|
Almond meal Levels
|
RBCs (106mm-3)
|
WBCs (103mm-3)
|
PLT
|
|
Test diet-I (control)
|
0%
|
2.16±0.13bc
|
6.85±0.16bc
|
59.00±0.25 d
|
|
Test diet-II
|
20%
|
2.47±0.19 b
|
7.18±0.24ab
|
64.03±0.34 b
|
|
Test diet-III
|
40%
|
2.98±0.15 a
|
7.77±0.22 a
|
66.93±0.30 a
|
|
Test diet-IV
|
60%
|
2.40±0.18 b
|
7.05±0.23bc
|
62.96±0.13 c
|
|
Test diet-V
|
80%
|
2.16±0.16bc
|
6.44±0.27cd
|
58.74±0.43d
|
|
Test diet-VI
|
100%
|
1.76±0.13 c
|
5.99±0.29 c
|
55.00±0.25 e
|
RBC = Red Blood Cell, WBC = White blood cell, PLT = Platelet, Hb = hemoglobin
Means within columns having different superscripts are significantly different at p ≤ 0.05
Data are means of three replicates
Table 8. Hematological parameters (Hb, PCV, MCHC, MCH and MCV) of L. rohita fingerlings fed on almond meal based diets
|
Experimental diet
|
Almond meal levels
|
Hb (g/100ml)
|
PCV (%)
|
MCHC (%)
|
MCH (pg)
|
MCV (fl)
|
|
Test diet-I (control)
|
0%
|
6.99±0.13 b
|
22.73±0.28b
|
26.17±0.40d
|
35.94±0.75e
|
101.56±0.35e
|
|
Test diet-II
|
20%
|
7.52±0.46ab
|
24.36±0.92a
|
28.46±0.49b
|
39.28±0.40d
|
97.77±0.45f
|
|
Test diet-III
|
40%
|
8.31±0.24 a
|
23.62±0.43a
|
31.42±0.43b
|
40.27±0.36d
|
165.32±0.47c
|
|
Test diet-IV
|
60%
|
7.20±0.23 b
|
25.02±0.45a
|
34.81±0.22a
|
47.13±0.33c
|
172.18±0.32b
|
|
Test diet-V
|
80%
|
6.11±0.27 c
|
22.77±0.37b
|
30.88±0.18b
|
50.93±0.39b
|
181.94±0.41a
|
|
Test diet-VI
|
100%
|
5.93±0.33 c
|
21.25±0.50c
|
26.49±0.36d
|
53.78±0.38a
|
154.18±0.37d
|
(Hb = Hemoglobin, PCV = Packed cell volume, MCHC = Mean corpuscular hemoglobin concentration MCH = Mean corpuscular hemoglobin, MCV = Mean corpuscular volume)
Means within columns having different superscripts are significantly different at p ≤ 0.05
Data are means of three replicates
Fish biologists determine the health status of fish by the application of hematological techniques. Our results are similar to the reference values reported by Siddiqui and Naseem, (1979), in which they described the hematological parameters of L. rohita as RBCs (2.29×106 mm-3), WBCs (6.25×103 mm-3), Hb (9g/100ml), MCV (155.22µm3), MCHC (25.38%) and MCH (39.56%). Furthermore, the normal hematological indices of other monogastric fishes like Nile tilapia and common carp were also reported by Ismail and Mahboub, (2016) and Witeska et al. (2016), respectively. They reported nearly similar results with reference to RBCs (1.13×106/µL; 1.51×106/µL), WBCs (28.58×103/µL; 51.3×103/µL) and Hb (4.70g/dL; 6.42g/dL). By comparing our results to the above cited reference values, we can estimate that health status of the fish was not affected by the replacement of fish meal with almond meal up to 40%. In line with our study, Nisha and Geetha, (2017) revealed that L. rohita fed with 30% Pistia stratiotes meal based diet resulted in improved level of RBCs (2.38 ×106 mm-3). Fagbenro and Adeparsuri, (2003) monitored hematological stress behavior of the fish. In his experimental study, increase in almond meal level in diet decreased the white blood cells count in fish body. Mmereole, (2008) findings supported our results that above 25% rubber seed meal in monogastric animal results in decrease in WBC. The mechanism responsible for the improved hematological indices of fish can be the flavonoid present in almond meal, which provokes erythropoiesis and limits RBCs hemolysis by strengthening the cellular membranes from oxidative damage (Asgary et al., 2005). Sotolu and Faturoti, (2009) demonstrated results opposite to our findings that 80% Leucaena seed meal resulted in high WBC count which was due to the presence of foreign antigen. However, Wedemeyer and Wood, (1974) found that changes in WBC count in stressed fish are the expression of the immune system and increases vulnerability to disease. In the hemoglobin analysis of the present study, fish fed with 40% almond meal based diet significantly improved values of Hb (8.31g/100ml). While, the second higher values of hemoglobin (7.20g/100ml) was found in fish fed with 60% almond meal based diet which were different from remaining almond meal based diets. The findings of Arsalan et al. (2016) supported our results that Hb concentration increased when given 10% (8.91g/100ml) and 20% (8.53g/100ml) Moringa oleifera leaf meal based diet to L. rohita fingerlings. Furthermore, Nugroho etal. (2016) stated the same values of Hb (8.581 g dL-1) by feeding almond leaves extract based diet up to 600ppm to Betta species. The highest significant values of PCV (25.02%) and MCHC (34.81%) were found at 60% almond meal as compared to remaining diets while MCH (47.13pg) and MCV values were maximum (181.94fl) at 80%. Accordingly, Arsalan et al. (2016) found the PCV (28.01%), MCHC (36.60%), MCH (72.47pg) and MCV (209.03fl) values by giving 30% and 40% MOLM based diets to L. rohita fingerlings.
Conclusion: In conclusion, replacement of fishmeal with almond meal has significantly improved the growth performance, nutrient digestibility and hematology of L. rohita fingerlings fed on almond meal. Whereas 40% replacement level based diet was found more effective as compared to other replacement levels based diet.
References
- Akpabio, U.D., A.E. Akpakpan, I.E. Matthew, and A.U. Akpan (2011). World J. Sci. Technol. 3(2): 27-34.
- Allan G.L. and S.J. Rowland (1992). Development of an experimental diet for silver perch (Bidyanus bidyanus). Austasia Aquacult. 6(3): 39-40.
- Annegowda H.V., C.W. Nee, M.N. Mordi, S. Ramanathan, and S.M. Mansor (2010). Hydrolyzed extracts of Terminalia catappa leaf. Asian J. Plant Sci. 9(8):479-485.
- AOAC (1995). Official methods of analysis. 15th Association of Official Analytical Chemists, Washington, D.C (USA).1094 p.
- Arsalan M.Z.H., S.M. Hussain, M. Asrar, H. Anwar, M.M.H. Rehan, M.M. Shahzad, and N. Wahab (2016). Effects of Moringa oleifera leaf meal (MOLM) based diets on carcass composition and hematology of Labeo rohita J. Biodiv. Environ. Sci. 9(1): 214-223.
- Asgary S., G. H. Naderi, and N. Askari (2005). Protective effect of flavonoids against red blood cell hemolysis by free radicals. Exp. Clin. Cardiol. 10(2):88-90.
- Blaxhall P.C., and K.W. Daisley (1973). Routine hematological methods for use with fish blood. J. Fish Biol. 5(6): 771-781.
- Barrows F.T. and J.B. Frost (2014). Evaluation of the nutritional quality of co-products from the nut industry, algae and an invertebrate meal for rainbow trout, Oncorhynchus mykiss. Aquaculture. 434: 315-324.
- Christian A. and M.E. Ukhun (2006). Nutritional potential of the nut of Tropical Almond (Terminalia catappa L.).Pak J. Nutr. 5(4): 334-336.
- Divakaran S., G.O. Leonard and P.F. Ian (2002). Note on the methods for determination of chromic oxide in shrimp feeds. J. Agric. Food Chem. 50(3): 464-467.
- Elezuok O. (2016). Growth performance of Clarias gariepinus juveniles fed processed almond (Terminalia catappa) kernel meal (doctoral dissertation). M.Sc. thesis. Department of Aquaculture and Fisheries Management, Univ. Ibadan, Nigeria.
- Fagbenro O.A., E. Adeparusi and O. Fapohunda (2003). Feed stuffs and dietary substitutions for farmed fish in Nigeria. In proceedings of the national workshop on fish feed development and feeding practices in aquaculture, National Fresh Water Fisheries Research Institute, 60-65 p.
- Falaye A.E., K.E. Elezuo, E.K. Ajani and A. Omoike (2016). Digestibility and nutrient utilization of differently processed tropical almond (Terminalia catappa) kernel meal and cake based diets by Clarias gariepinusJ. Agric. Sci. Technol. 17(1): 42-60.
- Hussain S.M., A. Javid, A.I. Hussain, N. Aslam, Q. Ali, M. Hussain, and D. Riaz (2018). Replacement of Fish Meal with Moringa oleifera Leaf Meal (MOLM) and its Effect on Growth Performance and Nutrient Digestibility in Labeo rohita Pak. J. Zool. 50(5): 1825-1834.
- Hussain, S. M., M. Afzal, A. Javid, N. Aslam, M. Hussain, S.Z.H. Shah, H. Azmat and M.S. Mubarik (2015). Role of phytase supplementation in improving nutrient digestibility in Labeo rohita (Hamilton, 1822) fingerlings fed on cottonseed meal based diet. Indian J. Fish., 62(1): 78-84.
- Ismail H. T. H., and H. H. H. Mahboub (2016). Effect of acute exposure to nonylphenol on biochemical, hormonal, and hematological parameters and muscle tissues residues of Nile tilapia; Oreochromis niloticus. Vet. World, 9(6): 616-625.
- Lazzarotto V., F. Médale, L. Larroquet and G. Corraze (2018). Long-term dietary replacement of fishmeal and fish oil in diets for rainbow trout (Oncorhynchus mykiss): Effects on growth, whole body fatty acids and intestinal and hepatic gene expression. PLoSONE. 13(1): e0190730.
- Lech, P.L. and R.C. Reigh (2012). Plant products affect growth and digestive efficiency of cultured Florida pompano (Trachinotus carolinus) fed compounded diets. PLoS One, 7, 1-11.
- Mmereole F.U.C. (2008). The effects of replacing groundnut cake with rubber seed meal on the hematological and serological indices of broilers. J. Poult Sci.7(6): 622-624.
- Nisha, S. N., and B. Geetha (2017). Effect of partial replacement of fishmeal with aquatic weed Pistia stratiotes meal on growth, biochemical composition, haematological parameters and digestive enzymes in Indian major carp Labeo rohita (Hamilton, 1822). Int. J. Fish. Aquat. Stud. 5(2):527-532.
- NRC (1993). Nutrient requirements of fish. National Research Council, National Academy Press, Washington, DC, USA, p. 233-289.
- Nugroho R. A., H. Manurung, D. Saraswati, D. Ladyescha and F.M. Nur (2016). The effects of Terminalia catappa leaves extract on the water quality properties, survival and blood profile of ornamental fish (Betta sp) cultured. Biosaintifika. 8(2): 240-247.
- Olsen R.L. and M.R. Hasan (2012). A limited supply of fishmeal: Impact on future increases in global aquaculture production. Trends Food Sci. Technol.27(2): 120-128.
- Rowland S.J. and B.A. Ingram (1991). Diseases of Australian native freshwater fishes with particular emphasis on the ectoparasite and fungal diseases of Murray cod (Maccullochella peeli), golden perch (Macquaria ambigua) and silver perch (Bidyanus bidyanus). NSW Fisheries Bulletin Number 4, Sydney.
- Siddiqui A.Q. and S.M. Naseem (1979). The haematology of Rohu, Labeo rohita. J. Fish Biol. 14(1): 67-72.
- Snedecor G.W. and W.G. Cochran (1991). Statistical Methods. 8th Iowa State University, Press American, USA.503 p.
- Soltan M.A., M.A. Hanafy and M.I.A. Wafa (2008). Effect of replacing fish meal by a mixture of different plant protein sources in Nile tilapia (Oreochromis niloticus L) diets.Global Vet. 2(4): 157-164.
- Sotolu A.O. and E.O. Faturoti (2009). Growth performance and hematology of Clarias gariepinus (Burchell, 1822) fed varying inclusions of Leucaenaleuco cephala seed meal based-diets. Cient. Agríc.9(4): 979-985.
- Steel R.G.D., J.H. Torrie, and D.A. Dickey (1996). Principles and procedures of statistics. 3rd McGraw Hill international Book Co. Inc., New York (USA). 336-352 p.
- Wagner A.D., J.D. Gabrieli, and M. Verfaellie (1997). Dissociations between familiarity processes in explicit recognition and implicit perceptual memory. Exp. Psychol: Learn, Mem., Cognit. 23(2): 305-323.
- Wedemeyer G.A and W.T. Yastuke (1977). Clinical methods for the assessment of the effects of environmental stress on fish health.U. S. Fish Wildl. Serv. Tech. Pap. 89.
- Wedemeyer G.A. and J.W. Wood (1974). Stress as a predisposing factor in fish diseases. S. Fish Wildl. Serv. 38.
- Witeska M., K. Lugowska, and E. Kondera (2016). Reference values of hematological parameters for juvenile Cyprinus carpio. Bull. Eur. Ass. Fish Pathol. 36(4): 169.
|





