AERVA JAVANICA MEDIATED SYNTHESIS, CHARACTERIZATION AND ANTIMICROBIAL EVALUATION OF ZINC OXIDE NANOPARTICLES
G. Afzal1, A. Jamal2’*, S. Kiran3, G. Mustafa4, F. Ahmad1, S. Saeed5, H. I. Ahmad6 and S. Dawood1
1Department of Zoology, The Islamia University of Bahawalpur, Bahawalpur, 36100, Pakistan.
2Sciences and Research, College of Nursing, Umm Al-Qura University, Makkah-715, Kingdom of Saudi Arabia.
3Department of Applied Chemistry, Government College University, Faisalabad, Pakistan.
4Department of Biochemistry, Government College University, Faisalabad, Pakistan.
5Institute of Physics, The Islamia University of Bahawalpur, Bahawalpur, 36100, Pakistan.
6Department of Animal Breeding and Genetics, University of Veterinary and Animal Sciences, Lahore, Pakistan.
*Corresponding Author’s Email: aajamal@uqu.edu.sa, adiljamalcemb@gmail.com
ABSTRACT
Zinc is an indispensable trace element in the human body. Nano sized zinc-based particles have extensive applications in different fields such as medicine, agriculture and biotechnology. The present study was designed to describe the rapid, simple and environment friendly synthesis of zinc oxide nanoparticles (ZnO NPs) using Aerva javanica. In the current study, biosynthesis and characterization of ZnO NPs using Aerva javanica leaf extract is reported for the first time. Synthesized ZnO NPs were characterized by X-Ray Diffraction (XRD) and Fourier Transform Infrared Spectroscopy (FTIR) techniques. Synthesized ZnO NPs were tested against antimicrobial activity using the agar diffusion method. Antibacterial activity of ZnO NPs (10 mg/mL) was measured and zones of inhibition (MIC) against S. aureus and P. aeruginosa were measured as 13.5 mm + 0.8 and 5 + 2.8 mm while ZnO NPs (40 mg/mL) exhibited 18.4 + 4.3 and 8 + 2.8 mm for S. aureus and P. aeruginosa respectively. Antifungal activity of ZnO NPs (10 mg/mL) was measured and MIC against C. albicans and A. niger were recorded as 2.6 mm + 1.7 and 2+ 0.5 mm while ZnO NPs (40 mg/mL) exhibited 6.6 + 2.0 and 2.5 + 0.5 mm for C. albicans and A. niger respectively. Our study concluded that MIC for S. aureus and C. albicans was found to be higher as compared to P. aeruginosa and A. niger. Data analysis showed that ZnO NPs had a significant inhibitory antibacterial effect as compared to antifungal. Thus, present study depicts that the plant extract of A. javanica can be a valuable natural resource to synthesize the functional nanomaterials for targeted applications particularly in molecular medicine.
Key words: Cholistani shrub, Anti-microbial activity, Aerva javanica, Green synthesis, ZnO Nanoparticles, Molecular-medicine, XRD, FTIR
http://doi.org/10.36899/JAPS.2022.2.0453
Published first online August 13. 2021
INTRODUCTION
Nanotechnology has significant progress in terms of applications and research. Augmented applications in nano biotechnology are a consequence of marvelous and astounding features obsessed by nanoparticles like versatile biological and chemical activities diffusivity and also enormous applications as that of their bulked and raw materials (Reddy, 2017; Sharmila et al., 2018). Molecular medicine is an extensive application of nano biotechnology. By the combination of nanotechnology and biology, this has been resulted in contending infectious diseases. Especially, the biologically synthesized nanoparticles have gained substantial interest globally. Nanoparticle size ranges from 10-10 nm (Bhosale et al., 2015).
Bacteria, fungi, viruses, yeast algae and plants, are successful candidates for the biological synthesis of nanoparticles (Fawcett et al., 2017; Gu et al., 2018). Nanostructured and metal oxide particles like CuO, ZnO, MgO, TiO2 and their composites are getting much popularity because of their antibacterial properties (Akhavan et al., 2011). Metal oxides-based nanoparticles are attaining tremendous popularity because of their antimicrobial efficacy and activity even at a minimum dose (Anagnostakos et al., 2008). ZnO nanoparticles among the metal oxides have been studied extensively both for industrial (Yan et al., 2011; Jeong et al., 2016) and scientific applications because of their antiviral, antifungal and antibacterial activities. ZnO nanoparticles have been reported to show promising activity against microbes, especially sturdier than bulk ZnO (Yamamoto, 2001). ZnO, at significantly low concentrations, is an effective antifungal and antibacterial agent (Applerot et al., 2009; Padmavathay and Vijayyararaghvan, 2008). ZnO nanoparticles with small size provide more surface area and hence allow electrostatic interactions of nanoparticles with the cell surface of microbes, generating pit in the cell envelope, leading to cytoplasmic contents leakage and persuading microbial cell death (Liu et al., 2009; Sharma et al., 2010). The action of nanoparticles partially relies on stability, size, and concentration in the growth medium for their antimicrobial activity.
Considering their broad applications, ZnO nanoparticles synthesis is a crucial task to the scientific community. ZnO nanoparticles production has been carried out by involving many chemical and physical approaches. The uncommon biological, physiochemical features have clasped extensive consideration of the scientists. These methods are harmful, energy consuming, costly and non-ecofriendly. Therefore, the biological synthesis approach using plants and their extracts, has evolved as a suitable approach with reduced cost and environment friendly for the ZnO nanoparticles preparation (Kowshik et al., 2002). Biologically synthesized ZnO nanoparticles have been reported against bacterial and fungal strains using different plants including Aloe vera (Gunalan et al., 2012), Cassia fistula (Suresh et al., 2015) etc. Aerva javanica commonly known as Cholistani shrub (kapok bush) is a member of the plant family Amaranthaceae perennial semi-shrubby growing in tropical and subtropical dry areas. In Pakistan, it exclusively grows in the desert area (Cholistan) of Punjab. Aerva genus is represented by 20 species in Indo Pakistan region (Sharif et al., 2011). This herb has been traditionally used as diuretic, diabetic, demulcent and kidney stones (Qureshi and Bhatti, 2009). Leaves of A. javanica are used in wound healing, joints inflammation. A. javanica displays cytogenetical (Soliman, 2006), cytotoxic (Al-Fatimi et al., 2007), anti-hyperglycaemic (Reddy and Reddy, 2009) and anti-plasmodial activities (Ahmed el et al., 2010).
It has been reported previously that metabolites of A. javanica contain triterpenoids, steroids, flavonoids and carbohydrates (Ahmed-el et al., 2010). Because of the rich metabolite features of A. javanica and their suggested role as bio reducing agents, consider them as a promising candidate for the ZnO nanoparticles synthesis, which is planned in the current study. In addition, it was also investigated that changing concentrations of precursor (plant extract and zinc salt) have any effect on antibacterial and antifungal activity.
MATERIALS AND METHODS
Preparation of Plant Extract: Fresh leaves of A. javanica were collected from Cholistan desert area, Bahawalpur, Punjab. Collected leaves were washed thoroughly using tap water several times and subsequently with distilled water. Leaves were kept under shadow drying at room temperature. For the extract preparation, 50g of cleaned dried A. javanica leaves were sliced and later boiled in 25 ml of sterilized deionized water at 60 oC for 1 hr. The solution was cooled down at room temperature and was later filtered using Whatman filter paper no.1. Cooled filtered extract was centrifuged at 4000 rpm for 25 minutes. The supernatant was further stored at 4 °C until later use.
Synthesis of ZnO Nanoparticles: ZnSO4 solution was prepared using 1M concentration in deionized water. The pH of reaction ZnSO4 solution was adjusted using 0.05 M NaOH in deionized water. A sample of ZnO NPs was prepared using 20 ml of leaf extract with 80 ml solution of 1M ZnSO4.7H2O. pH was adjusted (pH 10) using NaOH. The resultant light green color solution stirred for 2 hours at 60 °C under constant stirring till color changes to pale white. Mixture incubated overnight as centrifuged at 4000 rpm for 25 minutes to separate the precipitates. Pellet was washed thrice with distilled deionized water, dried at 120 oC in a hot air oven for 6 hrs. Oven dried pellet was ground to a fine powder. To obtain pure ZnO NPs, the ground powder was further calcinated at 150 oC to avoid any remaining impurities (organic compounds).
Characterization of Synthesized ZnO Nanoparticles: XRD and FT-IR analysis of the synthesized ZnO NPs from A. javanica leaf extract were done using the facilities of Central Research Lab (CRL), Institute of Physics, The Islamia University of Bahawalpur, Pakistan following the standard manufacturer protocols. XRD is the most valuable and important approach and the simplest method and this method was performed for the identification of the crystalline phase of the synthesized ZnO NPs using XRD (D8 Burker, Germany). Scanning was performed in the region of 20o- 80o. The nanoparticles crystallite size was measured using Scherre’s formula, d=Kλ/β cosθ; where d is the crystallite size, is the Scherrer's constant (K= 0.9), λ is the wavelength (λ=1.5406 A°), θ is the diffraction angle, β is the half width maximum. FT-IR (Burker, Germany) was done to identify the functional group presence and for the stabilization of biosynthesized ZnO NPs in the wavelength range 4000-500 cm-1.
Microorganisms: Antibacterial activity was carried out using Staphylococcus aureus and Pseudomonas aeruginosa. The antifungal activity evaluation was performed against Candida albicans and Aspergillus niger. These microorganisms were collected from the Microbiology Department, The Islamia University of Bahawalpur.
Antimicrobial Activity: The antimicrobial activity of synthesized ZnO NPs was performed using the standard disc diffusion method. The overnight grown S. aureus, P. aeruginosa, C. albicans and A. niger at 37 0C were standardized using McFarland standard corresponded to 1×106 CFU/ml. About 5 mm diameter discs were used using Whatman filter paper. Biosynthesized ZnO NPs of 10 and 40 mg/ml stocks were prepared. A 5g of solidified MHA was added with 45 ml of distilled water and later sterilized. The mixture prepared was transferred into four petri plates and microorganisms to be tested were swabbed over the surface of MHA plates. Microorganisms to be checked were inoculated in three labelled discs (5 mm diameter) dipped well in dilutions of ZnO NPs (10, 40 mg/ml) containing ofloxacin as an antibiotic. 100 µl of ZnO NPs suspension (10, 40 mg/ml), positive control DO 40 µg/ml (antibacterial), CAS 10 µg/ml (antifungal) and negative control (distilled water) were used. After incubation at 370C for 72 hrs (fungal plates) and 24 hrs (bacterial plates), inhibition zones diameter was measured.
RESULTS
X-Ray Diffraction (XRD) and Fourier Transform Infrared Spectroscopy (FT-IR) Analysis: XRD was carried out for biologically synthesized nanoparticles for characterization to conform the particles as zinc. The XRD pattern of synthesized ZnO NPs is shown in Figure 1. XRD pattern showed diffraction peaks at 2θ values of 31.66º, 34.32º, 36.15º, 47.46º, 56.52º, 62.80º, 66.44o, 67.89º, 69.02º and 76.92º. Peaks indexed as 100, 002, 101, 102, 110, 103, 200, 112, 201 and 202 were consistent with zinc oxide wurtzite structure (JCPDS- 36-1451). Almost both the samples synthesized biologically showed similar peak patterns (Figure 1). Scherrer equation was used to estimate the size of nanoparticles estimated from XRD peaks. The obtained XRD results clearly show that diffract graphs of prepared ZnO NPs declined as the concentration of ZnSO4 is increased. The wurtzite structure is most common and stable in an ambient environment. As XRD peaks characteristics highly conform to the ZnO peaks so this also verifies that synthesized nanoparticles were from any impurities (Bigdeli et al., 2010).
FT-IR was performed to reveal the composition of synthesized ZnO NPs. Figure 2 shows the typical FT-IR spectrum of ZnO NPs. The FT-IR response was performed through the wavenumber range from 500-4000 cm-1 (Figure 2). FT-IR spectrum confirmed the different chemical bonds presence responsible for the synthesis of ZnO NPs. FT-IR confirmed the functional groups hexagonal phase ZnO, C-O bonding in amino acid, C-N bond of amide-I in protein, C=C bond in the aromatic ring and C=O bond in polyphenol, O-H bond in COOH, C-H bond in alkanes, OH bonding vibrations. These possible functional groups indexed at frequency of 533.71, 1018.12, 1461.45, 2845.74, 2845.74, 2912.09, 3398.56 cm-1 (Figure 2). The peak of 533 cm-1 is a characteristic absorption of Zn-O bond feature while the peak at 3398 cm-1 showed the OH characteristics absorption.
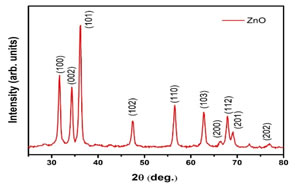
Figure 1. XRD pattern of ZnO nanoparticles
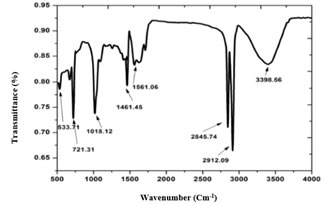
Figure 2. FT-IR spectrum of ZnO nanoparticles showing functional groups
In our study, the antimicrobial activity effects of synthesized zinc nanoparticles are studied on two different bacterial strains, including S. aureus (gram positive), P. aeruginosa (gram negative). Each sample of prepared ZnO NPs with varying concentrations, exhibited noteworthy antibacterial activity with a strong effect against the S. aureus as compared to P. aeruginosa. Lesser antimicrobial activity is observed P. aeruginosa as that of S. aureus while increasing (10, 40 mg/ml) the concentration of ZnO NPs (Table. 1). Among tested bacterial strains, S. aureus was observed as a more sensitive strain, whereas, P. aeruginosa was less sensitive to ZnO NPs. This antibacterial activity is based upon the total inhibition zones induced by different concentrations of synthesized ZnO NPs. The inhibition zone possessed by synthesized ZnO NPs against antifungal pathogens is mentioned in Table 2. The antifungal activities of two different concentrations of prepared ZnO NPs against tested fungal strains revealed C. albicans more sensitive as compared to A. niger. Lesser antifungal activity of synthesized ZnO NPs is observed against A. niger while in comparison to C. albicans with increasing concentration of Zn nanoparticles (Table. 2).
Table 1.Zone of ZnO NPs (mm) against tested bacterial strains
|
Sample / Microorganism
|
Zone of Inhibition (MIC) (mm)
|
|
Control
|
Dose of Green ZnO NPs (mg)
|
| |
10 mg
|
40 mg
|
|
Staphylococcus aureus
|
|
Sample 1
|
19
|
12
|
17.4
|
|
Sample 2
|
20
|
13.5
|
18.5
|
|
Sample 3
|
21
|
15
|
19.3
|
|
Mean values
|
20+0.5b
|
13.5+0.8b
|
18.4+4.3a
|
|
Pseudomonas aeruginosa
|
|
Sample 1
|
27
|
0
|
3
|
|
Sample 2
|
28
|
5
|
8
|
|
Sample 3
|
29
|
10
|
13
|
|
Mean values
|
28 + 0.5b
|
5+ 2.8b
|
8 + 2.8a
|
Alphabets (a,b) indicate the statistically significant differences at P ≤0.05 probability level.
Table 2.Zone of Inhibition of ZnO NPs (mm) against tested fungal strains
|
Sample / Microorganism
|
Zone of Inhibition (MIC) (mm)
|
|
Control
|
Dose of Green ZnO NPs (mg)
|
| |
10 mg
|
40 mg
|
|
Candida albicans
|
|
Sample 1
|
13
|
0
|
3
|
|
Sample 2
|
12
|
2
|
7
|
|
Sample 3
|
11
|
6
|
10
|
|
Mean values
|
12 + 0.5b
|
2.6 + 1.7a
|
6.6 + 2b
|
|
Aspergillus niger
|
|
Sample 1
|
10
|
1
|
1.5
|
|
Sample 2
|
9
|
3
|
2.7
|
|
Sample 3
|
11
|
2
|
3.3
|
|
Mean values
|
10 + 0.5b
|
2 + 0.5a
|
2.5 + 0.5a
|
Alphabets (a,b) indicate the statistically significant differences at P ≤0.05 probability level.
DISCUSSION
The ZnO NPs synthesized in this study were characterized using physical approaches XRD and FTIR. XRD pattern clearly showed the main peaks (Yedurkar et al., 2016; Irshad et al., 2020). XRD confirmed the presence or absence of bioactive compounds like terpenes, flavonoids, alkaloids, carbohydrates and phenols in supernatant layer crude leaves extract (Irshad et al., 2020). Unassigned peaks at 66.44o and 76.92º were weaker than those of zinc. This might be due to the presence of phytochemical and bioorganic compounds in the leaf extract. According to Kumar and Yadav (2009) and Jeeva et al. (2014), similar findings of XRD diffraction peeks were also observed in the range of 2θ values. The phytochemicals absence or weak presence can be explained as best that bioactive constituents play a role in the reduction and capping of metal oxides during nanoparticles synthesis (Malapermal et al., 2015). Earlier workers revealed similar comparable XRD data results (Yedurkar et al., 2016; Irshad et al., 2018, Irshad et al., 2020).
FTIR analysis of ZnO NPs showed a similar comparable pattern with other studies. A broad range between 3000-3500 cm-1 with maximum absorption at 3398.56 cm-1 that is attributed to the frequencies of amino and hydroxyl of phenols (phenolic groups), alcohols and amines while the weak absorption at 2912.09 cm-1 revealed the asymmetric and symmetric range of organic functional group (C-H stretch, alkanes) (Yedurkar et al., 2016; Irshad et al., 2020). ZnO NPs have one noticeable sharp peak at 533.71 cm-1 of hexagonal ZnO (Srinivasa and Basaveswara, 2015; Salem et al., 2016; Yedurkar et al., 2016). Moreover, peak at 1561.06 cm-1 (C=O stretch in polyphenol and C=C stretch (alkenes) in the aromatic ring) and peak at 1461.45 cm-1 (C-N stretching frequencies of amide-I in protein) in NPs FTIR spectrum. Our results are comparable with reported studies that stabilization during ZnO NPs synthesis is due to proteins (Irshad et al., 2020). A weak peak at 1018.12 cm-1 and 721.31 cm-1 correspond to C-O stretching in amino acid and alcohol respectively (Yedurkar et al., 2016; Moghaddam et al., 2017; Irshad et al., 2020). The presence of prominent and sharp peaks along with weak peak presence in ZnO NPs spectrum revealed that active ingredients having functional groups were performing the tasks of dispersing, capping and stabilizing agents for nanoparticles.
Results of previous studies documented that ZnO NPs using plant extracts exhibited promising antibacterial activity against gram positive as compared to gram negative bacteria (Premanathan et al., 2011; Azam et al., 2012; Mirhosseini et al., 2013). Sensitivity can be attributed to cell physiology, cell wall structure and composition, cellular metabolism and magnitude of contact of bacterial cell with nanoparticles (Slman, 2012). The cell wall structure is the key factor that can be ascribed to the susceptibility or tolerance to nanoparticles. According to our studies, ZnO NPs show promising activity towards the gram-positive bacteria; S. aureus, which may be described due to negatively charged bacterial surfaces and interaction between ZnO NPs (Fang et al., 2016; Murtaza et al., 2017). Previous studies documented that electrostatic interactions damage the bacterial cell integrity causing leakage of cytosolic constituents and cell death (Li et al., 2012; Zhang et al., 2015). Besides Zn+2 ions released from ZnO NPs displayed greater binding to the bacterial surface due to negatively charged bacterial surface, thereafter facilitating the entry into the cell where they impede with other different biochemical functions (Reddy et al., 2007; Applerot et al., 2009). While decreased susceptibility level in gram negative is due to an extra protective layer, the outer membrane, which functions as a selective barrier to decrease the injury by nanoparticles by their restricting entry to the cell’s interior (Russell, 2003). On the other hand, previous findings also displayed the higher susceptibility in gram negative P. aeruginosa to ZnO NPs as gram positive S. aureus (Tawale et al., 2010). This might be due to variations in intracellular antioxidants components and the occurrence of strong detoxifying agents within bacteria.
Similarly, the variation in the antifungal activity of ZnO NPs was studied. C. albicans in comparison to A. niger showed higher sensitivity which is similar to previous findings (Akbar et al., 2017). Different factors may be involved in the susceptibility or tolerance of microorganisms to nanoparticles which require more studies. Our study elucidates that resultant lower antimicrobial activity is attributed to decreased ZnO NPs concentration. Zn2+ ions in decreased concentration act as nutrients and enhances microbial growth; while higher Zn2+ concentration is harmful to microbes. Hence, augmenting particle concentration, ZnO NPs diffuse very well through growth media and its higher concentration of Zn2+ displays an amplifying antimicrobial efficacy (Stankovic et al., 2013).
Conclusion: Our study reveals that ZnO NPs synthesized from A. javanica documented the reducing, capping and stabilizing phytochemicals during characterization. ZnO NPs also showed promising antibacterial and antifungal potential. Therefore, A. javanica can be effectively used for the synthesis of ZnO nanoparticles to be used as antimicrobials in the future.
Acknowledgements: We are highly thankful to the Nanosciences and Technology Department (NS &TD), National Center for Physics, Islamabad, Pakistan and Institute of Physics, The Islamia University of Bahawalpur, Pakistan for providing the necessary facilities and technical extreme support during the study to accomplish the study objectives. The authors also articulate and expressive regards to all those who helped directly or indirectly to achieve the study goals.
Conflict of Interest: None
REFERENCES
- Ahmed-el., H.M, B. Y. Nour, Y. G. Mohammed and H.S. Khalid (2010). Antiplasmodial activity of some medicinal plants used in sudanese folk-medicine. Environ. Health Insights. 4: 1-6.
- Azam, A., A.S. Ahmed, M. Oves, M.S. Khan, S.S. Habib and A. Memic (2012). Antimicrobial activity of metal oxide nanoparticles against gram-positive and gram-negative bacteria: a comparative study. Int. J. Nanomedicine. 7: 6003-6009.
- Akbar, S., K.S. Haleem, I. Tauseef, W. Rehman, N. Ali and M. Hasan (2017). Raphanus sativus mediated synthesis, characterization and biological evaluation of zinc oxide nanoparticles. Nanosci. Nanotech. Lett. 9(12):1-8.
- Akhavan, O., R. Azimirad, S. Safad and E. Hasani (2011). CuO/Cu(OH)2 hierarchical nanostructures as bactericidal photocatalysts. J. Mater. Chem. 21: 9634-9640.
- Al-Fatimi, M., M. Wurster, G. Schroder and U. Lindequist (2007). Antioxidant, antimicrobial and cytotoxic activities of selected medicinal plants from Yemen. J. Ethnopharmacol. 111(3):657-666.
- Anagnostakos, K., P. Hitzler, D. Pape, D. Kohn and J. Kelm (2008). Persistence of bacterial growth on antibiotic-loaded beads: is it actually a problem? Acta. Orthopaedica. 79(2): 302-307.
- Applerot, G., A. Lipovsky, R. Dror, N. Perkas, Y. Nitzan, R. Lubart and A. Gedanken (2009). Enhanced antibacterial activity of nanocrystalline ZnO due to increased ROS-mediated cell injury. Adv. Func. Mater. 19(6): 842-852.
- Bhosale, R.S., K.Y. Hajare, B. Mulay and M. Kothawade (2015). Biosynthesis, characterization and study of antimicrobial effect of silver nanoparticles by Actinomycetes Int. J. Curr. Microbiol. App. Sci. 2: 144-151.
- Bigdeli, F., A. Morsali, and P. Retalleau (2010). Synthesis and characterization of different zinc (II) oxide nano-structures from direct thermal decomposition of ID coordination polymers. Polyhedron. 29: 801-806.
- Fang, L., H. Murtaza, D. Hao, H.S. Gul, M. Weiwei, D. Yulin and D. Rongji (2016). Optimized luteolin loaded solid lipid nanoparticle under stress condition for enhanced bioavailability in rat plasma. J. Nanosci. Nanotech. 16(9): 9443-9449.
- Fawcett, D., J.J. Verduin, M. Shah, S.B. Sharma and G. E. J. Poinern (2017). A review of current research into the biogenic synthesis of metal and metal oxide nanoparticles via marine algae and seagrasses. J. Nanosci. 8013850. https://doi.org/10.1155/2017/8013850.
- Gu, H., X. Chen, F. Chen, X. Zhou and Z. Parsaee (2018). Ultrasound-assisted biosynthesis of CuO-NPs using brown alga Cystoseira trinodis: characterization, photocatalytic AOP, DPPH scavenging and antibacterial investigations. Ultrasonics. Sonochem. 41:109-119.
- S., R. Sivaraj and V. Rajendran (2012). Green synthesized ZnO nanoparticles against bacterial and fungal pathogens. Prog. Nat. Sci: Mater. Int. 22(6):693-700.
- Irshad, S., A. Salamat, A.A. Anjum, S. Sana, R.S. Saleem, A. Naheed and A. Iqbal (2018). Green tea leaves mediated ZnO nanoparticles and its antimicrobial activity. Cogent. Chem. 4(1):1469207.
- Irshad, S., M. Riaz, A.A. Anjum, S. Sana, R.S.Z. Saleem and A. Shauka (2020). Biosynthesis of ZnO nanoparticles using Ocimum basilicum and determination of its antimicrobial activity. The. J. Anim. Plant Sci. 30(2):185-191.
- Jeeva, K., M. Thiyagarajan, V. Elangovan, N. Geetha and P. Venkatachalam (2014). Caesalpinia coriaria leaf extracts mediated biosynthesis of metallic silver nanoparticles and their antibacterial activity against clinically isolated pathogens. Ind. Crops. Prod. 52:714-720.
- Jeong, E.S., M. Kang and H.S. Kim (2016). Surface acoustic wave propagation properties with ZnO thin film for thermo-electric sensor applications. J. Nanosci. Nanotechnol. 16(10):10219-10224.
- Kowshik, M., N. Deshmukh, W. Vogel, J. Urban, S.K. Kulkarni and K.M. Paknikar (2002). Microbial synthesis of semiconductor CdS nanoparticles, their characterization, and their use in the fabrication of an ideal diode. Biotechnol. Bioeng. 78(5): 583-588.
- Kumar, V. and S.K. Yadav (2009). Plant mediated synthesis of silver and gold nanoparticles and their applications. J. Chem. Technol. Biotechnol. 84(2): 151-157.
- Li, X., F. Zhang, C. Ma, Y. Deng, Z. Wang, S. Elingarami and N. He (2012). Controllable synthesis of ZnO with various morphologies by hydrothermal method. J. Nanosci. Nanotechnol. 12(3): 2028-2036.
- Liu, Y., L. He, A. Mustapha, H. Li, Z. Q. Hu, M. Lin (2009). Antibacterial activities of zinc oxide nanoparticles against Escherichia coli O157:H7. J. Appl. Microbiol. 107:1193-201
- Malapermal, V., J. N. Mbatha, R. M. Gengan, and K. Anand (2015). Biosynthesis of biometallic Ag- Au nanoparticles Ocimum basilicum (L.) with antidiabetic and antimicrobial properties. Adv. Mater. Lett. 6, 1050-1057.
- Mirhosseini, M. and F.B. Firouzabadi (2013). Antibacterial activity of zinc oxide nanoparticle suspensions on food‐borne pathogens. Int. J. Dairy. Tech. 66(2): 291-295.
- Moghaddam, A.B., M. Moniri, S. Azizi, R.A. Rahim., A.B. Ariff, W.Z. Saad, F. Namvar and M. Navaderi (2017). Biosynthesis of ZnO nanoparticles by a new Pichia kudriavzevii yeast strain and evaluation of their antimicrobial and antioxidant activities. Molecules. 22(6): 872-890.
- Murtaza, H., W.L. Yang, Y. Ju, X. Chu, Y. Wang, Y. Deng, N. Mahmod and Y.L. Hou (2017). Biocompatibility of iron carbide and detection of metals ions signaling proteomic analysis via HPLC/ESI-Orbitrap. Nano. Res. 10(6): 1912-1923.
- Padmavathy, N and R. Vijayaraghavan (2008). Enhanced bioactivity of ZnO nanoparticles-an antimicrobial study. Sci. Technol. Adv. Mater. 9(3): 035004.
- Premanathan, M., K. Karthikeyan, K. Jeyasubramanian and G. Manivannan (2011). Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation Nanomedicine. 7(2): 184-192.
- Qureshi, R and G.R. Bhatti (2009). Folklore uses of Amaranthaceae family from Nara desert Pakistan. Pak. J. Bot. 41(4): 1565-1572.
- Reddy, K.M., K. Feris, J. Bell, D.G. Wingett, C. Hanley and A. Punnoose (2007). Selective toxicity of zinc oxide nanoparticles to prokaryotic and eukaryotic systems. Appl. Phys. Lett. 90(213902): 2139021-2139023.
- Reddy, K.S. and V.M. Reddy (2009). Anti-hyperglycaemic Activity of ethanol extract of Aerva javanica leaves in Alloxan-induced diabetic mice. J. Pharm. Res. 2(7): 1259-1261.
- Reddy, K.R. (2017). Green synthesis, morphological and optical studies of CuO nanoparticles. J. Mol. Struct. 1150: 553-557.
- A.D (2003). Similarities and differences in the responses of microorganisms to biocides. J. Antimicrob. Chemother. 52(5): 750-763.
- Salem, N.M., L.S. Albanna, A.O. Abdeen, Q.I. Ibrahim and A.M. Awwad (2016). Sulfur nanoparticles improves root and shoot growth of tomato. J. Agri. Sci. 8(4): 179-185.
- Sharif, A., E. Ahmed, A. Malik, M. Mukhtar-Ul-Hassan, A. Munawar, A. Farrukh, S.A. Nagra, J. Anwar, M. Ashraf and Z. Mahmood (2011). Antimicrobial constituents from Aerva javanica. J. Chem. Soc. Pak. 33(3): 439-443.
- Sharma, D., J. Rajput, B.S. Kaith, M. Kaur and S. Sharma (2010). Synthesis of ZnO nanoparticles and study of their antibacterial and antifungal properties. Thin. Solid. Films. 519(3): 1224-1229
- Sharmila, G., R.S. Pradeep, K. Sandiya, S. Santhiya, C. Muthukumaran, J. Jeyanthi, N.M. Kumar and M. Thirumarimurugan (2018). Biogenic synthesis of CuO nanoparticles using Bauhinia tomentosa leaves extract: characterization and its antibacterial application. J. Mol. Struct. 1165: 288-292.
- Slman, A.A (2012). Antibacterial activity of ZnO nanoparticle on some gram-positive and gram-negative bacteria. Iraqi. J. Phys. 10(18): 5-10.
- Soliman, M.A (2006). Cytogenetical studies on Aerva javanica (Amaranthaceae). Flora. Medit. 16: 333-339.
- Srinivasa Rao, N and M.V. Basaveswara (2015). Structural and optical investigation of ZnO nanopowders synthesized from zinc chloride and zinc nitrate. Am. J. Mater. Sci. 5(3): 66-68.
- Stanković, A., S, Dimitrijević and D. Uskoković (2013). Influence of size scale and morphology on antibacterial properties of ZnO powders hydrothemally synthesized using different surface stabilizing agents. Colloids. Surf. B: Biointerfaces.102: 21-28.
- Suresh, D., P.C. Nethravathi, R.H. Udayabhanu, H. Nagabhushana and S. C. Sharma (2015). Green synthesis of multifunctional zinc oxide (ZnO) nanoparticles using Cassia fistula plant extract and their photodegradative, antioxidant and antibacterial activities. Mater. Sci. Semicond. Process. 31:446-454.
- Tawale, J., K. Dey, R. Pasricha, K. Sood and A. Srivastava (2010). Modification at lattice scale for an optimized optical response of Alx(ZnO)1-x nanostructures. Thin. Solid. Films. 519(3): 1244-1247.
- Yamamoto, O. (2001). Influence of particle size on the antibacterial activity of zinc oxide Int. J. Inorg Mater. 3(7): 643-646.
- Yan, S., X. Xu, Y. Sheng, H. Sun, J. Wu and L. Tang (2011). Disposable biosensor based on Au nanoparticles modified CdS nanorod arrays for detection Cytochrome C. J. Nanosci. Nanotechnol. 11(12): 10320-10323.
- Yedurkar, S., C. Maurya and P. Mahanwar (2016). Biosynthesis of zinc oxide nanoparticles using Ixora coccinea leaf extract-a green approach. Open. J. Synth. Theo. App. 5(1): 1-14.
- Zhang, F., X. Li, N. He and Q. Lin (2015). Antibacterial properties of ZnO/Calcium alginate composite and its application in wastewater treatment. J. Nanosci. Nanotechnol. 15(5): 3839-3845.
|





