|
Volume 32, No. (4), 2022, August
(Impact Factor 0.570 ; JCR 2022) |
|
ASSESSMENT OF THE STATE OF THE TERRESTRIAL ENVIRONMENT IN SKIKDA REGION (ALGERIA) USING ENZYMATIC ACTIVITY AND ENERGY RESERVE CONTENTS OF LAND SNAILS, CANTAREUS ASPERSUS
N. Zaidi1*, L. Douafer2, A. Hamdani3 and N. E. Soltani3
1Laboratory for the Optimization of Agricultural Production in Subhumid Areas, Faculty of sciences, University of Skikda, 21000-Skikda, Algeria
2Faculty of Sciences, Department of Biology, University of Mila, 43000-Mila, Algeria
3Laboratory of Applied Animal Biology, Department of Biology, Faculty of Sciences, University Badji Mokhtar of Annaba, 23000-Annaba, Algeria
*Corresponding author’s email: n.zaidi@univ-skikda.dz , znedjoua@yahoo.fr
ABSTRACT
Environmental pollution is one of the most significant issues in industrial regions. Land snails are commonly used as suitable organisms to monitor environmental pollution. Therefore, the present study aims to investigate the effects of anthropogenic pollution on biochemical biomarkers in land snails, Cantareus aspersus. Snails were obtained from four sites of northeastern Algeria, namely El-hadaiek, Azzaba, Ben-Azzouz (Skikda city) and El-Kala (E-Taref city). The sites of Skikda city were selected in this study because of their proximity to industrial factories, while the El-Kala site was used as a reference site (control site) for its far location from pollution sources. Moreover, the enzymatic activities of glutathione S-transferase (GST) and acetylcholinesterase (AChE), and the three energy reserves (glycogens, lipids and proteins) were measured in the digestive gland of snails collected from each site during the winter and spring seasons. The results revealed a significant variation in the AChE activity and energy reserves between sites and seasons, along with significant correlationsbetween the enzymatic and non-enzymatic biomarkers. The highest activity of GST and total proteins were observed during spring in snails of Azzaba site, identified as the closest site to potential pollution sources. In addition, AChE activity was significantly decreased during spring, as well as depletion of lipid and glycogen contents with the highest values was recorded in the site of Azzaba. Conclusively, the land snails, C. aspersus are proven as sentinel organisms effectively used in biomonitoring of terrestrial environment in Skikda region.
Keywords: Pollution, Cantareus aspersus, Biomarkers, Energy reserves, Oxidative stress, Skikda city.
INTRODUCTION
Skikda city is located in the Northeast of Algeria (36° 52' 34N, 6° 54' 33E) at 510 km from Algiers (capital of Algeria). It is an excellent industrial and economic center in the country and the capital place of petrochemical products production in the northeastern part of Algeria. Among its most towns, Azzaba which had previously included a mercury factory, but it has been closed over several years due to its serious effect on human health (Megueddem et al., 2004; Benhamza et al., 2008; Seklaoui et al., 2016) and environmental quality (Alioua et al., 2004; Kahoul et al., 2014). Also, Skikda city includes many manufacturing industries, like the processing industry (wood, cork, plastic, and others), metal industry; building materials, mines and quarries, canning, agro-food industry, and textile industry. Despite the important economic role of these industries, they can cause a lot of mischiefs in city dwellers and on the city itself. As a result, Skikda city is threatened by environmental pollution (Fadel et al., 2016; Zeghdoudi et al., 2019), especially aquatic pollution (Kehal et al., 2004; Boutefnouchet et al., 2005).
The land snails, Cantareus aspersus(Syn: Helix aspersa) is the prevalent and widespread gastropod species in northeast Algeria (Larbaa and Soltani, 2013; Douafer and Soltani, 2014), and are used as bioindicatorsof metallic and organic pollution of soil (Gimbert et al., 2006), Owing to its easy accumulation of pollutants, including heavy metals in its body (Viard et al., 2004; Jordaens et al., 2006). Thus, land snails have been suggested as sentinel organisms to biomonitor environmental pollution (Regoli et al., 2006; Mleiki et al., 2017). Whilst the accumulation of pollutants in snail tissues affects their physiology (Berger and Dallinger, 1993; Gomot et al., 2000) and damages the antioxidant activities of many target organs (Ismert et al., 2002). The antioxidants can effectively remove or convert reactive oxygen species (ROS) into non-harmful metabolites, and maintain the cell function and metabolism (Mates, 2000; Li et al., 2003; Zhang et al., 2008). The hazards of the environmental contamination on the organism's bodies are basically assessed by determining the changes in some enzyme markers, in particular acetylcholinesterase (AChE) and glutathione-S-transferase (GST) which are often used as chemical pollution biomarkers. In addition, the alterations of glycogen, proteins, and lipids levels in tissue are used as good biomarkers in evaluating the pollution impact on the organism's health (Drobne et al., 2008; Dutra et al., 2009; Sancho et al., 2009; Javed and Usmani, 2014; Sifi and Soltani, 2018).
In vertebrates, the family of cholinesterases is divided into two groups: acetylcholinesterase and butyrylcholinesterase. Acetylcholinesterase group is involved in the mechanisms of transmission of nervous influx through the body. Also, its inhibition by many neurotoxicants leads to the accumulation of a chemical mediator, acetylcholine, in the synaptic cleft, which thereby maintains a permanent transmission of nerve impulses, and subsequently leads to muscle tetany and death of the organism (Bocquené et al., 1995; Bainy, 2000). Butyrylcholinesterase (BChE) is identified at high concentrations in several organs and tissues, including plasma, liver, lung, intestine, smooth muscle, skin, brain and heart (Massoulie et al., 1993; Cokugras, 2003), however, its physiological function is still unknown (Nicoletet al., 2003). In addition, BChE is a detoxifying enzyme able to trap many xenobiotics before they reach their target sites (Massoulie et al., 1993; Cokugras, 2003; Nicolet et al., 2003) and protect AChE against anticholinesterase substances (Whitaker, 1986). Nevertheless, most invertebrates possess one basic type of cholinesterases (AChEs) (Massoulie et al. 1993) having two different roles: role in the transmission of nervous influx, and analogous role to that of BuChE in vertebrates (Shi et al., 2012). The enzymatic activity of AChEs has been determined in a digestive gland of the land snail, Achatina fulica Bowdich (Manna, 1979; Zaĭtseva and Kuznetsova, 2008), Eobania vermiculata(Itziou and Dimitriadis, 2011) and Cantareus apertus (Mleiki et al., 2015).
Furthermore, glutathione S-transferases (GST) represent a family of multifunctional enzymes (Georges and Buchanan, 1990), involved in many physiological processes, including the detoxification of many xenobiotics (Habig et al., 1974; Lauterburg and Mitchel, 1981) and protection of cell against oxidative stress (Halliwell and Gutteridge, 2007). Indeed, these enzymes catalyse the addition reaction between the xenobiotics and the –SH group of glutathione, generating consequently more hydrophilic substrates by neutralization of the electrophilic sites of xenobiotics (Hayes and Pulford, 1995; Baussant et al., 2009). The utility of GST as biomarkers of chemical pollution in land snails has been well documented (El-Shenawy et al., 2012; Grara et al., 2012;El-Gendyet al., 2019).
Although environmental pollution harmfully affects organism metabolism, in addition to many other effects, pollution monitoring is a necessary part in terms of quantifying pollutant amounts and pollution detection problems. C. aspersus has been commonly used as sentinel species for monitoring metallic pollution (Larba and Soltani, 2014) and soil quality (Bairi et al., 2020) in several sites of Northeast Algeria. Seasonal responses of two biomarkers (GSH and AChE) in this land snail and its relationship with the soil physic-chemical factors were performed in five sites (National Park of El Kala, EL Bouni, Sidi Amar, Nechmaya and Guelma) of Northeast Algeria (Douafer et al., 2020). Since there is no corresponding study has been conducted in Skikda region. Therefore, the present study was undertaken to assess the utility of C. aspersus as a bioindicator in environmental pollution monitoring in some regions of this city. Our hypothesis is that land snails living near to the pollution sources and, exposed directly (inhaling industrial air pollutants) and indirectly (eating the contaminated plant) to a wide variety of pollutants can regulate their antioxidant enzyme activities and energy reserves for protection against the contamination, and therefore, these animals can be important sentinel organisms in a program of terrestrial pollution biomonitoring. To test this hypothesis, we would determine the activity of two biomarker enzymes (AChE and GST), and the content of three biomolecules (carbohydrate, protein, and lipid) in the digestive gland of land snail collected from three sites of Skikda city and El-kala which used as reference. The selected biomarkers were determined in two seasons (winter and spring) to provide sufficient understanding regardingthe effect of seasonal variations on the antioxidant defense system and energy reserves in terrestrial snails.
MATERIALS AND METHODS
Sample collection: Live samples of the land snails, Cantareus aspersus were collected during two seasons (winter and spring) in 2017 from four sites, among which three are located in Skikda city (northeast Algeria) ( El-Hadaiek (36° 49′ 32″ N, 6° 53′ 18″ E, Azzaba (36° 43′ 60″ N, 7° 06′ 00″ E) and Ben-Azzouz (36° 51′ 40″N, 7° 17′ 33″ E)), and were selected according to their proximity to industrial units and other potential pollution sources, including the previous factory of mercury of Azzaba, and cement factory of Hadjar-soud region (about 54km from Skikda city). Meanwhile, the fourth sampling site is El Kala (36° 53′ 48.55″ N, 8° 26′ 36.80″ E) located in E-Taraf city (northeast Algeria) and was used as a control site due to its far location from motorized traffic and other anthropogenic sources of metal contamination.The selected study sites were characterized by a Mediterranean climate with a hot, dry summer and a mild, rainy winter (Remenieras, 1972). In response to these environmental factors, the snails enter in a state of inactivity, aestivation and hibernation, respectively, during hot and cold periods, accompanied bya set of biological modifications to resist environmental stress (Storey, 2002;Reuner et al., 2008). In addition, in this climate (Mediterranean climate), the reproduction of snails is at the end of spring or the beginning of summer. The seasonal temperatures of each site are listed in Table 1. The adult specimens of C. aspersuswere collected in winter during the hibernation and in spring after the resumption of their biological activity,and then were transported to the laboratory. Whilst, 12 specimens (weight 12.85 ± 0.9 g, shell diameter 28.50 ± 1.5 mm) from each site were rapidly killed by deep freezing in liquid nitrogen (Ismert et al., 2001) and the digestive glands were dissected, weighed, frozen and stored at -20 C.
Table 1: seasonal temperatures (C°) recorded in the different studied sites.
| |
El-Hadaik
|
Azzaba
|
Ben-Azzouz
|
El-Kala
|
|
Winter
|
9.1
|
9.5
|
11.5
|
11.9
|
|
Spring
|
18.2
|
20.0
|
20.0
|
19.9
|
Enzymatic activity assays
Acetylcholinesterase activity: AChE activity was determined according to the method of Ellman et al. (1961) as previously described (Zaidi and Soltani, 2010). In brief, samples of digestive gland tissue from each site were homogenized with 1ml solution composed of 38 mg ethylene glycol tetra acetic acid (EGTA), 1 ml Triton X-100 %, 5.845 g NaCl, and 80 ml Tris buffer (0.01 M, pH 7), then the homogenate was centrifuged at 5000 rpm for 5 min, and the supernatant was used for enzyme activity determination. AChE activity was measured at 412 nm every 4 min for a period of 20 min using 5,5′-dithiobis-2-nitrobenzoic acid (DNTB), Tris buffer (0.1 M, pH 7) and acetylthiocholine iodide as substrate.
Glutathione-S-transferase activity: The glutathione-S-transferase activity was analyzed in the snail digestive gland by the method of Habig and Jakoby (1981) as previously described in Zaidi and Soltani (2011). Briefly, the digestive gland was individually homogenized in buffer phosphate (0.1 M, pH 6) and centrifuged at 1300 rpm for 30 min, and the supernatant was kept for the enzymatic determination. GST activity was determined by the addition of supernatant to the mixture GSH-CDNB in phosphate buffer (0.1 M, pH 7) using 1-chloro-2,4 dinitrobenzene (CDNB) as a substrate. Changes in absorbance were measured every 1 min for 5 min at 340 nm.
Biochemical assays: Carbohydrate, lipid and protein contents were extracted from the digestive gland following the procedure of Shibko et al. (1966). Each sample was individually homogenized in 1 ml of trichloroacetic acid (20%) and centrifuged at 5000 g for 10 min. The supernatant was used for the carbohydrates determination in accordance with the method of Duchateau and Florkin (1959) using anthrone as a reagent and glucose as a standard (Cerning-Beroard, 1975). Absorbance was measured at 620 nm. The obtained pellet was mixed with ether and chloroform (1V/1V) and centrifuged at 5000 g for 10 min. The resulted supernatant was used to estimate the lipid contents based on the method of Goldsworthy et al. (1972) using sulphophosphovanillin as a reagent and Sunflower oil (contains more than 99% of triglyceride) as a standard (Ghorab, 2016). Absorbance was read at 530 nm. The resulted pellet was resuspended in 1ml de NaOH (0.1 N), and was used to determining total protein contents according to Bradford (1976) assay using Coomassie brilliant blue (G 250, Merck) as reagent and bovine serum albumin (BSA) as a standard (Dauvillier, 1998). Absorbance was measured at 595 nm. All biochemical assays were expressed as µg/mg wet weight tissue.
Statistical analysis: All data are given as mean ± standard deviation (SD). The normality and homogeneity of variances were respectively tested by Levine’s and Kolmogorov-Smirnov tests. Data were analyzed with one and two-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test. Statistical tests were conducted using Minitab Software (Version 17, State College, and Penn, USA) where p < 0.05 was considered statistically significant.
RESULTS
Changes in the enzymatic activities:As shown in figures 1 and 2, the enzymatic activity of Acetylcholinesterase (AChE) in C. aspersus digestive gland from the polluted sites of Skikda city was significantly lower than that of snails collected from the reference site (El Kala) showing the highest AChE activity (8.039 ± 0.973pM/min/mg protein, and 6.900± 0.874pM/min/mg protein, in winter and spring, respectively). Besides, the lowest AChE activity was observed in the site of Azzaba during winter (4.299 ± 0.415 pM/min/mg protein) and spring (3.620 ± 0.313pM/min/mg protein). Two-way ANOVA (site, season) revealed significant effects of site (F3, 88= 237.07; p<0.001), season (F1, 88 = 151.59; p<0.001) and season-site interaction (F3,88 = 5.71; p= 0.001).Tukey's testshowed a significant difference (p< 0.001) inAChE activity in all the studied sites (El-Hadaiek, Azzaba, Ben-Azzouz and El-Kala) between winterand spring. Hence, the enzymatic activity of AChE activity is overall, high in winter compared to that noticed in spring and differs between sites in the following order: Azzaba< El-hadaiek < Ben-Azzouz < El-Kala. Furthermore, the GST activities were significantly increased in the snails collected from the three sites of Skikda city compared to that of snails collected from the control reference site (El Kala). The higher values of GST activity were found in Azzaba in winter (1.530 ± 0.235 nM/min/mg protein), and in spring (1.729 ± 0.256 nM/min/mg protein). Two-way ANOVA revealed a significant effects of site (F3, 88= 44.13 ; P <0.001) on GST activity, since the season (F1, 88 = 3.51 ; P > 0.05) and site-season interaction (F3, 88= 1.45, p >0.05) show no significant effect.On top of that, Tukey's testshowed no significant change (p> 0.05) in the enzymatic activity of GST between the two seasonsin all selected sites. Of note, the GST activity was found to be higher in spring than that seen in winter, and is different between sites in the following order: Azzaba > El-hadaiek > Ben-Azzouz> El-Kala.
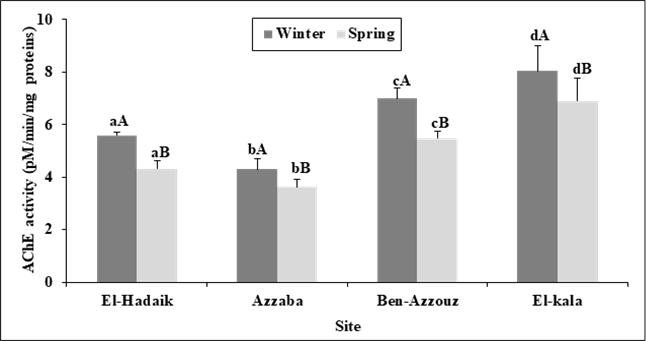
Figure 1. Acetylcholinesterase activity (pM/min/mg protein) in the digestive gland of C. aspersus collected from different sites during winter and spring (2017) (m ± SD, n=12). Analysis of variance (ANOVA) followed by Tukey's test. The lowercase letters compare the values of the same season between the sites and the uppercase letters compare the values of the same site between seasons.
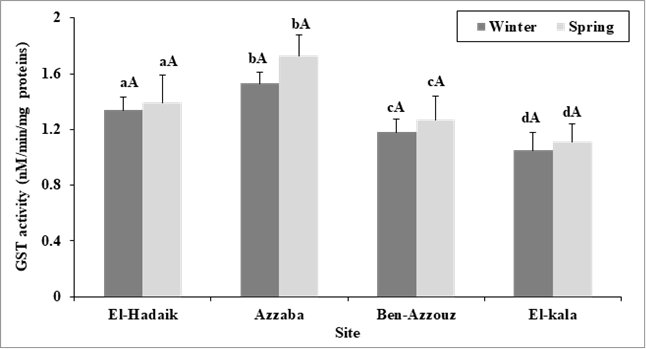
Figure 2. GST activity (nM/min/mg proteins) in the digestive gland of C. aspersus collected from different sites during winter and spring (2017) (m ± SD, n=12). Analysis of variance (ANOVA) followed by Tukey's test. For each season mean values followed by the same letter are not significantly different from each other at p> 0.05). The lowercase letters compare the values of the same season between the sites and the uppercase letters compare the values of the same site between seasons.
Changes in energy reserve contents: The results of glycogen contents showed a significant difference (P < 0.001) between the sampling sites of Skikda city and the control site (El Kala). The lowest values were recorded in Azzaba during the two seasons, while the highest values were observed in El-Kala site in winter (1.015 ± 0.136 μg. mg−1 ww) and spring (1.136 ± 0.143 μg.mg−1 ww). The two-way ANOVA revealed a significant effect of sites (F3, 88 = 120.62; P<0.001) and season-site interaction (F3, 88 = 5.60; P < 0.001), and no significant effect of season (F1, 88 = 1.89; P= 173) in all the study sites (Table 2). Pairwise comparisons (Tukey test) revealed a significant difference (p <0.05) between the two seasonsin the site of Ben-Azzouz and El-Kala, and no significant difference (p > 0.05) in the others sites. The lipid contents were significantly (P < 0.01) decreased in samples of all study sites of Skikda city as compared with that of El Kala. This decrease was significantly marked in the site of Azzaba with levels of 0.537 ± 0.084 μg.mg−1 ww, and 0.502 ± 0.080 μg.mg−1 ww, respectively, in winter and spring. Further, the highest contents of lipid were found in El-Kala site in winter (0.883 ± 0.192 μg.mg−1 ww) and spring (0.714 ± 0.136 μg.mg−1 ww). The contents of lipids were higher in winter than spring. This result was confirmed by two-way ANOVA test, exhibiting a significant effect of site (F3, 88 = 31.29; P < 0.001) and season (F1, 88 =17.11; P < 0.001) (Table 3). Moreover, Tukey's testshowed a significant change (p< 0.05) in lipid contents between the two seasonsin El-Hadaik, Ben-Azzouz and El-Kala, excludingAzzaba site (p=0.313).
Table 2. Glycogen content (μg.mg−1 ww) in the digestive gland of C. aspersus collected from different sites during winter and spring of 2017) (m ± SD, n=12). Analysis of variance (ANOVA) followed by Tukey's test, mean values followed by the same letter are not significantly different from each other at p> 0.05.
| |
El-Hadaik
|
Azzaba
|
Ben-Azzouz
|
El-Kala
|
|
Winter
|
0.634 ± 0.119 aA
|
0.561 ± 0.088 aA
|
0.793 ± 0.085 bA
|
1.015 ± 0.136 cA
|
|
Spring
|
0.546 ± 0.088 aA
|
0.516 ± 0.084 aA
|
0.684 ± 0.098 bB
|
1.136 ± 0.143 cB
|
The lowercase letters compare the averages of the same season between the sites.
The uppercase letters compare the values of the same site between seasons.
Table 3. Total lipids (μg.mg−1 ww) in the digestive gland of C. aspersus collected from different sites during winter and spring (2017) (m ± SD, n=12). Analysis of variance (ANOVA) followed by Tukey's test, mean values followed by the same letter are not significantly different from each other at p> 0.05
| |
El-Hadaik
|
Azzaba
|
Ben-Azzouz
|
El-Kala
|
|
Winter
|
0.627 ± 0.056 aA
|
0.537 ± 0.084 aA
|
0.657 ± 0.044 aA
|
0.883 ± 0.192 bA
|
|
Spring
|
0.567 ± 0.068 aB
|
0.502 ± 0.080 aA
|
0.571 ± 0.081 aB
|
0.714 ± 0.136 bB
|
The lowercase letters compare the averages of the same season between the sites.
The uppercase letters compare the values of the same site between seasons.
In the present study, a significant increase in total protein levels was observed in the digestive gland of snails collected from the three sites of Skikda city as compared with those of snails collected from a reference site (El Kala). Total protein contents were significantly higher in Azzaba in winter (2.191 ± 0.189 μg.mg−1 ww) and spring (2.480 ± 0.279 μg.mg−1 ww). The two-way ANOVA revealed a significant effect of site (F3, 88 = 77.57; P < 0.001) and season (F1, 88 = 21.44; P < 0.001), along with no significant effect of season-site interaction (F3, 88 = 2.02; P > 0.05) (Table 4). Tukey's testshowed a significant change (p< 0.05) in total protein contents between the two seasonsin El-Hadaik, Ben-Azzouz, and Azzaba, since El-Kala site showed no significant change in this parameter (p >0.05).
Table 4. Total protein (μg.mg−1 ww) in the digestive gland of C. aspersus collected from different sites during winter and spring (2017) (m ± SD, n=12). Analysis of variance (ANOVA) followed by Tukey's test, mean values followed by the same letter are not significantly different from each other at (p> 0.05).
| |
El-Hadaik
|
Azzaba
|
Ben-Azzouz
|
El-Kala
|
|
Winter
|
1.983 ± 0.171 aA
|
2.191 ± 0.189 bA
|
1.736 ± 0.136 cA
|
1.596 ± 0.127 cA
|
|
Spring
|
2.184 ± 0.185 aB
|
2.480 ± 0.279 bB
|
1.856 ± 0.127 cB
|
1.648 ± 0.128 cA
|
The lowercase letters compare the averages of the same season between the sites
The uppercase letters compare the values of the same site between seasons
Correlation between the enzyme activities (AChE and GST) and the energy reserve contents (glycogen, lipids and protein) in C. aspersus.
In table 5, statistical analyses of the correlation between the enzyme activities and the energy reserve contents in C.aspersus digestive gland recorded in all study sites showed a highly significant negative correlation between AChE activity and protein contents (p <0.001), and between GST activity and the contents of glycogen and lipids in all sites (p <0.001). Meanwhile, a highly significant correlation between GST activity and protein contents (p <0.001), and between AChE activity and the lipid contents (p <0.001) were observed in all sites. Besides, we noted a highly significant correlation between AChE activity and glycogen content (p <0.001) in Azzaba and Ben-Azzouz. Also, AChE activity was significantly (p <0.01) correlated with glycogen content in El-Hadaik site.
Table 5. Relationship between the AChE and GST activities and the glycogen, lipids and proteins content in C. aspersus. (R = coefficient of correlation, p = significance level)
|
Enzyme/site
|
|
El-Hadaik
|
Azzaba
|
Ben-Azzouz
|
El-Kala
|
| |
|
AChE
|
GST
|
AChE
|
GST
|
AChE
|
GST
|
AChE
|
GST
|
|
Glycogen
|
R
|
0.597
|
-0.823
|
0.753
|
-0.826
|
0.723
|
-0.723
|
0.277
|
-0.747
|
|
P
|
0.002
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
0.190
|
0.000
|
|
lipids
|
R
|
0.634
|
-0.813
|
0.693
|
-0.761
|
0.780
|
-0.819
|
0.827
|
-0.798
|
|
P
|
0.001
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
|
proteins
|
R
|
-0.661
|
0.859
|
-0.842
|
0.956
|
-0.703
|
0.829
|
-0.790
|
0.954
|
|
P
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
0.000
|
DISCUSSION
Mollusks, owing to their typical properties, including wide environmental distribution, easy sampling, stress tolerance and ability to accumulate contaminants are mainly used in environmental risk and pollution assessment (Laskowski and Hopkin, 1996;Beeby and Richmond, 2002; Regoli et al., 2005), such as land snails are considered as an effective biomonitor organism (Anim et al., 2011;Itziou and Dimitriadis, 2011;Vega et al., 2012; Radwan et al., 2020). The validity of land snails,Cantareus aspersus as bioindicator organisms of terrestrial pollution in the region of Skikda was assessed by determination of a battery of digestive gland enzymatic and non-enzymatic biomarkers. Hence, the digestive gland was selected, in this study, as the main pollutant-target organ for the biochemical evaluation due to its ability to accumulate various pollutants, and its effective involvement in detoxification processes (Berandah et al., 2010). This organ possesses an efficient antioxidant status and exhibits a model of free radical injury (Ramos-Vasconcelos et al., 2005). The use of biochemical alterations of gastropods digestive glands as biomarkers of xenobiotics contamination have been investigated (Snyman et al., 2005; Radwan et al., 2008; Attia et al., 2021)
In the present study, some biomarkers in the digestive gland of these snails, including enzymatic biomarker of neurotoxicity and immunotoxicity (AChE), enzymatic antioxidant (GST), non-enzymatic antioxidants (glycogen, total lipid and total protein) were determined. Previous studies have identified AChE as a biomarker of chemical exposure such as pesticides (Coeurdassieret al., 2002), and heavy metals (Dallinger et al., 2004: de Vaufleury, 2015). The enzymatic activity of AChE of snails collected from the three sites of Skikda city is lower than that of the control site (El-Kala), indicating thus the presence of pollution in the investigated areas. In addition, AChE activity was found to be significantly lower in snails of Azzaba as compared to the other sites. The site of Azzaba is known for its proximity to potential pollution sources (factory of mercury) and thus is classified as the most polluted site. Furthermore, the site of Ben-Azouz is located near the cement factory of Hadjar-Soud, generating contamination by the cement dust. Cement dust of Hadjar-Soud constitutes numerous types of metal oxides including calcium oxide (CaO), silicon oxide SiO2, aluminum trioxide Al2O3, ferric oxide Fe2O3, and other components (Mekti, 2018). In accordance with this result, the decrease in AChE activity was reported in the head of Cantareus aspersus collected from the same north-east Algerian sites compared to snails of the reference site (El Kala). This is likely explained by the metal pollution with high concentrations of Fe, Al2O3, MgO and FeO (Douafer et al., 2020) and Fe, Mn, Pb, and Cd (Larba and Soltani, 2014). Bairi et al. (2020) have reported inhibition of AChE activity in C. aspersus collected from the site of El Hadjar. In our study, the inhibition of AChE activity observed in snail’s digestive gland from the polluted site may be due to the neurotoxic or immunotoxic effects, or both, of pollutants against this land snail (Mleiki et al., 2015). Zaitseva and Kuznetsova (2008) reported that the activity of AChE in digestive gland tissue was found in the intraepithelial nerve plexus of the gut and the digestive gland and in the nerve fibers and subepithelial neurons located in muscle cells. It was also detected in the haemocytes of blood vessels and connective tissue (Moore, 1991). Similar to our result, a decrease of AChE activity was also observed in the digestive tractofC. aspersus treated with ferric oxide (Besnaci et al., 2019), mercury (Leomanni et al., 2016), and pesticides (Radwan andMohamed, 2013).
GST is an important stress marker in heavy-metals exposed land snails (El-Shenawy et al., 2012; Abdel-Halimet al., 2013). In the present study, GST activity of snails collected from the polluted sites of Skikda city was significantly increased in comparison with other snails of the reference site (El-Kala). The increase of GST activity refers to the organism's adaptation to the increased pollution stress (Astani et al., 2012). Hence, the increase of GST activity could be due to the pollutants-inducing the activation of the natural antioxidant defense systems (Elia et al., 2007). Our result coincides with that of Larba and Soltani (2014) who reported that GST activity was significantly increased in the digestive gland of the land snails C. aspersus collected from several sites in Northeast Algeria. An activation of GST has been also reported in this species collected from the same sites exposed to metallic pollution (Bairi et al., 2020; Abdel-Halim et al., 2013). Also, induction of GST was observed in the digestive gland of the land snails Eobania vermiculata collected from the metal-polluted areas (De Souza et al., 2015).
The measure of energy reserves is of potential interest for assessing the health quality of terrestrial and aquatic ecosystems (El-Shenawy et al., 2012; Bislimi et al., 2013; Sifi and Soltani, 2018). The findings showed also a significant difference between the study sites in the levels of glycogen, lipids, and proteins of the land snail digestive gland. Furthermore, the glycogen and lipid levels were significantly declined in snails of Azzaba site when compared with those of the reference site (El-Kala). Carbohydrates are the first and immediate source of energy in living organisms (Umminger, 1977), and are importantly involved in detoxification, biotransformation, and excretion of toxicants (Wiens and Gilbert, 1967). Noteworthy, the stress condition can lead to a decrease in the carbohydrate reserves to meet the energy demands (Arasta et al., 1996). Also, the results revealed a decrease in the level of carbohydrates of the digestive gland in snails collected from polluted sites as compared to the reference site (El Kala). Depletion of glycogen in this study is likely due to its mobilization to satisfy the energy demand during stressful conditions. This may result in the direct use of this compound for energy production (Ribeiro et al., 2001) or from an increased rate of glycogenolysis (Radwan et al., 2008). Among the overall carbohydrate molecules, especially glycogenand lipids are mainly utilized to overcome stress. Lipids serve as a highly reduced storage form of energy and play an important role in the structure of cell membranes and intracellular organelles (Kandil et al., 2009). In addition, total lipids in animals from Azzaba site were significantly lower than those found in animals of the reference site (El-Kala). This result might be explained by a drastic depletion of glycogen storage in the snail digestive gland, where lipid contents may be used for energy production (Radwan et al., 2008). Also, the decrease of total lipid may be due to the increased enzymatic activity of lipase involved in the breakdown of lipid (Merad et al., 2020) or reduction in synthesizing capacity of lipid (Abd-El-All, 2004)). It is reported that during chronic stress, lipid constitutes another source of energy offered to tissues when needed after carbohydrates (Moussard, 1999). Moreover, proteins are not only essential in tissue building and repair but also can, during chronic periods of stress, supply energy in metabolic activities and biochemical reactions. Our findings revealed significant differences in the total proteins (TP) between the study sites. TP content was significantly increased in the digestive gland of snails collected from Azzaba site as compared to El-kala (reference site). The increase in total protein level noticed in the present study could be attributed to the increase of protein synthesis to supply the increased energy requirement for body use. El-Shenawy et al. (2012) reported an increase in energy demand in the snail digestive gland under stress conditions to detoxify the toxicants and to overcome the induced stress. It is reported that Carbohydrates and lipid contents are limited in snail and the corresponding alternative source of energy to satisfy the increased energy demand in proteins (Radwan et al., 2008). Moreover, the increase in total protein amount of the snail digestive gland may be partly due to the destruction of cell organelles of snails collected from polluted sites that may lead to the inhabitation of protein synthesis (Eissa et al., 2002). Similarly to our results, depletions in glycogen and lipid contents with an increase in proteins were also observed in land snails after exposure to metal dust collected at the level of the iron and steel complex of EL-Hadjar (Eastern Algeria) (Grara et al., 2012), pesticides (Aït Hamlet et al., 2012) and Weatfert fertilizer (Attia et al., 2021). On the other hand, El-Shenawy et al. (2012) observed an increase in the level of lipid and decrease in the protein content in the digestive gland of E. vermiculata snails collected frompolluted areas as compared to non-polluted areas. The decrease in total protein reserves may be explained by the imbalance between the rate of protein synthesis and degradation.
Our results showed a positive correlation between AChE activity and metabolism of glycogen and lipids, and between GST activity and protein content, while, a negative correlation was noted between AChE and protein, and between GST and the amount of glycogen and lipid. Importantly, the digestive gland of the land sails is the principal organ of pollutants detoxification, requiring high energy, and hence the energy content would be reduced in this organ.
It is reported that in land snails C. aspersusseasonal variations can influence the capacity of the antioxidant defense system (Ramos-Vasconcelos et al., 2005) and energy reserves (Silva et al., 2012). Further, the AChE and carbohydrate and lipid levels were decreased, since protein contents in the digestive gland of C. aspersus collected in spring compared to winter were increased. In addition, we noted no significant difference in the GST activity between the two seasons. This may be attributed to the climate change between seasons (winter and spring), physiological conditions (hibernation aestivation), and reproductive cycle. Of note, these sites are characterized by a Mediterranean climate (Remenieras, 1972). The precipitation (humidity) and temperature are important environmental climatic factors (Ramade, 2003) able to affect the species distribution (Faurie et al., 2006; Somero, 2010) and vital functions of organisms by resulting in changes in biochemical and physiological processes (Somero, 1995; Kingsolver and Huey, 2008). Several studies (Dellali et al. 2001; Singh et al., 2009; Itziou and Dimitriadis, 2012) have suggested that AChE activity is related to seasonal variability and extreme physiological conditions such as hibernation and aestivation. Inhibition of AChE activity in C. aspersus has been demonstrated in spring as compared to winter (Bairi et al., 2020; Douafer et al., 2020). Snails can regulate their defense system seasonally in response to environmental factors such as temperature and humidity (Nowakowska et al., 2010; Staikou et al., 2016). In contradiction of our results, Bairi et al. (2020) and Larba and Soltani, (2014) have reported a seasonal variation in GST activity with an increase of GST activity in the spring and summer compared to winter. Energy reserves are considered as biomarkers of stressful xenobiotic exposure (lagadic et al., 1994), but their storage and variation are closely linked to environmental factors (Rosa et al., 2005). In contrast to our results, the highest values of glycogen and lipid were found in the digestive gland of Megalobulimus oblongus in spring compared to those found in winter (Da Silva and Zancan, 1994). These authors suggest that during hibernation, glycogen in tissues constitutes the major source of energy, while the amount of lipid in the digestive gland (hepatopancreas) remains constant in autumn and winter. The changes in the biochemical composition of the digestive gland are related to the reproductive cycle. Reductions of reserves are noted during spring, due likely to the mobilization of these reservesfor reproduction and aestivation (Da Silva and Zancan, 1994). The results of this study may be explained by increasing or decreasing the proximity to pollution sources (factory of mercury and cement factory of Hadjar-Soud).
Conclusion: This study provides original data on some enzymatic and non-enzymatic biomarkers in the digestive gland of land snails, Cantareus aspersus, collected from three sites in Skikda city, and El Kala town (Taref city, northeast Algeria) during winter and spring seasons. The results showed an alteration in the enzymatic activity of AChE and GST, and the energy reserves (glycogen, lipids and proteins) of snails from the sites located in the proximity of pollution sources as compared to the reference site (El Kala). A significant variation between seasons was noted in the levels of all measured biomarkers, except the GST activity. Also, the biological data suggest that Azzaba is identified as the most contaminated site because the study site is located near the factory of mercury which has been closed over several years. Further, C. aspersus is considered an efficient bioindicator of environmental pollution in Skikda region. These species can reduce oxidative damage by developing a defense system leading to remove or transform ROS into less toxic metabolites. In addition, changes in activities of AChE and GST and energy reserves can be used as environmental biomarkers in terrestrial animals exposed to cement dust, metals, and other contaminant sin field condition. For all these reasons, we recommended the use of snail C. aspersus as a sentinel organism of environmental pollution by chemical pollutants that might be present in the Skikda region. Other studies must be conducted in this region to complete the present findings like the analysis of metallic elements in soil and tissues of snails and the determination of histological and molecular effects of pollutants in this species.
Acknowledgments: This study was supported by the Algerian Fund for Scientific Research and by the Ministry of Higher Education and Scientific Research of Algeria (PRFU Projet to Pr. N. ZAIDI).
Conflicts of interest: No competing interests.
Ethics approval: The experimental studies were performed according to the WHO guidelines (1965).
REFERENCES
- Abd-El-All, S.M. (2004). Toxicity and biochemical response of Eobania vermiculata land snail to Niclosamide molluscicide under laboratory and field conditions. J. Agric. Sci. 29: 4751-4756.
- Abdel-Halim, K.Y., A.A. Abdo El-Saad, M.M. Talha, A.A. Hussein and N.M. Bakry (2013). Oxidative stress on land snail Helix aspersa as a sentinel organism for ecotoxicological effects of urban pollution with heavy metals. Chemosphere93(6): 1131-1138.
- Adrian D.J., J. Potus, A. Poiffait and P. Dauvillier (1998). Introduction à l’analyse nutritionnelle des denrées alimentaires. Tec & Doc Lavoisier (France). 254 p
- Aït Hamlet, S., S. Bensoltane, M. Djekoun, F. Yassi and H. Berrebbah (2012). Histological changes and biochemical parameters in the hepatopancreas of terrestrial gastropod Helix aspersa as biomarkers of neonicotinoid insecticide exposure. Afri. J. Biotech. 11(96): 16277-16283.
- Alioua, A., F. Semadi and H. Benrrabeh (2004). Cartography of mercurial pollution using bioaccumulators in the area of Azzaba (Eastern Algeria). C. R. Acad. Bulg. Sci. 57(2): 85-90.
- Anim, A.K., M. Ackah, J.R. Fianko, L. Kpattah, J. Osei, Y. Serfor-Armah and E.T. Gyamfi (2011). Trace elements composition of Achatina achatina samples from the Madina Market in Accra, Ghana. Res. J. Environ. Earth sci. 3(5): 564-570.
- Arasta, A.T., V.S. Bais and P. Thakur (1996). Effect of Nuvan on some biological parameters of Indian cat fish, (Mystus vittatus). J. Environ. Biol. 17(2):167-169.
- Astani, M., A.R. Vosoughi, L. Salimi and M. Ebrahimi (2012). Comparative study of heavy metal (Cd, Fe, Mn, and Ni) concentrations in soft tissue of gastropod Thais mutabilis and sediments from intertidal zone of Bandar Abbas. Adv. Environ. Biol. 6: 319-326.
- Attia, L., S. Tine, F. Tine-Djebbar and N. Soltani (2021). Potential hazards of an inorganic fertilizer (Weatfert) for the brown garden snail (Eobania vermiculata Müller, 1774): growth, histological and biochemical changes and biomarkers. Appl. Ecol. Environ. Sci. 19(3):1719-1734.
- Bainy, A.C.D. (2000). Biochemical responses in Peneids caused by contaminants. Aquacul. 191: 163-168.
- Bairi, Y., K. Sifi and S. Kilani-Morakchi (2020). Biomarkers response in the snail Cornu aspersum (Gastropoda, Helicidae) used as bioindicator of soil pollution in extreme Northeast of Algeria. Eco. Env. & Cons. 26(1): 47-54.
- Baussant, T., R.K. Bechmann, I.C. Taban, B.K. Larsen, A.H. Tandberg, A. Bjørnstad S. Torgrimsen, A. Naevdal, K.B. Øysaed, G. Jonsson and S. Sanni (2009). Enzymatic and cellular responses in relation to body burden of PAHs in bivalve molluscs: A case study with chronic levels of North Sea and Barents Sea dispersed oil. Mar. Poll. Bull. 58: 1796-1807.
- Beeby, A. and L. Richmond (2002). Evaluating Helix aspersa as a sentinel for mapping metal pollution. Ecol. Indic. 1(4):261-270.
- Benhamza, M., N. Kherici, F. Picard-Bonnaud and A. Nezzal (2008). Qualité des eaux souterraines de la zone mercurielle nord numidique (Azzaba), nord est Algérie. Évaluation de la contamination de la population par le mercure inorganique. Bul. Serv. Géo. N. 19(2): 135-149.
- Berandah, F.E., Y.C. Kong and A. Ismail (2010). Bioaccumulation and distribution of heavy metals (Cd, Cu, Fe, Ni, Pb and Zn) in the different tissues of Chicoreus capucinus Lamarck (mollusca: muricidae) collected from Sungai Janggut, Kuala Langat, Malaysia. EnvironmentAsia. 3(1): 65-71.
- Berger, B. and R. Dallinger (1993). Terrestrial snails as quantitative indicators of environmental metal pollution. Environ. Monit. Assess. 25(1): 65-84.
- Besnaci, S., S. Bensoltane and M. Djekoun (2019). Stress oxydatif et changements histologiques induitent par les nano-fe2o3 chez Helix aspersa. Sci. Study Res. Chem. & Chem. Eng. Biotec. F. Indus. 20(2):119-133.
- Bislimi , K., A. Behluli , J. Halili , I. Mazreku , F. Osmani , F. Halili (2013): Comparative analysis of some biochemical parameters in hemolymph of garden snail (Helix pomatia L.) of the Kastriot and Ferizaj regions, Kosovo. Int. J. Eng. Appl. Sci. 4(6): 11-18.
- Bocquené, G., C. Bellanger, Y. Cadiou and F. Galgani (1995). Joint action of combinations of pollutants on the acetylcholinesterase activity of several marine species. Ecotoxicol. 4:266-279.
- Boutefnouchet, N., N. Bouzerna and H. Chettibi (2005). Assessment of the petrochemical industry pollution on the Skikda bay, Algeria. Int. J. Environ. Res. Public Health. 2(3): 463-468.
- Bradford, M.M. (1976). A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principal of protein-dye binding. Anal. Biochem. 72(1-2): 248-254.
- Cerning-Beroard, J. (1975). The use of invertase for determination of sucrose. Application to cereals, cereal products and other plant materials. Cereal. chem. 52: 431-438.
- Coeurdassier, M., A. Gomot-De Vaufleury, M. Saint-Denis, D. Ribera, J.F. Narbonne and P.M. Badot (2002). Effects of dimethoate on snail B-esterase and growth as a function of dose, time and exposure route in a laboratory bioassay. Biomarkers. 7(2):138-50.
- Cokugras, A.N. (2003). Butyrylcholinesterase: Structure and Physiological Importance. Turkish J. Biochem. 28(2):54-61.
- Da Silva, R.S. M. and D.M. Zancan (1994). Seasonal variation of the carbohydrate and lipid metabolism in a land pulmonate gastropod, Megalobulimus oblongus, omp. Biochem. Physiol. 108A (2-3) : 337-341.
- Da Silva, R.S.M. and D.M. Zancan (1994). Seasonal variation of the carbohydrate and lipid in a land pulmonate gastropod, Megalobulimus oblongus. Comp. Biochem. Physiol. 108: 337-341.
- Dallinger R (1994). Invertebrate organisms as biological indicators of heavy-metal pollution. Appl. Biochem. Biotechnol. 48(1): 27-31.
- De Souza, M.R., F.R. da Silva, C.T. de Souza, L. Niekraszewicz, J.F. Dias, S. Premoli, D.S. Corrêa, C. Soares Mdo, N.P. Marroni, M.I. Morgam-Martins and J. da Silva (2015). Evaluation of the genotoxic potential of soil contaminated with mineral coal tailings on snail Helix aspersa. Chemosphere. 139 : 512-517.
- de Vaufleury, A. (2015). Landsnail for ecotoxicological assessment of chemicals and soil contamination – ecotoxicological assessment of chemicals and contaminated soils using the terrestrial snail, Helix aspersa, at various stage of its life cycle: A Review. Ecol. Indic. 365-391.
- Dellali, M., M. Gnassia-barelli, M. Romeo and P. Aissa (2001). The use of acetylcholinesterase activity in Ruditapes decussatus and Mitillus galloprovincialis in the biomonitoring of Bizerta lagoon. Comp. Biochem. Physiol. 130(2): 227-235.
- Douafer, L. and N. Soltani (2014). Inventory of land snails in some sites in the Northeast Algeria: Correlation with soil characteristics. Adv. Environ. Biol. 8(1): 236-243.
- Douafer, L., N. Zaidi and N. Soltani (2020). Seasonal variation of biomarker responses in Cantareus aspersus and physic-chemical properties of soils from Northeast Algeria. Environ. Sci. Pollut. Res. 27:24145-24161.
- Drobne, D., M. Blažič, C.A.M. Van Gestel, V. Lešer, P. Zidar, A. Jemec and P. Trebše (2008). Toxicity of imidacloprid to the terrestrial isopod Porcellios caber (Isopoda, Crustacea). Chemosphere. 71(7) :1326-1334.
- Duchateau, G.H. and M. Florkin (1959). Sur la tréhalosémie des insectes et sa signification. Arch. Insect Biochem. Physiol. 67: 306-314.
- Dutra, B.K., F.A. Fernandes, A.L. Lauffer and G.T. Oliveira (2009). Carbofuran-induced alterations in the energy metabolism and reproductive behaviors of Hyalella castroi (Crustacea, Amphipoda). Comp. Biochem. Physiol. Part - C: Toxicol. Pharmacol. 149(4): 640-646.
- Eissa, S.H., E.T. Rizk, A.E. Abou-Shafey, M.H. Mona and A. Atlum (2002). Toxicological effect on Euphorbia peplus water suspension on heamocytes of the fresh water snails, Biomphalaria alexandrina and Lanistes carinatus. Proc LCBS 2: 417-447.
- El-Gendy, K.S., M.A. Radwan, A.F. Gad, A.E. Khamis and E.H. Eshra (2019). Physiological traits of land snails Theba pisana as simple endpoints to assess the exposure to some pollutants. Environ. Sci. Pollut. Res. 26: 6922-6930.
- Elia, A.C., R. Galarini, A.J.M. Dorr and M.I. Taticchi (2007). Heavy metal contamination and antioxidant response of a freshwater bryozoan (Lophopus crystallinus Pall., Phylactolaemata). Ecotoxicol. Environ. Saf. 66(2):188-194.
- Ellman, G.L., K.D. Courtney, V.Jr. Andres and R.M. Feather-stone (1961). A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7(2): 88-95.
- El-Shenawy, N.S., A. Mohammadden and Z.H. Al-Fahmie (2012). Using the enzymatic and non-enzymatic antioxidant defense system of the land snail Eobania vermiculata as biomarkers of terrestrial heavy metal pollution. Ecotoxicol. Environ. Saf. 84(1):347-354.
- Fadel, D., N. Hadjoudja, B.E. Badouna and R. Djamaï (2016). Bio-qualitative estimation of diffused air pollution of a city in North-eastern Algeria by using epiphytic lichens. Int. J. Soc. Res. Methodol.Soci. Sci. 2(3):34-43.
- Faurie, C., C. Ferra, P. Medori, J. Devaux and J.L. Hemptinne (2006). Écologie, Approche scientifique et pratique, ed., Tec et Doc, Paris (France). 488 p
- Georges, G.S. and G. Buchanan (1990). Isolution, properties and inducation of piace liver cytosolic glutathione S-trnsferase. Fish. Physiol. Biochem. 8(6): 437-449.
- Ghorab, I. (2016). Evaluation de la valeur nutritionnelle et effets de facteurs environnementaux chez les crustacés. Thesis (published). Deptt. of Biology, Univ. of Annaba, Algeria.
- Gimbert, F., A. De Vaufleury, F. Douay, R. Scheifler, M. Coeurdassier and P.M. Badot (2006). Modelling chronic exposure to contaminated soil: a toxicokinetic approach with the terrestrial snail Helix aspersa. Environ. Int. 32(7): 866-875.
- Goldsworthy, G.J., W. Mordue and J. Guthkelch (1972). Studies on insect adipokinetic hormones. Gen. Comp. Endocrinol. 18(3): 545-551.
- Gomot de Vaufleury, A. and F. Pihan (2000). Growing snails used as sentinels to evaluate terrestrial environment contamination by trace elements. Chemosphere, 40(3):275-284.
- Grara, N., A. Atailia, M. Boucenna, F. Khaldi, H. Berrebbah and M.R. Djebar (2012). Effects of heavy metals on the snail Helix aspersa bioindicators of the environment pollution for human health. Int. Conf. Appl. Life Sci. 241-246.
- Habig, W.H. and W.B. Jakoby (1981). Glutathione S-transferase (rat and human). Meth. Enzymol. 77: 218-231.
- Habig, W.H., M.J. Pabst and W.B. Jakoby (1974). Glutathione S-transferases the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249: 7130-7139.
- Halliwell, B. and J.M.C. Gutteridge (2007). Free radicals in biology and medicine. 4 th Ed. OUP Oxford. Clarendon (Canada). 888 p
- Hayes, J.D. and D.J. Pulford (1995). The glutathione S-transferase supergene family: regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit. Rev. Biochem. Mol. Biol. 30(6): 445-600.
- Ismert, M., T. Oster and D. Bagrel (2002). Effects of atmospheric exposure to naphthalene on xenobiotic-metabolising enzymes in the snail Helix aspersa. Chemosphere. 4(2):273-280.
- Itziou, A and V.K. Dimitriadis (2011). Introduction of the land snail Eobania vermiculata as a bioindicator organism of terrestrial pollution using a battery of biomarkers. Sci. Total. Environ. 409(6): 1181-1192.
- Itziou, A. and V.K. Dimitriadis (2012). Multiple pollution biomarker application on tissues of Eobania vermiculata during two periods characterized by augmented and reduced snail activity. Ecotoxicol. Environ. Saf. 86: 13-22.
- Javed, M. and N. Usmani (2014). Stress response of biomolecules (carbohydrate, protein and lipid profiles) in fish Channa punctatus inhabiting river polluted by Thermal Power Plant effluent. Saudi J. Biol. Sci. 22(2): 237-242.
- Jordaens, K., H. De Wolf, B. Vandecasteele, R. Blust and T. Backeljau (2006). Associations between shell strength, shell morphology and heavy metals in the land snail Cepaea nemoralis (Gastropoda, Helicidae). Sci. Total. Environ. 363(1-3): 285-293.
- Kahoul, M., A. Alioua, N. Derbal and W. Ayad (2014). Behavior of soil micromycetes regarding the mercury pollution in the area of Azzaba (Algeria). J. Mater. Environ. Sci. 5(5): 1470-1476.
- Kandil, M. A., H. I. El-Deeb, S. A. Mobarak and E. A. Eweis (2009). Biochemical impacts of methomyl and abamectin and their binary mixtures with acetylsalicylic acid against the land snails Eobania vermiculata and Monacha obstructa. Zeits. Fur. Die. Ges. Experim. Med. 135: 545-561.
- Kehal, M.A, A. Mennour, L. Reinert and H. Fuzellier (2004). Heavy metals in water of the Skikda Bay. Environ. Technol. 25(9): 1059-65.
- Kingsolver, J.G. and R.B. Huey (2008). Size, temperature, and fitness: three rules. Evol. Ecol. Res. 10: 251-268.
- Lagadic, L., T. Caquet and F. Ramade (1994). The role of biomarkers in environmental assessment. Invertebrate population and communities. Ecotoxicol. 3: 1 93-208.
- Larbaa, R. and N. Soltani (2013). Diversity of the terrestrial gastropods in the Northeast Algeria: spatial and temporal distribution. Eur. J. Exp. Biol. 3(4): 209-215.
- Larba, R. and N. Soltani (2014). Use of the land snail Helix aspersa for monitoring heavy metal soil contamination in Northeast Algeria. Environ. Monit. Assess. 186(8): 4987-4995.
- Laskowski, R. and S.P. Hopkin (1996). Accumulation of Zn, Cu, Pb and Cd in the garden snail (Helix aspersa): implications for predators. Environ. Pollut. 91(3): 289-297.
- Lauterburg, D.J. and J.R. Mitchel (1981). Gillette, bromobenzene-induced liver nicrosis, protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatic intermediate. Pharmacol.11:151-169.
- Leomanni, A., T. Schettino, A. Calisi and M.G. Lionetto (2016). Mercury induced haemocyte alterations in the terrestrial snail Cantareus apertus as novel biomarker. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 183-184: 20-27.
- Li, X.Y., Y.D. Liu, L.R. Song and H.T. Liu (2003). Responses of antioxidant systems in the hepatocytes of common carp (Cyprinus carpio L.) to the toxicity of microcystin-LR. Toxicon. 42(1): 85-89.
- Manna, B. (1979). Acetylcholinesterase in the central nervous system and digestive gland of Achatina fulica Bowdich. Acta. Biol. Acad. Sci. Hung. 30(4):373-80.
- Massoulie, J., L. Pezzementi, S. Bon, E. Krejci and F.M. Vallette (1993). Molecular and cellular biologyof cholinesterases. Prog. Neurobiol. 41(1):31-91.
- Mates, J. M. (2000). Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology. 153(1-3): 83-104.
- Megueddem, M., H. Belmahi, M. Azzouz and M. Reggabi (2004). Quantitative determination of blood mercury in professionally exposed subjects. Rev. Francoph. Lab. 363: 41-46.
- Mekti, Z. (2018). Comportement du laitier de l’usine sidérurgique d’El-Hadjar sur le mélange clinker : Cas ERCE Hadjar-Soud, Skikda. Thesis. Deptt. of Mining. Univ. Annaba, Algeria.
- Merad, I. and N. Soltani (2017). Sublethal effects of cadmium on energy reserves in the edible Mollusk Donax trunculus. J. Entomol. Zool. Stud. 5(1): 100-105.
- Mleiki, A., I. Marigómez and N. Trigui (2015). Effects of Dietary Pb and Cd and their Combination on Acetyl Cholinesterase Activity in Digestive Gland and Foot of the Green Garden Snail, Cantareus apertus (Born, 1778). Int. J. Environ. Res. 9(3): 943-952.
- Mleiki, A., I. Marigómez and N. Trigui (2017). Green garden snail, Cantareus apertus, as biomonitor and sentinel for integrative metal pollution assessment in roadside soils. Environ. Sci. Pollut. Res. 24(31): 24644-24656.
- Moore, M.N. (1991). Lysosomal changes in the response of molluscan hepatopancreatic cells to extracellular signals. Histochem. J. 23: 495-500.
- Moussard, C. (1999). La biochimie, Biochimie structurale et métabolique. Médecine, Pharmacie, Sciences. De Boek, Bruxelles. 294 p.
- Nicolet, Y., O. Lockridge, P. Masson, J.C. Fontecilla-Camps and F. Nachon (2003). Crystal structure of human butyrylcholinesterase and of its complexes with substrate and products. J. Biol. Chem. 278: 41141-41147.
- Nowakowska, A., M. Caputa and J. Rogalska (2010). Natural aestivation and antioxidant defence in Helix pomatia: effect of acclimation to various external conditions. J. Mollus. Stud.76(4): 354-359.
- Oliveira, M.M., M.V. Silva Filho, F.C. Fernandes and J. Cunha Bastos (2007). Brain acetylcholinesterase as a marine pesticide biomarker using Brazilian fishes. Mar. Environ. Res. 63(4): 303-312.
- Radwan, M.A. and M.S. Mohamed (2013). Imidacloprid induced alterations in enzyme activities and energy reserves of the land snail. Helix aspersa. Ecotoxicol. Environ. Saf. 95: 91-97.
- Radwan, M.A., A.E. Essawy, N.E. Abdelmeguied, S.S. Hamed, A. Ahmed (2008). Biochemical and histochemical on the digestive gland of Eobania vermiculata snails treated with carbamate pesticides. Pestic. Biochem. Physiol. 90: 154-167.
- Radwan, M.A., K.S. El-Gendy and A.F. Gad (2020). Biomarker responses in terrestrial gastropods exposed to pollutants: A comprehensive review. Chemosphere. 257:127218.
- Ramade, F. (2003). Élément d’écologie, écologie fondamentale. 3 rd Ed., Dunod, France. 690 p.
- Ramos-Vasconcelos, G.R., L.A. Cardoso and M. Hermes-Lima (2005). Seasonal modulation of free radical metabolism in estivating land snails Helix aspersa. Comp. Biochem. Physiol. Part - C: Toxicol. Pharmacol. 140(2): 165-174.
- Regoli, F., S. Gorbi, D. Fattorini, S. Tedesco, A. Notti, N. Machella, R. Bocchett, M. Benedetti and F. Piva (2006). Use of the land snail Helix aspersa as sentinel organism for monitoring ecotoxicologic effects of urban pollution: an integrated approach. Environ. Health Perspect. 114(1): 63-69.
- Regoli, F., S. Gorbi, N. Machella, S. Tedesco, M. Benedetti, R. Bocchetti, A. Notti, D. Fattorini, F. Piva and G. Principato (2005). Pro-oxidant effects of extremely low frequency electromagnetic fields in the land snail Helix aspersa. Free Radic. Biol. Med. 39(12): 1620-1628.
- Remenieras, G. (1972). L'hydrologie de l'Ingénieur. ed., Eyrolles, Collection du centre de recherche et d’essais de chatou. France. 456 p.
- Reuner, A., F. Brümmer and R.O. Schill (2008). Heat shock proteins (Hsp70) and water content in the estivating Mediterranean Grunt Snail (Cantareus apertus). Comp. Biochem. Physiol. B, Biochem. Mol. Biol. 151(1):28-31.
- Ribeiro, S., J.P. Sousa, A.J.A. Nogueira and A.M.V.M. Soares (2001). Effect of endosulfan and parathion on energy reserves and physiological parameters of the terrestrial isopod Porcellio dilatatus. Ecotoxicol. Environ. Saf. 49(2): 131-138.
- Rosa, R., P.R. Costa, N. Bandarra and M.L. Nunes (2005). Changes in tissue biochemical composition and energy reserves associated with sexual maturation in the ommastrephid squids Illex coindetii and Todaropsis eblanae. Biol. Bull. 208: 100-113.
- Sancho, E., M.J. Villarroel, E. Andreu and M.D. Ferrando (2009). Disturbances in energy metabolism of Daphnia magna after exposure to tebuconazole. Chemosphere. 74:1171-1178.
- Seklaoui, M., A. Boutaleb, H. Benali, F. Alligui, W. Prochaska (2016). Environmental assessment of mining industry solid pollution in the mercurial district of Azzaba, northeast Algeria. Environ. Monit. Assess. 188(11):621-646.
- Shi, X., Z. Zhou, L. Wang, F. Yue and M. Wang (2012). The immunomodulation of acetylcholinesterase in zhikong scallop Chlamys farreri. PLoS ONE 7(1), e30828.
- Shibko, S., P. Koivistoinen, C. Tratyneck, A. Newhall and L. Freidman (1966). A method for the sequential quantitative separation and glycogen from a single rat liver homogenate or from a sub cellular fraction. Analyt. Biochem. 19: 415-428.
- Sifi, K. and N. Soltani (2018). Seasonal changes of two biomarkers of oxidative stress (LDH, MDA) in the edible mollusc Donax trunculus (Mollusca: Bivalvia) from the Gulf of Annaba (Algeria): correlation with carbohydrate and lipid contents. Molluscan Res. 39(1): 44-52.
- Silva, L., L. Meireles, F.O. Junqueira, J. Pinheiro and E.C. de Almeida Bessa. (2012). Seasonal changes of energy reserves in Bulimulus tenuissimus (d’Orbigny, 1835) (Mollusca, Bulimulidae). Anim. Biol. 62: 111-118.
- Singh, P., P. Kumar, V.K. Singh and D.K. Singh (2009). Effect of snail attractant pellets containing plant molluscicides on certain enzyme in the nervous tissue of Lymnaea acuminata (Lamark). The Bioscan. 4(3): 395-398.
- Snyman, R. G., A. J. Reinecke and S.A. Reinecke (2005). Quantitative damages in the digestive gland cells of the snail Helix aspersa after exposure to the fungicide copper oxychloride. Ecotoxicol. Environ. Saf. 60(1): 47-52.
- Somero, G. (2010). The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine “winners” and “losers”’. J. Exp. Biol. 213(6): 912-920.
- Somero, G.N. (1995). Proteins and temperature. Annu. Rev. Physiol. 57: 43-68.
- Staikou, A., G. Tachtatzis, K. Feidantsis and B. Michaelidis (2016). Field studies on the annual activity and the metabolic responses of a land snail population living in high altitude. Comp. Biochem. Physiol., Part AMol. Integr. Physiol.191: 1-8.
- Storey, K. B. (2002). Life in the slow lane: molecular mechanisms of estivation. Comp. Biochem. Physiol. 133A: 733-754.
- Umminger, B. L. (1977): Relation of whole blood sugar concentrations in vertebrates to standard metabolic rate. Compar. Biochem. Physiol. 56: 457-460.
- Vega, I.A., M.A. Arribere, A.V. Almonacid, G.S. Ribeiro and A. Castro-Vazquez (2012). Apple snails and their endosymbionts bioconcentrate heavy metals and uranium from contaminated drinking water. Environ. Sci. Pollut. Res. 19:3307-3316.
- Viard, B., A. Maul and J. Pihan (2004). Standarduse conditions of terrestrial gastropods in active biomonitoring of soil contamination. J. Environ. Monit. 6(2):103-107.
- Whittaker, M. (1986). Cholinesterase. Monogr Hum Genet. Basel. Karger. Switzerland. 134 p
- WIENS, A.W. and L.I. GILBERT (1967). Regulation of carbohydrate mobilization and utilization in Leucophaea maderae. J. Insect. Physiol. 13: 779-794.
- Zaidi, N. and N. Soltani (2011). Environmental risks of two chitin synthesis inhibitors on Gambusia affinis: chronic effects on growth and recovery of biological responses. Biol. Control. 59(2): 106-113.
- Zaidi, N. and N. Soltani (2010). Chronic toxicity of flucycloxuron in the mosquitofish, Gambusia affinis: acetylcholinesterase and catalase activities and pattern of recovery. Ann. Biol. Res. 1(4):210-217.
- Za-tseva, O.V. and T.V. Kuznetsova (2008). Distribution of acetylcholinesterase activity in the digestive system of the gastropod molluscs Littorina littorea and Achatina fulica. Morfologiia. 133: 55-59.
- Zeghdoudi, F., L.M. Tanjir, N. Ouali, I. Haddidi and M. Rachedi (2019). Concentrations of trace-metal elements in the superficial sediment and the marine magnophyte, Posidonia oceanica (L) Delile, 1813 from the Gulf of Skikda (Mediterranean coast, East of Algeria). Cah. Biol. Mar. 60: 223-233.
- Zhang, X., F. Yang, X. Zhang, Y. Xu, T. Liao, S. Song and J. Wang (2008). Induction of hepatic enzymes and oxidative stress in Chinese rare minnow (Gobiocypris rarus) exposed to waterborne hexabromocyclododecane (HBCDD). Aquat. Toxicol. 86: 4-11.
|
|
|
|
|





