|
Volume 32, No. (4), 2022, August
(Impact Factor 0.570 ; JCR 2022) |
|
ANTIFUNGAL ACTIVITY AND PHYTOCHEMICAL PROFILE OF CHLOROFORM SOLUBLE FRACTION OF DATURA METEL FRUIT
N. Jabeen1,2, *A. Javaid1 and E. Ahmed3
1Department of Plant Pathology, Faculty of Agricultural Sciences, University of the Punjab, Quaid-i-Azam Campus, Lahore 54590, Pakistan
2School of Botany, Minhaj University Lahore, Pakistan
3 School of Chemistry, University of the Punjab, Quaid-i-Azam Campus, Lahore 54590, Pakistan
*Corresponding author’s email: drnadia.bot@mul.edu.pk
ABSTRACT
Datura metel is a weed of family Solanaceae. In the present study, dried and powdered fruits of this medicinal plant species were extracted in methanol for two weeks. After evaporating the solvent, the remaining gummy extract was mixed in water and fractionated using n-hexane to separate the non-polar fraction. The remaining aqueous phase was partitioned with chloroform in a separating funnel. The chloroform fraction was separated and evaporated on a rotary evaporator. The gummy biomass of this fraction obtained after complete evaporation of the solvent was dissolved in dimethylsulphoxide (DMSO) and its different concentrations (3.125 to 200 mg/ml) were prepared in malt extract broth. Antifungal activity of the extract was checked against a highly destructive soil-borne phytopathogen Sclerotium rolfsii isolated from bell pepper plants suffering from collar rot disease. Results revealed that there was 35–51% reduction in biomass of S. rolfsii due to different concentrations of the chloroform fraction. Twelve compounds were identified through GC-MS analysis of this fraction. The predominant were 1-hexacosanol (15.45%), 1,2-benzenedicarboxylic acid, bis (2-methylpropyl) ester (14.15%), 1-octadecanol (12.81%), 1-octadecene (12.80%) and 1-eicosanol (8.12%), which could be responsible for antifungal activity against S. rolfsii.
Keywords: Bell pepper, Datura metel fruit, methanolic extract, natural fungicides, Sclerotium rolfii.
INTRODUCTION
Datura metel L., a herb of family Solanaceae, is known for its numerous biological activities (Su et al., 2017). It grows as a wild plant in warmer regions all over the world. Having origin in Tropical America, the plant is also cultivated in various temperate and tropical regions (Mai et al., 2017). The plant has been used in folk medicines for centuries to treat pain, insanity, cough, convulsion convulsions, asthma, and rheumatism (Fu et al., 2017). Presence of tropane class of alkaloids in D. metel makes this plant to be used as mydriatic, anti-spasmodic and sedative agent (Nuhu, 2002). Main chemical constituents of D. metel are withanolide-type steroids (Yang et al., 2010). Furthermore, some amide alkaloids and megastigmane sesquiterpenes have also been isolated from this plant (Kuang et al., 2008; Yang et al., 2010). The plant is known to have insecticidal activity against cotton bollworm Helicoverpa armigera (Singh and Singh, 2008), herbicidal activity against Parthenium hysterophorus and Phalaris minor (Javaid et al., 2010), antifungal activity against Macrophomina phaseolina and Colletotrichum gloeosporioides (Javaid and Saddique, 2011; Karim et al., 2017), antibacterial activity against Bacillus cereus, Escherichia coli and Klebsiella pneumoniae (Akharaiyi, 2011), antioxidant activity (Akharaiyi, 2011), and anticancer activity (Pan et al., 2007).
Sclerotium rolfsii is a serious soil-borne plant pathogen with extensive host range. Above 500 plant species belonging to about 100 families are susceptible to this pathogen (Deepthi and Reddy, 2013). Mostly members of Leguminoseae, Cucurbitaceae, Solanaceae and Brassicaceae families are host of this fungus (Wavare et al., 2017; Sharf et al., 2021). A number of strategies are being used to control this fungal pathogen including application of fungicides (Augusto et al., 2010; Khan and Javaid, 2015). However, this strategy is undesirable especially in food crops like bell pepper because of ill effects of synthetic fungicides on environment and health risks associated with the application of these agrochemicals (Westlund et al., 2018). Many recent studies have shown that extracts and pure compounds of plant species namely Coronopus didymus, Chenopodium album, Chenopodium quinoa, Acacia nilotica and Melia azedarach can be used as alternatives to synthetic fungicides for the control of S. rolfsii (Javaid and Iqbal, 2014; Sana et al., 2016, 2017; Ali et al. 2020) and other fungal pathogens such as Macrophomina phaseolina (Khan and Javaid, 2020a; Banaras et al., 2021). However, studies regarding antifungal activity of D. metel fruit especially against S. rolfsii are rare. Therefore, the present study was carried out to investigate antifungal activity of chloroform fraction of methanolic fruit extract of D. metel against S. rolfsii and the identification of possible antifungal constituents through GC-MS analysis.
MATERIALS AND METHODS
Preparation of extract: Fruits of D. metel (5 kg) were collected from Lahore, Pakistan, dried and crushed, and dipped in methanol (10 l) for 10 days. After that, material was passed through doubled layered muslin cloth. Residues were re-extracted with methanol and filtered through a double layered muslin cloth again followed by filtration by a filter paper sheet. The solvent was evaporated under reduced pressure on a rotary evaporator at 45 °C to yield crude methanolic extract of D. metel fruit. Crude gummy methanolic extract was added to 300 ml distilled water and shaked well to homogenized the mixture. The mixture was transferred to a separating funnel and extracted successively with n-hexane (500 ml × 5) followed by chloroform (500 ml). Thereafter, chloroform was evaporated under vacuum in a rotary evaporator and 19.9 g of the chloroform sub-fraction were obtained (Javaid et al., 2018). Chloroform fraction was selected on the basis of various previous studies where this fraction showed more antifungal activity as compared to n-hexane sub-fraction (Banaras et al., 2020; Khan et al., 2021).
Antifungal bioassay: Antifungal activity of chloroform sub-fraction was investigated in vitro against S. rolfsii. The fungus was isolated on malt extract agar medium from a bell pepper plant suffering from collar rot disease. The isolated fungus was identified on morphological basis especially on the basis of pattern of mycelial spreading and sclerotia formation. Koch’s postulates confirmed the pathogenicity of the fungus.
For antifungal bioassays, 1.2 g of the chloroform sub-fraction was dissolve in 0.5 ml of DMSO followed by addition of 5.5 ml malt extract broth to prepare a 200 mg/ml solution. This stock solution was serially double diluted to make lower concentrations of 100, 50, 25, 12.50, 6.25 and 3.125 by adding malt extract broth. For control, 0.5 ml of DMSO were mixed with 5.5 ml malt extract broth and serially double diluted to get various concentrations of DMSO in control corresponding to various concentrations of DMSO in the extract treatments. Each treatment was replicated three times with 1 ml medium in each 10-ml volume test tube. Tubes were inoculated by taking standard droplets of 10 µl from fungal suspension. Test tubes were incubated for seven days at 25 °C. After seven days, fungal biomass was filtered, dried and weighed (Khan and Javaid, 2020b).
Percentage decrease in fungal biomass in each treatment over corresponding control treatment was calculated by applying the following formula:
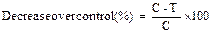
Where C and T represent fungal biomass in control and extract treatments, respectively.
GC-MS analysis: Chloroform sub-fraction was dissolved in chloroform to make it diluted, and filtered. The filtrates were subjected to GC-MS analysis for identification of the chemical constituents present in it. GC-MS was conducted on Shimadzu GC-2010 plus system coupled with an auto injector AOC-20i, an auto sampler AOC-20s and a gas chromatograph using helium as a carrier gas.
Statistical analysis: Data regarding antifungal activity of the chloroform sub-fraction was presented as means ± standard errors of three replicates. The data were analyzed by one-way analysis of variance (ANOVA) followed by LSD test for significance of difference in treatment means (P≤0.05). Computer software Statistix 8.1 was used for data analysis.
RESULTS AND DISCUSSION
Different concentrations of chloroform sub-fraction of fruit extract exhibited highly pronounced adverse effect on fungal growth causing 36–50% reduction in biomass of S. rolfsii. (Fig. 1). Previous studies mostly showed antifungal activity of other parts of D. metel such as leaves, roots, seeds and flowers. Rinez et al. (2013) recorded 69% in radial growth of Tichoderma viride due to aqueous flower extract and 24–76% suppression in growth of Fusarium oxysporum f. sp. melonis due to various organic solvent leaf extracts of D. metel. A 1.5% seed extract of D. metel reduced the growth of Colletotrichum gloeosporioides by 80% (Karim et al., 2017). Similarly, ethanolic extract of D. metel leaves caused 100% inhibition in spore germination of Colletotrichum camelliae, Botryodiplodia theobromae and Pestalotiopsis theae (Saha et al., 2005).
Twelve compounds were identified in the chloroform fraction of methanolic fruit extract of D. metel. Molecular weight, molecular formulae and peak areas of these compounds are illustrated in Table 1 and their antifungal activities, if any, are presented in Table 2. The most abundant compound was 1-hexacosanol (15.45%) followed by 1,2-benzenedicarboxylic acid, bis (2-methylpropyl) ester (14.15%), 1-octadecanol (12.81%),1-octadecene (12.80%), 1-eicosanol (8.12%), 1-heptadecene (7.90%), 1,3(15),10-bisabolatriene (7.61%),1,6,10- farnesatrien-3-ol (7.65%), 1-pentadecanol (5.80%), 1-pentadecene (2.68%), 1-tetradecanol (2.61%) and 1-dococene (1.10%). Compounds identified in the chloroform soluble fraction of methanolic fruit extract generally belonged to alcohols, alkenes and esters. According to Tuney et al. (2006) antimicrobial activity of plants extracts may be due to volatile compounds such as terpenoid, volatile fatty acids, phenols, phytol (acyclic diterpene alcohol), alkenes and esters. Similarly, Kim and Park (2012) found significant antifungal activity of alcohols (citronellol and geraniol) isolated from Acorus gramineus against Phytophthora cactorum, Fusarium circinatum and Cryponectria parasitica. Manohar (2015) identified 21 bioactive components from Geodorum densiflorum through GC-MS where alcohols and esters had antimicrobial activity. Kolawole et al. (2015) identified 78 organic compounds in Senna alata through GC-MS and found that aldehydes, alkenes, fatty alcohols, acetic acid, ketones and esters had good antifungal activity. Pohl et al. (2011) reported that alkenes and fatty acids have antifungal activity. Alkenes inhibited the myrisoylation of proteins of cell membrane. They also inhibited b-oxidation, triacylglycerol and sphingolipid synthesis and topoisomerase activity. Kiruthika and Sornaraj (2011) identified the alkenes in the D. metel flower through GC-MS having antimicrobial activity.
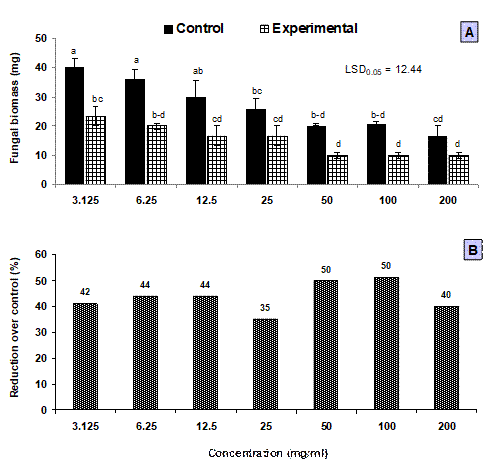
Fig. 1. Effect of chloroform sub-fraction of methanolic fruit extract of Datura metel on in vitro growth of Sclerotium rolfsii. A: Effect of chloroform sub-fraction on fungal biomass, B: Percentage inhibition in fungal biomass over corresponding control treatments due to different concentrations of chloroform sub-fraction. Vertical bars show standard errors of means of three replicates. Values with different letters at their top show significant difference (P≤0.05) as determined by LSD Test.
The most abundant compound in the present study was 1-hexacosanol. In previous studies, it also exhibited antimicrobial activity against many bacteria and yeast (Castilho et al., 2012). Apart from its antifungal activity, this compound is also known to exhibit a variety of biological activities. Mosquito larvicidal activity of an insect repellent plant Chromolaena odorata is reported to be due to this compound (Gade et al.,2017). This compound has also been isolated from Euphorbia peplus and is expected to be one of the major compounds in n-hexane fraction of methanolic extract having leishmanicidal activity (Amin et al., 2017).
The second most predominant compound found in the present study was 1,2-benzenedicarboxylic acid bis (2-methylpropyl) ester. Previously, this compound has been identified in diverse groups of organisms with a number of important properties. Klusaite et al. (2016) isolated this compound from bacterial strain 1410WF1-TSA30-2 and found that it had antibacterial activity against Gram- positive strains. Dong et al. (2016) identified it from an algal species Cladophora fracta and reported its algicidal property against Gymnodinium breve and Heterosigma akashiwo. It was among the dominant compounds of a flowering plant Achillea pachycephala, collected from Iran (Rahimmalek et al., 2012). This compound has also been reported from seaweed Ulva pertusa with allelopathic activity against Gymnodinium breve (Wang et al., 2008).
1,6,10-Farnesatrien-3-ol, also known as nerolidol, is a sesquiterpene alcohol naturally occurring in essential oils of many plants and flowers including lemon grass, tea tree, ginger, jasmine, neroli and others (Kaiser, 1993). Apart from exhibiting a number of other biological activities, it also possesses antifungal activity against a variety of fungal species infecting both human and plants (Chan et al., 2016). It exhibited fungicidal activity against Candida albicans (Curvelo et al., 2014). In a similar study, it stopped the growth of Trichophyton mentagrophytes at a 0.4 mg/ml concentration (Park et al., 2009). Lee et al. (2007) described strong antifungal effects of this compound against Microsporum gypseum causing dermatophytosis, an infection of keratinized tissues such as hair, nail and skin. The compound is present in cis- and trans- forms. Trans-nerolidol, isolated from essential oil of Lantana radula showed strong fungistatic activity against a plant pathogenic fungus Corynespora cassiicola (Passos et al., 2012). Likewise, essential oil of Piper chaba with Trans-nerolidol as main constituent showed antifungal activity against numerous phytopathogenic fungi namely Fusarium solani, F. oxysporum, Rhizoctonia solani, Colletotrichum capsici and Phytophthora capsici (Rahman et al., 2011).
Table 1. Structures of various constituents isolated from chloroform fraction of methanolic fruit extract of D. metel.
|
Sr. #
|
Names of the compounds
|
Molecular
Formula
|
Molecular
Weight
|
Peak
Area(%)
|
Structures
|
|
1
|
1-Pentadecanol
|
C15H32O
|
228
|
5.80
|

|
|
2
|
1-Dococene
|
C22H44
|
308
|
1.10
|
|
|
3
|
1-Heptadecene
|
C17H34
|
238
|
7.90
|
|
|
4
|
1-Octadecanol
|
C18H36
|
252
|
12.81
|
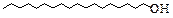
|
|
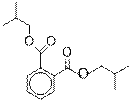
5
|
1,2-Benzenedicarboxylic acid, bis (2-methylpropyl) ester
|
C16H22O4
|
278
|
14.15
|
|
|
6
|
1-Pentadecene
|
C15H30
|
210
|
2.68
|
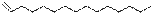
|
|
7
|
1-Tetradecanol
|
C14H30O
|
214
|
2.61
|
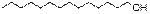
|
|
8
|
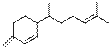
1,3(15),10-Bisabolatriene
|
C15H24
|
204
|
7.61
|
|
|
9
|
1-Octadecene
|
C18H36
|
252
|
12.80
|
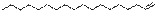
|
|
10
|
1-Eicosanol
|
C20H42O
|
298
|
8.12
|

|
|
11
|
1-Hexacosanol
|
C26H54O
|
382
|
15.45
|

|
|
12
|
1,6,10- Farnesatrien-3-ol
|
C15H26O
|
222
|
7.65
|
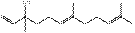
|
Table 2. Nature and properties of compounds identified from chloroform fraction of methanolic fruit extract of Datura metel through GC-MS analysis.
|
Comp. No.
|
Names of compounds
|
Nature
|
Property
|
Reference
|
|
1
|
1-Pentadecanol
|
Fatty alcohol
|
Antimicrobial
|
Kubo et al. (1994)
|
|
2
|
1-Docosene
|
Alkene
|
-
|
-
|
|
3
|
1-Heptadecene
|
Alkene
|
-
|
-
|
|
4
|
1-Octadecanol
|
Fatty alcohol
|
Antibacterial
|
Al-Hakami et al. (2013)
|
|
5
|
1,2-Benzenedicarboxylic acid, bis (2-methylpropyl) ester
|
Ester
|
Antibacterial, algicidal, allelopathic
|
Wang et al. (2008); Dong et al. (2016); Klusaite et al. (2016)
|
|
6
|
1-Pentadecene
|
Alkene
|
-
|
-
|
|
7
|
1-Tetradecanol
|
Fatty alcohol
|
Therapeutic
|
Hasturk et al. (2009)
|
|
8
|
1,3(15),10-Bisabolatriene
|
Sesquiterpene
|
Antibacterial
|
Sharma et al. (2016)
|
|
9
|
1-Octadecene
|
Alkene
|
Antioxidant
|
Adeosun et al. (2013)
|
|
10
|
1-Eicosanol
|
Alcohol
|
-
|
-
|
|
11
|
1-Hexacosanol
|
Ceryl alcohol
|
Antimicrobial,
larvicidal, leishmanicidal
|
Castilho et al. (2012); Amin et al. (2017); Gade et al. (2017)
|
|
12
|
1,6,10- Farnesatrien-3-ol
|
Sesquiterpene alcohol
|
Antifungal, antibacterial,
|
Chan et al. (2016)
|
Conclusion: The present study concludes that chloroform fraction of methanolic fruit extract of D. metel exhibited antifungal activity against S. rolfsii was possibly because of presence of 1,2-benzenedicarboxylic acid bis (2-methylpropyl) ester; 1,6,10- farnesatrien-3-ol; and 1-hexacosanol. This study will be helpful in preparation of natural products-based fungicides for the control of S. rolfsii by using the structures of potential antifungal compounds as analogues.
REFERENCES
- Adeosun, C.B., S. Olaseinde, A.O. Opeifa and O. Atolani (2013). Essential oil from the stem bark of Cordia sebestena scavenges free radicals. J. Acute Med. 3: 138-141.
- Akharaiyi, F.C. (2011). Antibacterial, phytochemical and antioxidant activities of Datura metel. Int. J. Pharm Technol. Res. 3: 478-483.
- Al-Hakami S.M., A.B. Khalil, T. Laoui and M.A. Atieh (2013). Fast disinfection of Escherichia coli bacteria using carbon nanotubes interaction with microwave radiation. Bioinorg. Chem. Appl. 2013: Article 458943.
- Ali, A., A. Javaid, A. Shoaib and I.H. Khan (2020). Effect of soil amendment with Chenopodium album dry biomass and two Trichoderma species on growth of chickpea var. Noor 2009 in Sclerotium rolfsii contaminated soil. Egypt. J. Biol. Pest Control 30: Article 102.
- Augusto, J., T.B. Brenneman, A.K. Culbreath and P. Sumner (2010). Night spraying peanut fungicides I. Extended fungicide residual and integrated disease management. Plant Dis. 94: 676-682.
- Banaras, S., A. Javaid and I.H. Khan (2020). Potential antifungal constituents of Sonchus oleraceous against Macrophomina phaseolina. Int. J. Agric. Biol. 24: 1376-1382.
- Banaras, S., A. Javaid and I.H. Khan (2021). Bioassays guided fractionation of Ageratum conyzoides extract for the identification of natural antifungal compounds against Macrophomina phaseolina. Int. J. Agric. Biol. 25: 761-767.
- Castilho, P.C., S. Savluchinske-Feio, T.S. Weinhold and S.C. Gouveia (2012). Evaluation of the antimicrobial and antioxidant activities of essential oils, extracts and their main components from oregano from Madeira Island, Portugal. Food Control. 23: 552-558.
- Chan, W.K., L.T. Tan, K.G. Chan, L.H. Lee and B.H. Goh (2016).Nerolidol: a sesquiterpene alcohol with multi-faceted pharmacological and biological activities. Molecules. 21: Article 529.
- Curvelo, J.A.R., A.M. Marques, A.L.S. Barreto, M.T.V. Romanos, M.B. Portela and M.A.C. Kaplan (2014). Soares. A novel nerolidol-rich essential oil from Piper claussenianum modulates Candida albicans biofilm. J. Med. Microbiol. 63: 697-702.
- Deepthi, K.C. and N.P.E. Reddy (2013). Stem rot disease of groundnut (Arachis hypogaeal.) induced by Sclerotium rolfsii and its management. Int. J. Life Sci. Biotechnol. Pharmacol. 2: 26-38.
- Dong, S.J., X.D. Bi, N. Wang, L. Song, W. Dai and S.L. Zhang (2016). Algicidal activities of Cladophora fracta on red tide-forming microalgae Heterosigma akashiwo and Gymnodinium breve. Allelopathy J. 37: 231-240.
- Fu, Y.H., Z.H. Si, P.M. Li, M.H. Li, H. Zhao, L. Jiang, Y.X. Xing, W. Hong, L.Y. Rua andJ.S. Wang (2017). Acute psychoactive and toxic effects of Datura metel on mice explained by 1H NMR based metabolomics approach. Metabolic Brain Dis. 32:1295-1309.
- Husturk, H., E. Goguet-Surmenian, A. Blackwood, C. Andry and A. Kantarci (2009). 1-Tetradecanol complex: therapeutic actions in experimental periodontitis. J. Periodontol. 80: 1103-1113.
- Javaid, A., S. Shafique and S. Shafique (2008). Herbicidal activity of Datura metel against Phalaris minor. Pak. J. Weed Sci. Res. 14: 209-220.
- Javaid, A., S. Shafique and S. Shafique (2010). Herbicidal effects of extracts and residue incorporation of Datura metel against parthenium weed. Nat. Prod. Res. 24: 1426-1437.
- Javaid, A. and A. Saddique (2011). Management of Macrophomina root rot of mungbean using dry leaves manure of Datura metel as soil amendment. Span. J. Agric. Res. 9: 901-905.
- Javaid, A. and D. Iqbal (2014). Management of collar rot of bell pepper (Capsicum annuum L.) by extracts and dry biomass of Coronopus didymus shoot. Biol. Agric. Hort. 30: 164-172.
- Javaid, A., L. Afzal and A. Shoaib (2017). Antifungal potential of a brassicaceous weed Sisymbrium irio against Macrophomina phaseolina. Planta Daninha. 35: Article e017164280.
- Javaid, A., G.R. Shahzad, N. Akhtar and G. Ahmed (2018). Alternaria leaf spot disease of broccoli in Pakistan and management of the pathogen by leaf extract of Syzygium cumini. Pak. J. Bot. 50: 1607-1614.
- Kaiser, R. (1993). The Scent of Orchids. Elsevier. pp. 199-200.
- Karim, M., K. Jabeen, S. Iqbal andA. Javaid (2017). Bioefficacy of a common weed Datura metel against Colletotrichum gloeosporioides. Planta Daninha. 35: Article I e017164676.
- Khan, I.H. and A. Javaid (2015). Chemical control of collar rot disease of chickpea. Pak. J. Phytopathol. 27: 61-68.
- Khan, I.H. and A. Javaid (2020a). Comparative antifungal potential of stem extracts of four quinoa varieties against Macrophomina phaseolina. Int. J. Agric. Biol. 24: 441-446.
- Khan, I.H. and A. Javaid (2020b). Antifungal activity and GC-MS analysis of n-butanol extract of quinoaleaves. Bangladesh J. Bot. 49: 1045-1051.
- Khan, I.H., A. Javaid and S.F. Naqvi (2021). Molecular characterization of Penicillium expansum isolated from grapes and its management by leaf extract of Chenopodium murale. Int. J. Phytopathol. 10: 29-35
- Kim, E. and I.K. Park (2012). Fumigant antifungal activity of Myrtaceae essential oils and constituents from Leptospermum petersonii against three Aspergillus species. Molecules.17: 10459-10469.
- Kiruthika, K.A. and R. Sornaraj (2011). Screening of bioactive components of the flower Datura metel using the GC-MS technology. Int. J. Pharm. Technol Res. 3: 2025-2028.
- Klusaite, A., V. Vickackaite, B. Vaitkeviciene, D. Bukelskis, Kieraite-Aleksandrova and N. Kuisiene (2016). Characterization of antimicrobial activity of culturable bacteria isolated from Krubera-Voronja Cave. Int. J. Speleol. 45(3): 275-287.
- Kolawole, R., B. Thomas, J. Folorunso and A. Oluwadun (2015). GC-MS analysis and antifungal activity of Senna alata Linn. Int. J. Microbiol Res. 6: 100-107.
- Kuang, H.X., B.Y Yang., Y.G. Xia and W.S. Feng (2008). Chemical constituents from the flower of Datura metel L. Arch. Pharmacol Res. 31(9): 1094-1097.
- Kubo, I., H. Muroi and A. Kubo (1994). Naturally occurring antiacne agents. J. Nat. Prod. 57: 9-17.
- Lee, S.J., J.I. Han, G.S. Lee, M.J. Park, I.G. Choi, K.J. Na and E.B. Jeung (2007). Antifungal effect of eugenol and nerolidol against Microsporum gypseum in a guinea pig model. Biol. Pharmaceut. Bull. 30 (1): 184-188.
- Mai, N.T., N.T. Cuc, H.L.T. Anh, N.X. Nhiem, B.H. Tai and C.V Minh (2017). Steroidal saponins from Datura metel. Steroids. 121: 1-9.
- Manohar, K. (2015). Bioactive compound evaluation of ethanol extract from Geodorum densiflorum Lam. by GC-MS analysis. Int. J. Pharmacol. Res. 5:139-144.
- Nuhu, H. (2002). Alakaloid content of the leaves of three Nigerian Datura species. Nigerian J. Nat. Prod. Med. 6(1): 15-18.
- Pan, Y., X. Wang and X Hu (2007). Cytotoxic withanolides from the flowers of Datura metel. J. Nat. Prod. 70: 1127-1132.
- Park, M.J., K.S. Gwak, I. Yang, K.W. Kim, E.B. Jeung, J.W. Chang and I.G. Choi (2009). Effect of citral, eugenol, nerolidol and α-terpineol on the ultrastructural changes of Trichophyton mentagrophytes. Fitoterapia. 80: 290-296.
- Passos, J.L., L.C.A. Barbosa, A.J. Demuner, E.S. Alvarenga, C.M.D. Silva and R.W. Barreto (2012). Chemical characterization of volatile compounds of Lantana camara L. and L. radula Sw. and their antifungal activity. Molecules.17:11447-11455.
- Pohl, C.H., J.L.Kock andV.S. Thibane (2011). Antifungal free fatty acids: a review. Science against microbial pathogens: current research and technological advances. Formatex. 3: 61-71.
- Rahimmalek, M., S.A.H. Goli, N. Etemadi and S. Gharbi (2012). Essential oil and total phenolic compounds variation in Achillea pachycephala collected from different regions of Iran. J. Essent. Oil Bear. Plant. 15: 217-226
- Rahman, A., S. Al-Reza and S. Kang (2011). Antifungal activity of essential oil and extracts of Piper chaba Hunter against phytopathogenic fungi. J. Am Oil Chem. Soc. 88: 573-579.
- Rinez, A., M. Daami-Remadi, A. Ladhari, F. Omezzine, I. Rinez and R. Haouala (2013). Antifungal activity of Datura metel L. organic and aqueous extracts on some pathogenic and antagonistic fungi. Afr. J. Microbiol. Res. 7: 1605-1612.
- Saha, D., S. Dasgupta and A. Saha (2005). Antifungal activity of some plant extracts against fungal pathogens of tea (Camellia sinensis). Pharmaceut. Biol. 43: 87-91.
- Sana, N., A. Shoaib and A. Javaid (2016). Antifungal potential of leaf extracts of leguminous trees against Sclerotium rolfsii. Afr. J. Trad. Complement. Altern. Med. 13: 54-60.
- Sana, N., A. Javaid and A. Shoaib (2017). Antifungal activity of methanolic leaf extracts of allelopathic trees against Sclerotium rolfsii. Bangladesh. J. Bot. 46: 987-993.
- Sharf, W., A. Javaid, A. Shoaib and I.H. Khan (2021). Induction of resistance in chili against Sclerotium rolfsii by plant growth promoting rhizobacteria and Anagallis arvensis. Egypt. J. Biol. Pest Control. 31: Article 16.
- Singh, V. and R. Singh (2008). Effect of Datura metel seed methanol extract and itsfractions on the biology and ovipositional behaviour of Helicoverpa armigera. J. Med. Aromat. Plant Sci. 30:157-163.
- Sharma, P., G.C. Shah, R. Sharma and P. Dhyani (2016). Chemical composition and antibacterial activity of essential oil of Nepeta graciliflora Benth. (Lamiaceae). Nat. Prod. Res. 30:1332-1334.
- Su, Y., Q. Wang, B. Yang, L. Wu, G. Cheng and H. Kuang (2017). Withasteroid B from Datura metel L. regulates immune responses by modulating the JAK/STAT pathway and the IL-17+RORγt+/IL-10+FoxP3+ ratio. Clin. Exp. Immunol. 190: 40-53.
- Tuney, I., B.H. Cadirci, D. Unal and A. Sukatar (2006). Antimicrobial activities of the extracts of marine algae from the coast of Urla Izmir, Turkey. Turk. J. Biol. 30: 171-175.
- Wavare, S.H., R.M. Gade and A.V. Shitole (2017). Effect of plant extracts, bio agents and fungicides against Sclerotiumrolfsiicausing collar rot in chickpea. Indian J. Pharmaceut. Sci. 79: 513-520.
- Wang, Z.Y., Z.J. Tian, F.M. Li, Z. An and H.Y. Hu. (2008). Allelopathic effects of large seaweeds on red tide dinoflagellate Gymnodinium breve. Allelopathy J. 22: 181-187.
- Westlund, P., D. Nasuhoglu, S. Isazadeh and V. Yargeau (2018). Investigation of acute and chronic toxicity trends of pesticides using high-throughput bioluminescence assay based on the test organism Vibrio fischeri. Arch. Environ. Contam. Toxicol. 74(4): 557-567.
- Yang, B.Y., Y.G. Xia, Q.H. Wang, D.Q. Dou and H.X. Kuang (2010). Four new withanolide glucosides from the flower of Datura metel L. Arch. Pharm. Res. 33: 1143-1148.
|
|
|
|
|





