ROLE OF HERBS AS ANTHELMINTIC IN THE CONTROL OF PARASCARIASIS IN EQUINES
Z. A. Beg1, N. Roohi1, Z. Iqbal2* , M.A. Iqbal1 and A. Zulfiqar3
1Department of Zoology, University of the Punjab, Lahore, Pakistan2
2Department of Animal Production, Riphah College of Veterinary Sciences, Riphah International University, Lahore, Pakistan
3Rawalpindi Medical University, Rawalpindi
*Corresponding Author’s email: dr.zahid@riphah.edu.pk
ABSTRACT
Parascariasis is the most common disease of horses, which needs strict control to prevent losses in the equine industry. The present study was conducted to evaluate the effectiveness of locally accessible herbal plants, as natural remedies, in managing Parascaris equorum infection relative to a most efficacious but expensive allopathic medicine Piperazine Adipate (PA). One hundred and ten horses naturally infected with Parascaris equorum were randomly allocated to five treatment groups each having ten animals. The treatments included aqueous extracts of three herbal drugs that is Nigella sativa (N. sativa) seeds, Fumaria parviflora (F. parviflora ) leave, Flemingia macrophylla (F. macrophylla) leaves, positive control with PA and untreated negative control. Plant extracts were administered through a nasogastric tube, on day 0 and 18 at a dose rate of 0.05, 0.1 and 0.15 g/kg body weight (BW) of horses. Piperazine adipate was given at a dose rate of 0.113 g/kg BW. All of the herbal extracts, at a dosage of 0.15 g/kg BW, showed increased efficacy as indicated by a significant (p<0.01) reduction in eggs per gram (EPG) of faecal samples on day 18 of the first treatment. Further reduction (p<0.01) in EPG was noticed on day 28 of the 2nd treatment. Efficacy was highest for N. sativa on the 28th-day post-treatment which was 86.08% as compared to 82.11% and 73.74% for F. parviflora and F. macrophylla, respectively. Piperazine adipate presented an efficacy of 85.94% and 100% on the 18th and 28th day of the trial, respectively. All of the herbal extracts were found to be potent anthelmintics, however, N. sativa proved to be most effective.
Keywords: Horse; Parascaris equorum; Nigella sativa; Fumaria parviflora;Flemingia macrophylla; Piperazine adipate.
https://doi.org/10.36899/JAPS.2023.1.0615
Published first online October 14, 2022
INTRODUCTION
A healthy horse population can contribute to the economic and social growth of the country. In developing or undeveloped world, equines still provide a great source of draught to the world's rural population (Binswanger, 1986). These logistic supporting animals remain exposed to multiple risk factors which adversely affect their health (Pritchard et al., 2005). Parasitism, a globally occurring problem, is the most critical of these hazards (Goncalves et al., 2002) which causes substantial economic losses to the horse industry. Symptoms of a parasitic infection begin to appear when parasites dominate the immunity of the host. Most of the equines are owned by poor farmers, and due to lack of resources, parasitic infections of the animals are not treated well in time, which causes death in most cases (Soulsby, 2012). Losses include increased management cost, low growth, and poor performance in the field of sports as well as draught. Host infected by the parasite poorly absorbs nutrients, suffers deficiency diseases, and sustains physical damage like obstruction of the intestine, allergic reaction, and even death (Morales et al., 2012).
Different types of roundworms infect horses, out of which P. equorum, causes “Parascariasis”, which has a 29% infection rate (Lyons et al., 1981). Young horses are more prone to parasitism. The infected animal faces immune deficiency, debility, and poor growth. Poor body condition score (BCS), decrease in blood cells, rough skin, and obstructive colic can occur in the parasitized animal (Goncalves et al., 2002). It is a dimorphic parasite. The size of the male is 15-28 cm, while the female is 50 cm long. The sexual stage occurs in the small intestine. The female parasite lays around 0.2-6 million eggs annually which are defecated with faeces. Even the latest diagnostic techniques and treatment regimes could not eradicate the disease in most part of the world, which necessitates more studies on parascariasis.
To combat the parasites, anti-parasitic (anthelmintic) allopathic drugs are used. These medicines are not easily available and some are even imported, hence, quite expensive that an ordinary person cannot afford these for the treatment of infected animals. This may result in worsening of the disease and consequent losses to the equine industry. Moreover, drug resistance developed by repeated deworming and over usage of these conventional allopathic medicines is making the control difficult. A survey conducted in Poland reported increased anthelmintic resistance developed in Parascaris species (Studzinska et al., 2020). Thus there is a dire need to find some locally available, cost-effective anthelmintic as a natural remedy against P. equorum, in horses (Kaplan and Vidyashankar, 2012; Canever et al., 2013).
Historically, many herbs have been used in the subcontinent to treat various diseases in humans and animals (Rupani and Chavez, 2018). These herbs have been of great importance as they can reduce the economic burden on farmers and control allergic responses (Mali and Dhake, 2011). Nigella sativa (N. sativa) is one of the most popularly known medicinal plant reported to have a large number of therapeutic properties like antibacterial, antiparasitic, antiviral, anticarcinogenic, anti inflamatory and anti diarrheal (El-Sayed et al.2019). The aqueous, ethanolic and oil extracts of N. sativa has shown strong anthelmintic action against nematodaris parasites of livestock (Zavadzkas Garza, 2022). Fumaria parviflora (F. parviflora) is also a medicinal herb found worldwide and used for gastrointestinal ailments. Its anthelmintic effects have also been described due to its phtochemical contents like alkaloids, flavonoids, glycosides, tannins and fumericine etc (Kumar et al. 2017). Flemingia macrophylla (F. macrophylla) is another natural medicinal herb having number of pharmacological properties including anti parasitic due to its various bioactive compounds (Gahlot et al. 2011) .The present study was, therefore, designed to check the curative effects of N. sativa (Kalwanji), F. parviflora (Burgi Shatara), F. macrophylla (Jantaan) plant extracts, commonly found in Pakistan, to manage the infection of Parascaris equorum in local equine fauna.
MATERIALS AND METHODS
The trial was carried out at Remount Depot Mona, Tehsil Malikwal District Mandi Bhauddin and Remount Depot Sargodha from March to September 2018. Guidelines provided by the world association for the advancement of veterinary parasitology (WAAVP) regarding animal selection and allocation, adequacy of infection and efficiency of anthelmintics were followed (Duncan et al., 2002). Initially, 200 male horses of mixed breeds having age 2-6 years were enrolled in the study. The animals were already having an identification number branded on their thighs. At the request of the authors the earmarked horses were not dewormed during the year of the experiment. Faecal samples of these horses were collected from the rectum in sterile bags and shifted to the Army College of Veterinary Sciences Sargodha laboratory under refrigeration for subsequent examination. Horses with positive faecal tests (FTs) showing a minimum EPG of 500 were marked. One hundred and ten out of these P. equorum positive horses, were selected and distributed in five major groups (1-5), for the study under a completely randomized (CR) design. Herbal treatment groups (1-3), each having 30 positive animals, were further divided into nine subgroups, with ten P. equorum positive horses, in each. Group -4 was given standard dose of PA, whereas group-5 was given no medicine and served as a control. These subgroups were randomly allocated to treatments as given in Table-1. The treatments were three herbal drugs each at three dosage levels, one conventional allopathic anthelmintic and a negative control.
The three aqueous herbal extracts used were made from N. sativa (Kalwanji), F. parviflora (Burgi Shatara) and F. macrophylla (Jantaan). Reduction in fecal egg count was used as an indicator to check the field efficacy of the three herbals as well as allopathic medicine PA as anthelmintics (Geary et al., 2012).
The above three herbal products Kalwanji seeds, Burgi Shatara and Jantaan leaves were obtained, dried out, finely ground, and placed in a sealed container at 4°C for further procedure. Their aqueous solution was prepared according to Parida et al. (2002). A 10% solution containing dried plant material and purified water with a ratio of 1:10 was prepared and placed in an incubator. The solution was filtered via muslin cloth, boiled for half an hour, and kept overnight. Following evaporation in decreased pressure, the viscid residue was collected and dipped in normal saline for dose preparation. Using the nasogastric tube (NT), the calculated dose was given to selected animals twice, that is, on days 0 and 18 of the research trial. A 5% solution of the calculated dose, i.e., 0.05, 0.1 and 0.15 g/kg body weight of each of the three herbs was given to the horses in the relevant subgroups. On days 0, 3, 7, 18, 21 and 28, the faecal egg count was performed in untreated control and treated animals. This study also observed the side effects, like allergic reactions, salivation, urticaria, shivering, diarrhoea, fever, etc., of herbal drugs through clinical observations.
For the collection of eggs, the flotation technique was used, and for counting the eggs, the McMaster chamber was used (Taylor et al. 2007). Percent efficacy for each treatment option and dosage was calculated as follows (Cabaret and Berrag, 2004).
Efficacy%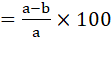
a = Pre-treatment EPG of faeces
b = Post-treatment EPG of faeces
Statistical analysis: Faecal EPG values of the three herbal drugs and control, on the 3rd, 7th, 18th, 21st, and 28th day were statistically analyzed and compared in software SPSS 20. One way ANOVA and Tukey’s Post Hoc analysis were used for multiple comparisons (Steel et al., 1997).
RESULTS
The results are presented in Table 2. Parascariasis positive horses of the control group, without any treatment, exhibited a steady increase in EPG in their faecal samples where N.sativa showed the highest reduction in EPG out of the three herbal drugs. A mild diarrhoea was noticed in F. macrophylla treated horses whereas no adverse effects were noted with N. sativa,F. parviflora as well as PA. First treatment with N. sativa, at dose rates of 0.05, 0.10 and 0.15 g/kg BW, to infected horses resulted in a consistent reduction in faecal EPG thus, indicating the enhanced efficacy of 66, 70.88 and 72.78%, respectively, at day 18. Repeated dose on day 18 resulted in further reduction in EPG with more intensified efficacy up to day 28 of the trial. A significantly pronounced reduction in EPG was observed at a dose rate of 0.15 g/kg BW. A similar dose-dependent and frequency related trend of increased efficacy and reduced faecal EPG of infected horses was also observed when treated with F. parviflora. In this case, once again, increasing dosage strength resulted in a linear increase in the effectiveness of the herb. Treatment with F. macrophylla, proved to be less effective than N. sativa and F. parviflora but still, the efficacy enhanced pronouncedly as reflected by a significant reduction in faecal EPG. Again a linear and dose-dependent enhancement of effectiveness was noticeable with a higher dose rates as well as after second dose. Piperazine adipate, at a standard dose rate of 0.113 g/kg BW, twice, i.e., on day 0 and 18 of the trial, was found to be most effective than all other therapeutic options (N. sativa, F. parviflora and F. macrophylla) with 100% effectiveness as indicated by zero faecal EPG at day 28 of the trial.
In the untreated control group, horses remained positive for the parasite eggs showing a persistent increase in faecal EPG up to day 28 of the experiment. Signs and symptoms like diarrhoea, debility, cough, and intermittent colic, of parasitic infection, were also noticeable in this group.
DISCUSSION
In the present study efficacy of treatments was recorded through a reduction in faecal egg count and presented as EPG of faecal samples. All the herbal drugs proved to be potent anthelmintic; however, N. sativa although less potent than PA, proved to be the most effective among the herbal drugs used in the trial. The efficacy of N. Sativa against P. equorum larvae has also been established recently by Rakhshandehroo et al. (2020), where in vitro use of N. Sativa in the concentration of 50, 100 and 200 mg/ mL 200 significantly (p<0.05) inactivated P. equorum larvae in 40, 20 and 10 minutes respectively. The results are also comparable with Al-Shaibani et al. (2008) who reported a reduction of 69.5% and 54.2 % respectively in faecal egg count of gastrointestinal nematodes of sheep treated with ethanolic and aqueous extracts of N. sativa at the dose of 200 mg/kg. The anti- parasitic effects shown in our results are also in match with the study of Mahmoud et al. (2002) where N. Sativa oil, administered at a dose of 2.5-5.0 ml/kg BW within two weeks, showed a significant decrease in the number of Schistosoma mansoni in the liver of mice. In a quite recent study effect of various concentrations of N. sativa oil was observed on an egg hatch assay of unidentified nematode eggs collected from sheep and highly significant reduction was reported as compared to negative control (Zavadzkas Garza, 2022).
The N. Sativa is a miraculous plant and its seeds have traditionally been used to treat all diseases. Its therapeutic properties appear mainly due to its non-volatile compounds like flavonoids, phenolic acid, tannins, and terpenes (Dalli et al., 2021a). In particular, thymoquinone contained in the essential oil of N. sativa has an anti-parasitic role (Ghedira, 2006, Ahmad et al., 2013, Dalli et al., 2021b). The pharmacological studies reveal that N. Sativa seeds contain various quinine compounds like thymoquinone, cymene, terpineol, as well as alkaloids like isoquinoline and pyrazole (Ahmad et al., 2013), which are supposed to have potent antiparasitic effects. In particular potent role of thymoquinone in inhibition of various protozoans like Theleria. equi, Babesia. bigemina and Babesia. bovis in vitro and Babeasia. microti in mice has been reported earlier (El-Sayed et al. 2019) which also substantiate our findings and make N. sativa a potential candidate for drug development against all types of parasites including P. equorum .The experimental results of F. parviflora showed its efficacy as an effective herbal anthelmintic. However, in reducing the faecal egg count, N. sativa was more effective than F. parviflora. The herbal extract of F. parviflora has an anthelmintic effect mainly due to its alkaloids such as perfume, berberine, fumarine, etc. (Kumar et al., 2017). Similar results have also been observed in research conducted on sheep (Al-Shaibani et al., 2009).
Flemingia macrophylla also showed a significant drop in EPG at higher dosage rate. This confirms that F. macrophylla is also an effective herbal anthelmintic against P. equorum infection in horses, however, N. sativa and F. parviflora, were found to be more potent anthelminitics and superior to F. macrophylla. Flemingia macrophylla is a Chinese shrub/ herb containing an isoflavone fleminiginand a number of compounds having cytotoxic, anti-inflammatory and other medicinal properties (Lai et al., 2013, Suharmiati et al., 2021).
Bio-anthelmintic activity of such herbs has also been reported due to their condensed tannin content when they were used as fodder (Niderkorn and Jayanegara, 2021). Parascaris equorum untreated infection as well in case of resistance against conventional anthelmintics can lead to clinical and subclinical intestinal damages in horses, particularly at a younger age (Peregrine et al., 2014), therefore locally available herbal drugs at appropriate dosage can be included in parasitic control management of equines, especially where stable / farm density is higher. From a therapeutic point of view, N. Sativa, F. parviflora and F. macrophylla have shown positive anti-parasitic activity in reducing worm exposure in affected horses. Further research is required to determine a more lethal dose of these herbal medicines to obtain even higher efficacy.
Conclusion: Conclusively, N. Sativa proved to be a more potent and effective herbal drug in controlling P. equorum infection in horses than F. parviflora and F. macrophylla. Additionally, no side effects of this herbal treatment were observed. Hence, it can be used as a cheaper, safer, and readily available anthelmintic remedy against parascariasis in horses. A further trial for the calculation of LC50 and LC90 of these herbal extracts as well as determination of withdrawl period, is suggested.
Acknowledgements: The authors are thankful to Remount Depot Mona and Sargodha Depot Pakistan as well as Army college of veterinary sciences authorities for permission to carry out our research at their setups and use their existing facilities.
Statement of animal rights: All applicable international, national, and institutional guidelines for the care and use of animals were followed. The experimental protocol was approved by the Animal Care and Use Committee, University of the Punjab, Lahore, vide number D/2365/ACAD.
Conflict of Interest:The authors declare that they have no conflict of interest.
Author’s contribution: All authors contributed to the study conception and design as well as manuscript writing. ZAB conducted the entire experiment and wrote the manuscript. NR supervised the research and finalized the manuscript. ZI and AZ critically reviewed and improved the manuscript. MAI helped in arranging and statistically analyzing the data.
Declaration: The present investigation is a part of the Ph.D. thesis entitled “Epidemiological, Biochemical and Therapeutic studies in control of Parascariasis in horses” submitted to the higher education commission (HEC) Pakistan by the first author of this manuscript.
REFERENCES
- Ahmad, A., A. Husain, M. Mujeeb, S. A. Khan, A. K. Najmi, N. A. Siddique, Z. A. Damanhouri. and F. Anwar (2013). A review on therapeutic potential of Nigella sativa: A miracle herb. Asian Pac. J. Trop. Biomed. 3(5):337-352.
- Al-Shaibani, I., M. Phulan, M. Shiekh, A. Arijo, A.Qureshi and A. M. Kumbher (2008). Anthelmintic activity of Nigella sativa L., seeds on gastrointestinal nematodes of sheep. Pakistan J. Nematol. 26(2):207-218.
- Al-Shaibani, I., M. Phulan and M. Shiekh (2009). Anthelmintic activity of Fumaria parviflora (Fumariaceae) against gastrointestinal nematodes of sheep. Int. J. Agric. Biol. 11:431-436.
- Binswanger, H., (1986). Agricultural mechanization: a comparative historical perspective. The World Bank Research Observer 1:27-56.
- Canever, R. J., P. R. Braga and A. Boeckh (2013). Lack of Cyathostomin sp. reduction after anthelmintic treatment in horses in Brazil. Vet. Parasitol. 194:35-39.
- Cabaret, J., and B. Berrag (2004).Faecal egg count reduction test for assessing anthelmintic efficacy: average versus individually based estimations. Parasitol. 121:105-11.
- Dalli, M., S.-e Azizi, H. Benouda, A. Azghar, M. Tahri, B. Bouammali, Maleb and N. Gseyra (2021a). Molecular composition and antibacterial effect of five essential oils extracted from Nigella sativa l. Seeds against multidrug-resistant bacteria: A comparative study. Evidence-Based Complementary and Alternative Medicine 2021.
- Dalli, M., O. Bekkouch, S. –e Azizi, A. Azghar, N. Gseyra and B. Kim (2021b). Nigella sativa Phytochemistry and pharmacological activities: A review (2019–2021). Biomolecules 12(1):20.
- Duncan, J., Abbott, J. Arundel, M. Eysker, T. Klei, R. Krecek, E. Lyons, C. Reinemeyer. and J. Slocombe. (2002). World association for the advancement of veterinary parasitology (waavp): Of guidelines for evaluating the efficacy of equine anthelmintics. Vet. Parasitol. 103(1-2):1-18.
- El-Sayed, S.A.E.S., M.A. Rizk., and N. Yokoyama (2019). Evaluation of the in vitroand in vivo inhibitory effect of thymoquinone on piroplasm parasites. Parasites Vectors, 37 (12). https://doi.org/10.1186/s13071- 019-3296-z
- Geary, T. G., C. Barry and Hosking (2012). Guideline: Anthelmintic combination products targeting nematode infection of ruminants and horses. Vet. Parasitol. 190: 306-316.
- Gahlot, K., V. Lal and S. Jha. (2011). Phytochemical and pharmacological potential of flemingia roxb. Ex wt aiton (fabaceae). Int. J. Phytomedicine 3(3):294-307.
- Ghedira, K., (2006). La nigelle cultiv’ée: Nigella sativa(ranunculaceae). Phytothérapie 4(5):220-226.
- Goncalves, .S, V. Julliand and A. Leblond (2002). Risk factors associated with colic in horses. Vet. Res. 33:641-652.
- Kaplan, R.M. and A. N. Vidyashankar (2012). An inconvenient truth: global worming and anthelmintic resistance. Vet. Parasitol. 186:70-78.
- Kumar, S., A. K. Sharma and A. Kamboj (2017). Fumaria parviflora(Fumitory): A traditional herbal medicine with modern evidence. Asian J. Pharm. Pharmacol. 3(6):200-207.
- Lai, W.C., Y.T. Tsui, A.N.B. Singab, M.El-Shazly, -C. Du, T.-L. Hwang, , C.-C.Wu, , M.H.Yen, e C.K. Le, and M.F.Hou (2013). Phyto-serm constitutes from Flemingia macrophylla. Int. j. Mol. Sci. 14(8):15578-15594
- Lyons, E., J.Drudge and T. Swerczek (1981). Prevalence of Strongylus vulgaris and Parascarisequorum in Kentucky thoroughbreds at necropsy. J. Am. Vet. Med. Assoc. 179:818-
- Mahmood, K. and M.Ashraf (2010). Pharmacokinetics of ecofriendly meloxicam in healthy horses. Pak J Sci 62:198-201.
- Mahmoud, M.R., S.El-Abhar and S.Saleh (2002). The effect of Nigella sativa oil against the liver damage induced by Schistosoma mansoni infection in mice. J. Ethnopharmacol ; 79(1): 1-11.
- Mali, R. G. and A.S. Dhake ( 2011). A review on herbal antiasthmatics. Orient. Pharm. Exp. Med. 11(2), 77-90.
- Morales, A.A., Belloh. and D.Villoria (2012). Prevalence of gastrointestinal parasites in thoroughbred horses during the quarantine period of 2012 at the racetrack La Rinconada, Caracas, Venezuela, Parasitology. 71 (2): 179-182.
- Niderkorn, V. and A.Jayanegara (2021). Opportunities offered by plant bioactive compounds to improve silage quality, animal health and product quality for sustainable ruminant production: A review. Agronomy. 11(1):86.
- Parida, M., C.Upadhyay and G. Pandya, (2002). Inhibitory potential of neem (Azadirachta indica Juss) leaves on dengue virus type-2 replication. J. Ethnopharmacol. 79:273-278.
- Peregrine, A.S., B.Molento and M. Kaplan (2014). Anthelmintic resistance in important parasites of horses: does it really matter? Vet. Parasitol. 201:1-8.
- Pritchard, J., A.Lindberg and Main (2005). Assessment of the welfare of working horses, mules and donkeys, using health and behaviour parameters. Prev. Vet. Med. 69:265-283.
- Rakhshandehroo, E., G.Anbari and M.Ghane (2020). An investigation of the anthelmintic effect of Nigella sativa extracts on the infective larva of Parascaris equorum in vitro. Vet. Res. Bio. Prod. 33:33-40.
- Rupani, R. and A.Chavez (2018). Medicinal plants with traditional use: Ethnobotany in the Indian subcontinent. Clin. Dermatol. 36(3), 306-309.
- Soulsby, E. (2012). Helminths, Arthropods and Protozoa of Domesticated Animals, 7th Edn., Baillire. Blackwell Publications.
- Studzinska, M.B., G.Salle, M. Roczen-Karczmarz (2020). A survey of ivermectin resistance in Parascaris species infected foals in south-eastern Poland. Acta. Veterinaria. Scandinavica. 62:1-5.
- Suharmiati, S., Z.A.Agustina and D.Effendi (2021). Medicinal plants knowledge and traditional healing practices of mentawai indigenous people in indonesia: An ethnomedicine approach. Indian J. Forensic Med.Toxicol. 15(3):4191.
- Steel, R.G, J. H. Torrie and D. A. Dickey (1997). Principles and Procedures of Statistics-A Biometerical Approach. McGraw‐Hill Book Company, New York,.
- Taylor, M., R.Coop and R.Wall (2007). Parasitol. Blackwell, Science (3rd Ed), UK, 1827-1828.
- Zavadzkas Garza, R. (2022). Nigella sativa oil as a Novel Anthelmintic Against Internal Nematode Parasites of Sheep. Senior Projects Spring 2022. https://digitalcommons.bard.edu/senproj_s2022/42
|
Subgroups
|
Pretreatment
EPG
|
Post treatment EPG
|
| |
Nigellasativa (Group 1)
|
P-value
Summary
|
| |
Day 0
|
Day 3
|
Day 7
|
Day 18
|
Day 21
|
Day 28
|
|
0.05g/kgBW(A)
|
679 ± 35.04
|
606.3±34.24
|
421.9±34.28
|
230.8±22.50
|
159.9±17.11
|
127.3±12.3
|
< 0.0001*
|
|
Efficacy %
|
|
10.70
|
37.86
|
66
|
76.45
|
81.25
|
|
0.1g/kgBW(B)
|
784.5± 48.84
|
716.5±46.73
|
514.9±34.48
|
228.4±15.22
|
187.5±13.71
|
148.1±12.79
|
< 0.0001*
|
|
Efficacy %
|
|
8.66
|
34.36
|
70.88
|
76.09
|
81.12
|
|
0.15g/kgBW(C)
|
758.9± 70.51
|
672.3±51.45
|
498.4±46.40
|
206.5±21.82
|
140.8±13.26
|
105.6±11.75
|
< 0.0001*
|
|
Efficacy %
|
|
11.41
|
34.32
|
72.78
|
81.44
|
86.08
|
|
Fumariaparviflora (Group 2)
|
|
0.05g/kgBW(D)
|
765.8 ± 19.21
|
701±66.20
|
612.3±68.75
|
454.2±49.42
|
334.8±48.87
|
266±49.71
|
< 0.0001*
|
|
Efficacy %
|
|
8.46
|
20.04
|
40.68
|
56.28
|
65.26
|
|
0.1g/kgBW (E)
|
750.3± 53.29
|
685.4±53.38
|
586±46.48
|
218.5±17.48
|
193.2±15.17
|
165.9±18.33
|
< 0.0001*
|
|
Efficacy %
|
|
8.6
|
21.89
|
70.87
|
74.25
|
77.88
|
|
0.15g/kgBW(F)
|
644.1 ± 69.25
|
576.2±60.05
|
476.4±52.29
|
186.8± 19.19
|
150±16.88
|
115.2±17.04
|
< 0.0001*
|
|
Efficacy %
|
|
10.54
|
26.03
|
70.99
|
76.71
|
82.11
|
|
Flemingiamacrophylla (Group 3)
|
|
0.05g/kgBW(G)
|
664.9 ± 66.10
|
599.3 ±62.35
|
514.3 ±59.61
|
327.1 ±39.19
|
236.5 ±36.61
|
227.3±34.35
|
< 0.0001*
|
|
Efficacy %
|
|
9.86
|
22.65
|
50.8
|
64.43
|
65.81
|
|
0.1g/kgBW(H)
|
670 ±56.37
|
592.3 ±45.19
|
484 ±42.00
|
278.2 ±13.35
|
237.2 ±14.16
|
191.1 ± 19.04
|
< 0.0001*
|
|
Efficacy %
|
|
11.59
|
27.76
|
58.47
|
64.59
|
71.47
|
|
0.15g/kgBW (I)
|
655 ± 46.82
|
580 ± 36.67
|
445 ± 27.34
|
255 ± 22.91
|
215 ±18.33
|
172 ± 16.85
|
< 0.0001*
|
|
Efficacy %
|
|
11.45
|
32.06
|
61.06
|
67.17
|
73.74
|
|
Piperazine adipate (Group 4)
|
|
0.113 g/kg BW
|
671.5± 68.16
|
556 ± 73.92
|
377 ± 46.59
|
94.4 ± 9.76
|
14.5 ± 7.45
|
0
|
<0.0001*
|
|
Efficacy %
|
|
17.20
|
43.85
|
85.94
|
97.84
|
100
|
|
Untreated Control (Group 5)
|
|
EPG
|
731.9± 53.24
|
753.8± 64.51
|
787.8 ± 63.22
|
816.2± 60.15
|
832.6 ±64.36
|
853.2 ±59.47
|
0.7 ns
|
|
Difference %
|
.
|
2.99↑
|
7.63↑
|
11.51↑
|
13.75↑
|
16.57↑
|
Table 1. Experimental design of therapeutic trial in Parascaris equorum positive horses (n=110).
|
Groups
|
1
|
2
|
3
|
4
|
5
|
|
Treatment
|
N. sativa
|
F. parviflora
|
F. macrophylla
|
Piperazine adipate
(n=10)
|
Control
(n=10)
|
|
Subgroups A-I
(n=10 each)
|
A
|
B
|
C
|
D
|
E
|
F
|
G
|
H
|
I
|
|
Dosage
g/kg body weight
|
0.05
|
0.1
|
0.15
|
0.05
|
0.1
|
0.15
|
0.05
|
0.1
|
0.15
|
0.113
|
No treatment
|
Table 2. Faecal EPG (Mean±SEM) in Parascaris equorum positive horses, following treatments and their effectiveness (n=10 in each trial) compared to untreated control (n=10).
↑increase, *significant at P< 0.001, ns: non-significant, EPG: eggs per gram, BW: body weight |





